When exposed to short-range adsorption of water molecules, such as hydroxide (OH–) and hydronium (H3O+) ions, at room temperature in ambient atmospheres, surfaces with unpaired ionic bonds have the tendency to reduce their surface energy.1
For instance, a monolayer of OH– ions is typically found bound on a ferroelectric surface, creating a layer of M···OH surface dipoles (where M is an electropositive metal atom).
This article focuses on ferroelectric single-crystal periodically-poled lithium niobate (PPLN) as a characteristic polar surface.
On such a surface’s up domains (unprotected positively charged sites), it is possible to screen the dipoles via a so-called “rumpling” conformational change of the layer of −OH groups. However, on the down domains (unprotected negatively charged sites), surface layers of ions restructure to screen the polarization with an additional two layers of H3O+ ions on top of the bound −OH layer.
Relative humidity (RH) regulates the availability of OH– and H3O+ for these screening phenomena and plays a key part in the extent of surface screening. Humidity has an additional impact on the kinetics of re-screening in circumstances where the surface has unfinished screening as a result of some physical perturbation.
The Charge Gradient Microscopy (CGM) technique precisely provokes this type of physical perturbation by high-force scraping a surface using an AFM tip. The collection and quantification of surface screening charges simultaneously can unveil the fundamental polarization domain structure at rapid scanning rates (see a schematic in Figure 1).
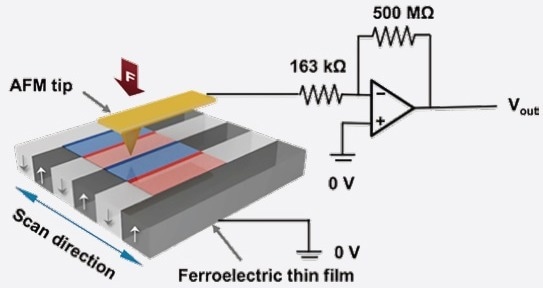
Figure 1. Schematic diagram of CGM [adapted from Reference 2]. Image Credit: Asylum Research - An Oxford Instruments Company
In reality, CGM tip velocities can exceed ~5 mm/second, facilitating quantification of both the bound surface charge density, as well as the related surface screening dynamics on polar surfaces in ambient conditions.2,3
Additional scanning probe techniques such as piezoresponse force microscopy (PFM), electrostatic force microscopy (EFM), or Kelvin probe force microscopy (KPFM) are also capable of mapping ferroelectric domains at high resolutions, but only in the quasistatic regime, as a result of their tip velocities being much slower (usually < 0.1 mm/second).
As a nanoscale technique, CGM can be understood through an analogy to a macroscale measurement: monitoring electrical currents at fixed locations on Earth (like the AFM tip) enables the variations in the charge densities of passing storm clouds (like the surface-adsorbed water) to be mapped out.4
References
- S. Hong, S. M. Nakhmanson, D. D. Fong, (2016) “Screening mechanisms at polar oxide heterointerfaces,” Rep. Prog. Phys. 79, p. 076501.
- S. Hong et al., (2014) “Charge Gradient Microscopy,” Proc. Nat’l. Acad. Sci. USA 111, pp. 6566–6569.
- A. Roelofs and S. Hong, (2018) “Charge Gradient Microscopy,” US Patent 9, p. 885, 861.
- J. M. Gregg and A. Kumar, (2014) “Trawling for complements,” Nature 510, pp. 481–482 (2014).

This information has been sourced, reviewed and adapted from materials provided by Asylum Research - An Oxford Instruments Company.
For more information on this source, please visit Asylum Research - An Oxford Instruments Company.