Attenuated total reflection Fourier transform infrared (ATR-FTIR) spectroscopy is an excellent tool for analyzing biofluids. A biological fingerprint can be extracted from the sample as an IR spectrum by monitoring molecular bond vibrations when the sample is stimulated with IR light.
Healthcare researchers and doctors are interested in “biofluid biopsies” since they constitute a minimally intrusive sample-collecting technique. ATR-FTIR can be employed to examine a variety of biofluids, including blood, saliva, and urine. It is possible to distinguish between healthy and sick patients by comparing the spectral differences between biofluid samples.
The most often utilized biofluid in diagnostic medicine is blood, which comprises platelets, plasma, red blood cells, and white blood cells. Plasma or serum are frequently employed for spectroscopic analysis since both solutions can be frozen for preservation.
The freezing procedure in whole blood would cause cell disintegration, and hemoglobin has a significant spectral influence.
As shown in Figure 1, a serum separates the blood cells and platelets by allowing the blood to clot, whereas plasma is an aqueous solution that is separated by centrifuging in an anti-coagulant tube. The serum is utilized more frequently because it removes red blood cells more effectively than other preparation methods.

Figure 1: Plasma vs. serum. Image Credit: Edinburgh Analytical
The fundamental vibrations of the functional groups in biological samples, such as proteins, lipids, and amino acids, can be detected using ATR-FTIR in the mid-IR range (4000 cm–1–400 cm–1). In addition to producing rapid results and being a simple, low-cost approach for diagnostics, the procedure only needs a modest sample volume in the order of microliters.
ATR-FTIR spectroscopy has been proven effective for serum analysis in various disease investigations, including cancer, endometriosis, brain diseases, and viral infections.1–6
Impact of Water on Biofluid Measurements
The impact of water on the IR spectrum is a shortcoming of ATR-FTIR for biofluid analysis, as seen in Figure 2.

Figure 2: ATR-FTIR spectrum of. Image Credit: Edinburgh Analytical
Due to its polarity, water has a high IR response, which obscures the biological information in the sample when biofluids are analyzed in the liquid phase utilizing ATR-FTIR. Typically, while studying biofluids, the user must wait for the sample to dry before the biological spectrum is visible. However, this slows down the entire acquisition process.
Using a heated ATR accessory is a faster and more convenient way. The sample can be heated to cause the water to evaporate more quickly, reducing the time needed for collection. This article removed water from the sample while quickly measuring the serum’s spectrum using an IR5 FTIR Spectrometer with a heated ATR accessory.
Materials and Methods
An Edinburgh Analytical IA30 FTIR Spectrometer with a heated ATR accessory was used to measure a sample of human serum albumin. Diamond served as the ATR's internal reflective element (IRE), a common IRE due to its high refractive index and durability. Diamond ATRs are ideally suited to heated experiments due to their superior thermal conductivity.

Figure 3: Edinburgh Analytical IA30 FTIR Spectrometer. Image Credit: Edinburgh Analytical
To show how heating the ATR affects drying times, 3 μL of the sample was pipetted onto the ATR without being heated, and spectra were obtained. The ATR was heated beforehand and maintained at 50 °C for this procedure.
The IR spectra were gathered with a resolution of 4 cm–1, an average of 10 spectra, and an acquisition duration of 35 seconds for each spectrum.
Results and Discussion
Figure 4 displays the Human Serum Albumin IR spectra after two minutes of drying at room temperature (blue) and heated (red) ATR. Strong water absorption bands dominate the room temperature ATR measurement spectrum, obscuring the biological spectrum.

Figure 4: ATR-FTIR spectra after 2 minutes drying time of Human serum albumin at room temperature (blue) and heated to 50 °C (red). Image Credit: Edinburgh Analytical
A whole biological spectrum free of any water interference was achieved for the heated ATR. The time it would take for the biological spectrum to be sufficiently disclosed for analysis after the blue spectrum had dried longer at room temperature would be over 15 minutes, significantly slowing down measurement throughput.
Figure 5 lists the band assignments of the serum spectrum and details the crucial information ATR-FTIR offers about the biological components in a biofluid sample. The high-wavenumber region of an IR bio-spectra is thought to span between 2600 cm–1 and 3800 cm–1, whereas the low-wavenumber region, which includes double bond stretching and the fingerprint region, spans between 2000 cm–1 and 500 cm–1.
The most discriminatory data are generated in the low-wavenumber range, where most diagnostics are performed.7
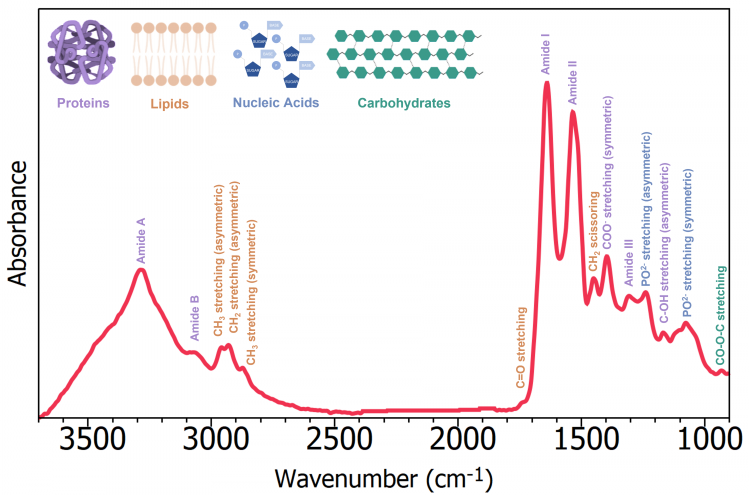
Figure 5: ATR-FTIR spectrum with band assignments of Human serum. Image Credit: Edinburgh Analytical
Conclusion
The biological fingerprint of a serum sample highlights the exceptional sensitivity of IA30 to the analysis of biofluids. The IA30 was given a heated ATR accessory to solve parasitic water absorption. The drying time before the spectral acquisition was significantly shortened by utilizing a heated ATR without sacrificing spectral quality.
References and Further Reading
- Soares Martins et al., (2020) “Potential of FTIR Spectroscopy Applied to Exosomes for Alzheimer’s Disease Discrimination: A Pilot Study,” J. Alzheimer’s Dis., 74(1), pp. 391–405.
- J. Pabico et al., (2023) “Diagnostic Efficiency of Serum-Based Infrared Spectroscopy in Detecting Breast Cancer: A Meta-Analysis,” Lab. Med., 54(1), pp. 98–105.
- Guo et al., (2022) “Fast and Deep Diagnosis Using Blood-Based ATR-FTIR Spectroscopy for Digestive Tract Cancers,” Biomolecules, 12(12), pp. 1–15.
- Roy et al., (2019) “Spectroscopy goes viral: Diagnosis of hepatitis B and C virus infection from human sera using ATR-FTIR spectroscopy,” Clin. Spectrosc., 1, p. 100001.
- Naseer et al., (2021) “ATR-FTIR spectroscopy as the future of diagnostics: a systematic review of the approach using bio-fluids,” Appl. Spectrosc. Rev., 56(2), pp. 85–97.
- Kokot et al., (2022) “ATR-IR Spectroscopy Application to Diagnostic Screening of Advanced Endometriosis,” Oxid. Med. Cell. Longev., 2022, 2022
- Rohman et al., (2019) “The use of FTIR and Raman spectroscopy in combination with chemometrics for analysis of biomolecules in biomedical fluids: A review,” Biomed. Spectrosc. Imaging, 8(3–4), pp. 55–71.

This information has been sourced, reviewed and adapted from materials provided by Edinburgh Analytical.
For more information on this source, please visit Edinburgh Analytical.