CO oxidation is a widely studied catalysis reaction that is central to a number of industrial processes, including the abatement of gaseous waste in petrochemical industries, fuel production, vehicle pollution reduction and pure H2 production for proton-exchange membrane fuel cells.

Hiden Analytical CO-A. Image Credit: Hiden Analytical
It can be challenging to accurately measure CO and CO2 in N2 using a mass spectrometer (MS) because of overlapping spectral fragments resulting from gases at m/z 28.
While other fragments are available and the m/z 12 signals for CO + CO2 can be deconvoluted in combination with m/z 44 for CO2, it should be noted that m/z 12 is a relatively low-intensity fragment. Therefore, overlap removal can lead to additional noise and errors in the resulting signal.
One potential means of addressing this limitation lies in the use of a discrete CO analyzer such as the Hiden CO-A (carbon monoxide analyzer). Such an instrument can be used to unambiguously measure the CO component of the mixture alone, combining this with analysis from the mass spectrometer.
This article outlines a study into the CO oxidation reaction using a Pt/Al2O3 catalyst with 1% CO + 1 % O2 gas mix in an N2 carrier gas.
Experimental
First, a single 0.05 g pellet of 1% Pt/Al2O3 was inserted into a CATLAB microreactor from Hiden. A gas flow of 1% CO + 1% O2 + 98% N2 was passed over the sample. This was done at a total flow rate of 100 ml per minute.
Next, the CATLAB reactor’s exhaust was connected to the Hiden CO-A and HPR-20 gas analyzer. This was done using 1/8” stainless steel tubing. Once the gas flows had stabilized, the temperature was increased to 250 °C at a rate of 15 °C per minute.
Results - MS Only
The results below illustrate the result of analyzing the m/z 44 signal for CO2 (red) and the m/z 12 signal for CO + CO2 (green) using only the HPR-20.
The increases in CO2 signal on m/z 44 are clearly visible when CO oxidation occurs. The m/z 12 signal also increases, however, due to the combination of the CO + CO2 fragments present at this m/z value.
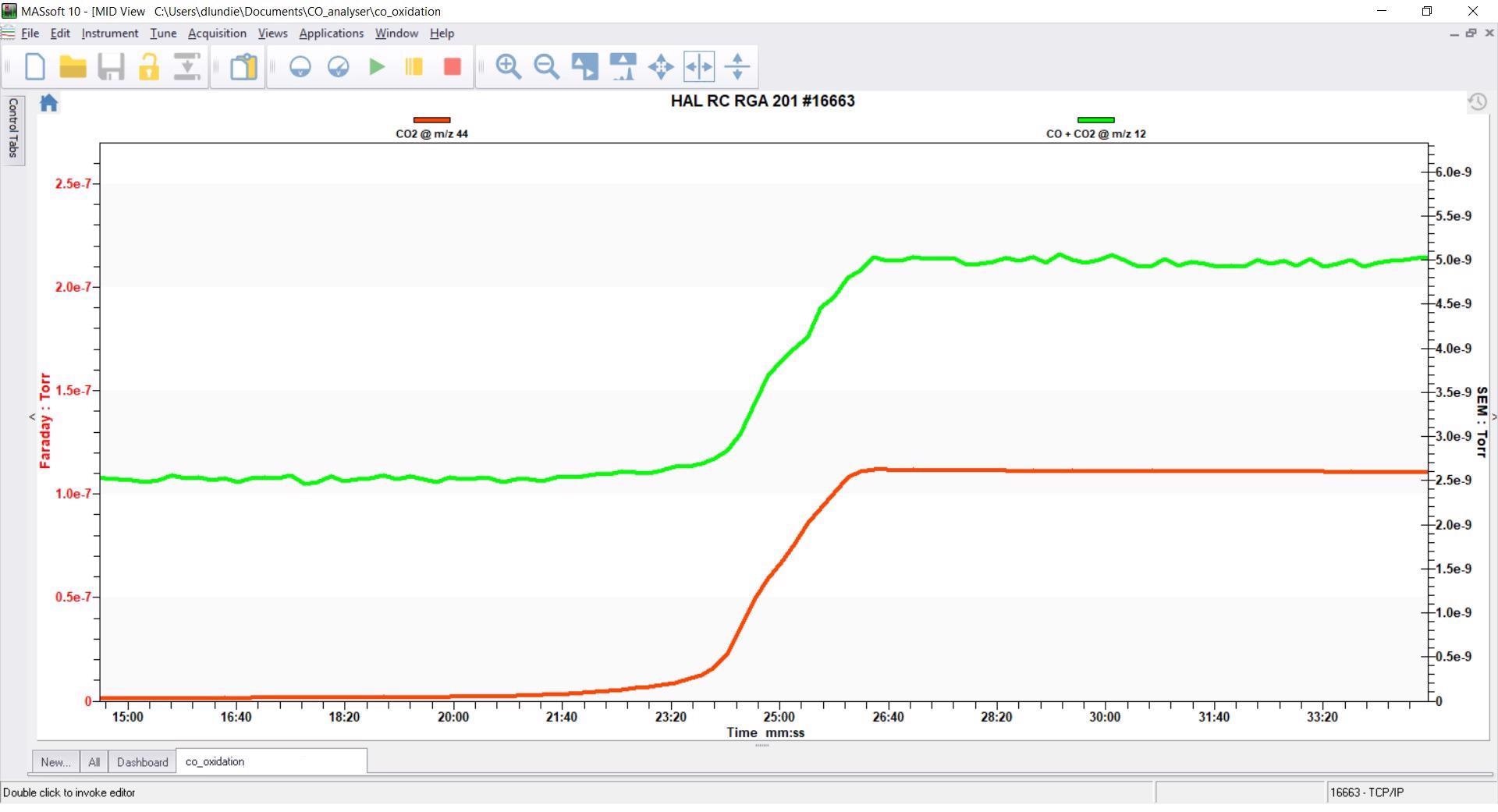
Image Credit: Hiden Analytical
Results - MS + CO-A
The results shown below were obtained by employing the MS in combination with the Hiden CO-A. The data plainly demonstrates the reaction of CO (blue) to form CO2 (red) with the complete conversion of CO with zero interference from the CO2 signal.
Spectral deconvolution represents a viable solution for specific applications, but the CO analyzer is able to eliminate uncertainty in the CO measurement – a key consideration in cases where there may be spectral interference that cannot be accounted for in deconvolution methods or for applications that involve higher amounts of CO2 and nitrogen than anticipated.
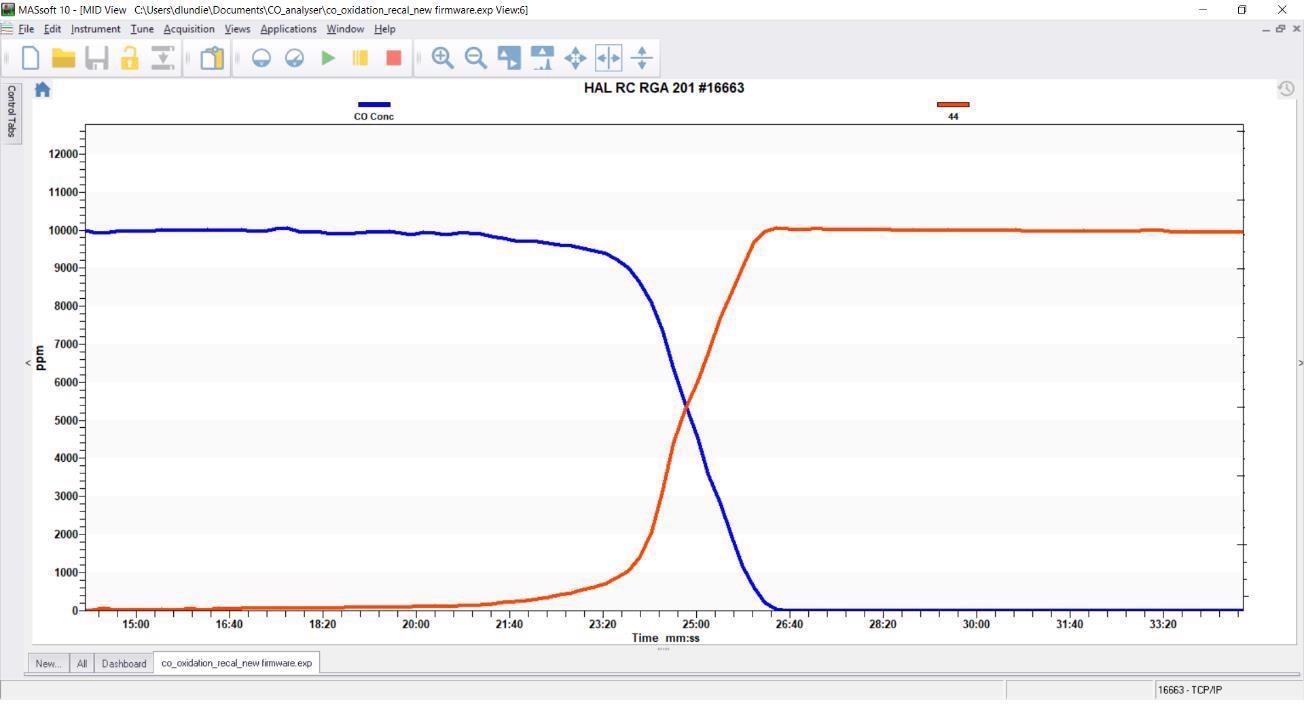
Image Credit: Hiden Analytical
Conclusion
Leveraging a combination of the Hiden CO-A and the HPR-20 affords users a means of unambiguously detecting CO in environments where standalone MS analysis may struggle, for example, CO oxidation reactions within an N2 atmosphere.
The CO-A unit is suitable for integration with any Hiden atmospheric gas analysis system, such as the ExQ, QGA, HPR-20 R&D, HPR-20 EGA and the HPR-20 TMS. It is ideally suited to applications that necessitate accurate CO measurement in the presence of overlapping species such as N2 and CO2.

This information has been sourced, reviewed and adapted from materials provided by Hiden Analytical.
For more information on this source, please visit Hiden Analytical.