The longevity of plasma treatment is the most commonly asked question regarding plasma treatment. This article outlines the key considerations that impact the longevity of plasma surface treatments and discusses how these issues can impact various materials.
Plasma surface treatment is an effective and long-standing technique to enhance the adhesion capabilities of a broad range of materials.
Even for the most difficult polymers, for example, PPS, PET, PTFE, PEEK, polyamides, polyolefins and acetals (POM), plasma treatment is an efficient and dependable method to enhance a material’s surface energy, thereby making it wettable.

Image Credit: Henniker Plasma
Wettability is a term for the capability of adhesive, ink or paint to diffuse on a surface and produce strong contact and fixed chemical bonds with the molecules of a surface.
The majority of polymers are naturally hydrophobic, which means that liquids do not produce the large quantities of fixed bonds that are a requirement for effective adhesion and form beads on the surface.
Ceramics, metals and glass are naturally hydrophilic in contrast. Surface contamination is frequently found from the production and machining procedures which can also impact the adhesion to these materials.
Surface contamination can also be found on several engineering polymers in the form of antioxidants, pigments, plasticizers and mold release agents, for example.

Image Credit: Henniker Plasma
Plasma surface treatment performs through a double mechanism, the first is removing contamination from the surface (cleaning) and the second is where the surface energy is increased through the insertion of molecules containing oxygen (activation).
Activation mostly applies to polymers, whereas cleaning occurs on all surfaces.
What Causes Plasma Treatments to Decay?
In the example of plasma cleaning, components will usually stay in a cleaned state forever, so long as they are kept in a clean environment and are free from the transfer of salts and oils (for example, from being touched by bare skin) or contaminants in the atmosphere.
In the case of polymers, the situation is slightly more challenging. The cleanliness elements mentioned above are still applicable, but surface activation is now dominant in enhancing the material’s surface energy.
The technical term for this mechanism is hydrophobic recovery, where the plasma treatment decreases in effectiveness over time due to the decrease in surface energy.
The main characteristics of a material that has been plasma treated are consistent compared to the surface of the material.
In time, thermodynamically driven reorientation can migrate the molecules from within the bulk to the surface, which relocates the oxygen-containing polar groups to below the surface.
This recovery is mainly a feature of the material instead of the plasma treatment and the reasons for this are discussed below.
Type of Material
One of the most crucial factors that influence the speed of hydrophobic recovery is the characteristics of the material subjected to plasma treatment.
Certain polymers are packed more tightly, which means the sub-surface molecules cannot move as freely, reducing their ability to move to the surface, decreasing the rate of the recovery process.
Opposed to this, polymers of a lower density can relocate more efficiently, which causes migration of the oxygen-containing surface molecules to the bulk. If present, low molecular weight additives like pigments and plasticizers will also move back to the surface in time.
Figure 1 presents this variation by contrasting the hydrophobic recovery of high-density polyethylene (HDPE) and low-density polyethene (LDPE).
Compared to HDPE, LDPE recovers to a lower energy state (higher water contact angle), but both materials maintain higher surface energy compared to the materials that were untreated, even after multiple weeks.
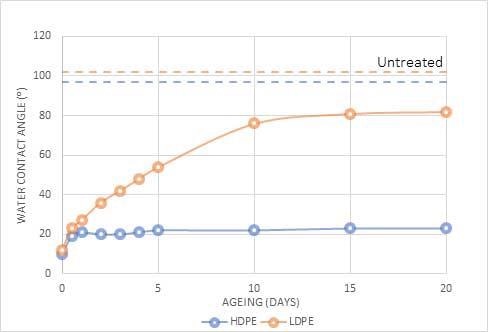
Figure 1. Change in water contact angle(⁰) of High-Density Polyethylene (HDPE) and Low-Density Polyethylene (LDPE) after plasma treatment. Image Credit: Henniker Plasma
There are a number of additional intrinsic characteristics of a polymer that may influence the speed of recovery, for example, conductivity, crystallinity and polarity, among others.
Figure 2 shows the variations between three separate polymers after being subjected to the same plasma treatment and being kept in equivalent environments.
It should be noted that not only is the speed of recovery different, but certain polymers are treated much more successfully compared to others, with polystyrene (PS) keeping in a high activation state even after multiple weeks.
Once again, it can also be observed that all of the materials continue to be in a state of activation and never completely recover to the low energy state level of untreated material.
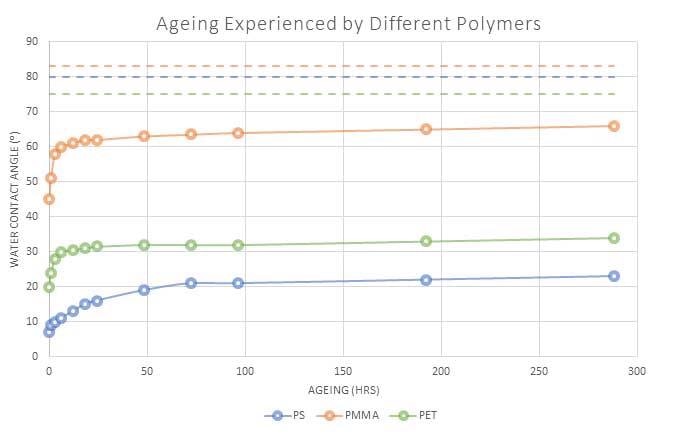
Figure 2. Water contact angle(⁰) of 3 different polymers after undergoing the same plasma treatment. Image Credit: Henniker Plasma
Duration of Plasma Treatment
There is some evidence that a prolonged exposure time to plasma may create a higher quantity of subsurface and surface bonds of the material being broken by the plasma’s UV radiation, resulting in simpler chain motion of molecules in the material’s bulk and subsequently a more efficient recovery mechanism.
Some reports have even shown that plasma durations of just 5 seconds are enough. This short duration is not always sufficient for the first stage where contamination is eliminated which makes it crucial to achieve a balance.
Figure 3 demonstrates the surface energy recovery behavior of plasma treated PET for various durations of plasma treatment.
While the speed of recovery is more efficient for longer plasma treatment, this continues to be the most effective treatment duration and produces a lower contact angle (higher surface energy) overall.
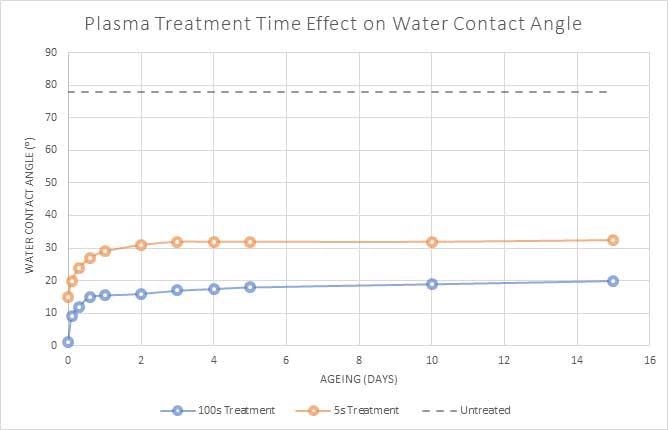
Figure 3. Plasma treatment of PET polymer for 5 seconds and 100 seconds. Image Credit: Henniker Plasma
Storage Conditions
The energy state of the surface can be greatly affected by the storage conditions of plasma-treated materials. For example, a more efficient rate of hydrophobic recovery can be achieved in higher temperatures due to the enhanced ease of mobility of chain structures underneath the surface.
The influence of storage temperature on a water contact angle of PET that has been plasma treated is presented in figure 4.
It can be observed that the fast recovery of the sample is accelerated by the higher temperature storage conditions.
In this case, the results for the water contact angle demonstrate that the surface energy returns to nearly 90% of its value in under one hour.
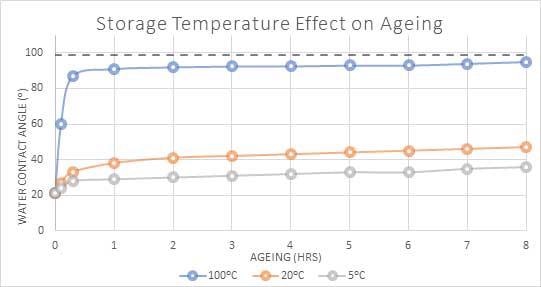
Figure 4. Water contact angle(⁰) of PET polymer over time when stored at different temperatures. Image Credit: Henniker Plasma
The transfer of salts and oil from handling with bare hands and airborne contamination also have a negative impact on surface energy.
Recontamination can be prevented by covering the plasma-treated material but consideration must be made to ensure the covering itself does not leech or transfer contamination.
Figure 5 contrasts two samples after plasma treatment showing one that has been left uncovered and one that has been covered throughout.
A significant change in contact angle over time can be seen in the uncovered elements, likely due to different degrees and types of contamination on various areas of the surface, whereas a more stable expected recovery can be seen in the covered samples over time.
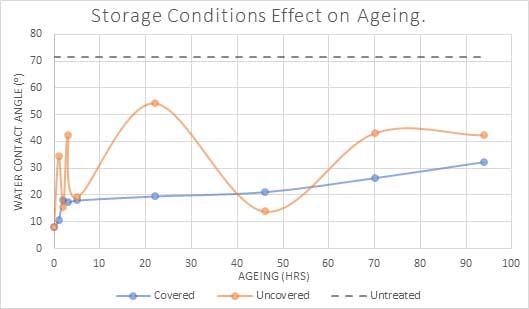
Figure 5. Water contact angle(⁰) of PET polymer over time when covered and handled with gloves, compared to uncovered and handled without gloves. Image Credit: Henniker Plasma
Type of Plasma Treatment
The type of gas utilized in the plasma process is a further factor that can influence the speed of recovery in a plasma-treated material. Various gases can present unique functional groups onto the surface, which consequently results in slightly modified surface characteristics.
Figure 6 shows a comparison of the quantified water contact angle of plasma-treated PET for various process gases. Air demonstrated the highest initial decline, while the oxygen-treated PET sample maintained the lowest contact angle.
Due to the amount of oxygen in ambient air, air plasma procedures are often highly suited for the treatment of materials and are less expensive compared to the use of bottled gas.
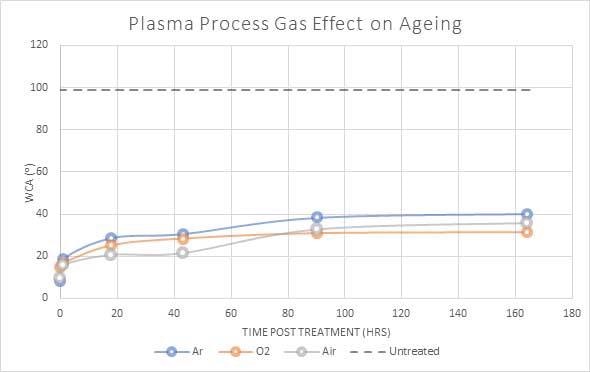
Figure 6. Water contact angle(⁰) of PET polymer when plasma treated with differing process gasses. Image Credit: Henniker Plasma
Example Water Contact Angles
The images below demonstrate the change in water contact angle overtime for a sample that has been plasma treated. Immediately after plasma treatment, the sample is seen to be fully wetted, which suggests superior adhesion characteristics and high surface energy.
As the sample gradually recovers, the water contact angle increases over a seven-hour period but continues to have a state of higher surface energy compared to the untreated sample.
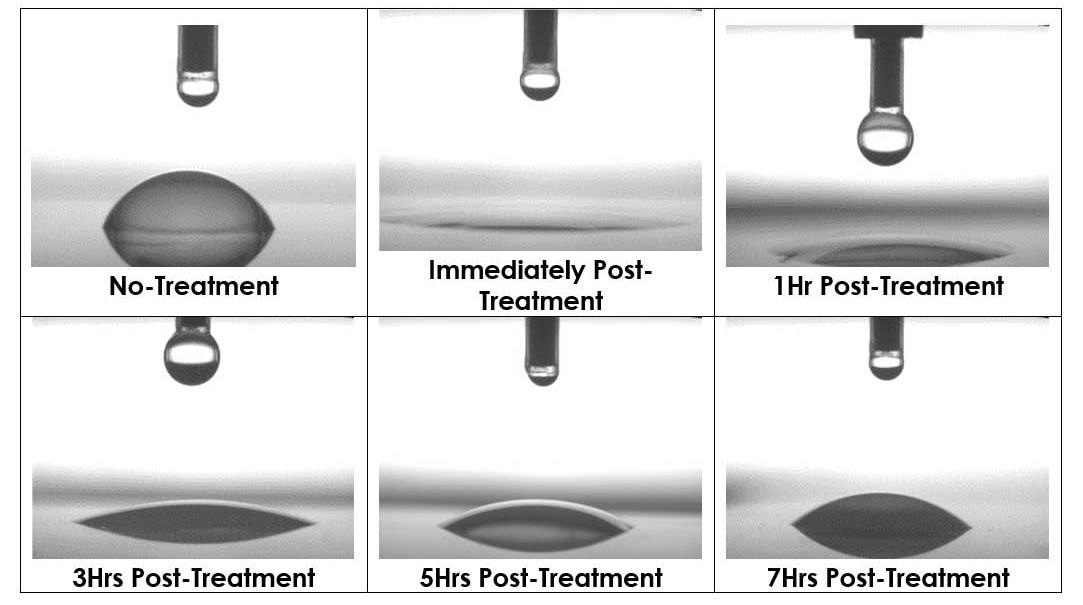
Image Credit: Henniker Plasma
Conclusions
When questioning how long a particular plasma treatment will last, each material must be considered individually. Generally, the majority of polymers will maintain a higher surface energy compared to the untreated polymer for many weeks.
Overall, for optimal results, it is preferred to conduct bonding/printing processes as soon as practically viable after plasma treatment. The following factors should be considered if this is not possible:
- Always utilizing powder-free gloves for plasma-treated part handling.
- Covering the components where possible with the use of foil and not leaving the treated surface vulnerable to ambient air.
- Decreasing lengthy treatment times. Oxygen plasma is able to fully activate and clean a polymer surface in a few minutes.
- Storing parts that have been treated with plasma in the coolest location possible, avoiding areas that are warm.
Henniker has a thoroughly equipped plasma testing and processing laboratory in its UK headquarters, where investigations can be performed to establish both the lifetime and effectiveness of plasma treatment on a broad range of samples.
Contact Henniker today to learn more.

This information has been sourced, reviewed and adapted from materials provided by Henniker Plasma.
For more information on this source, please visit Henniker Plasma.