ELTRA GmbH has introduced a strong and powerful elemental analyzer, called ELEMENTRAC ONH-p 2, for measuring hydrogen, oxygen, and nitrogen levels in inorganic materials, such as iron, steel, ceramics, or copper.
The NDIR and thermal conductivity detectors are extremely sensitive and consistently detect element concentrations from high percentages to low ppm content.
Oxygen, nitrogen and hydrogen analyser ONH-p 2 - Short introduction | ELTRA
Video Credit: ELTRA GmbH
The novel sample port system features a vertical sample drop and pulsed chamber rinsing functions, permitting convenient and user-friendly analysis of powdery, granular, or rod-shaped samples weighing up to 2 g.
The ELEMENTRAC ONH-p 2 elemental analyzer now meets or exceeds the requirements of all relevant international standards, like DIN EN 3976 or ASTM E 1019.
Benefits at a glance
- Short analysis time
- Pins, granules, and powders can be applied easily
- A closed-gas system ensures high sensitivity and minimal gas consumption
- Robust impulse furnace with 8.5 kW
- Low-cost argon can be used as carrier gas
- Analysis of inorganic samples, such as non-ferrous metals, steel, ores, slags, ceramics, etc.
- Autocleaner (optional)
Configurations
The ELEMENTRAC ONH-p 2 elemental analyzer is supplied as a single-element analyzer for hydrogen, nitrogen, or oxygen only, or comes in a multi-element configuration for quantifying NH, OH, ON, or ONH. While oxygen is determined as CO2 in up to two infrared cells, both hydrogen and nitrogen are identified in their elemental form in a thermal conductivity cell.
Besides this, the ELEMENTRAC ONH-p 2 elemental analyzer benefits from the sensitive thermal conductivity cell by using an OH mode with nitrogen carrier gas and ON mode with a helium carrier gas (optionally argon).
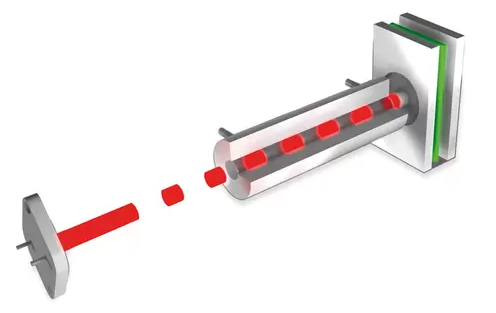
Cuvette with variable length. Image Credit: ELTRA GmbH
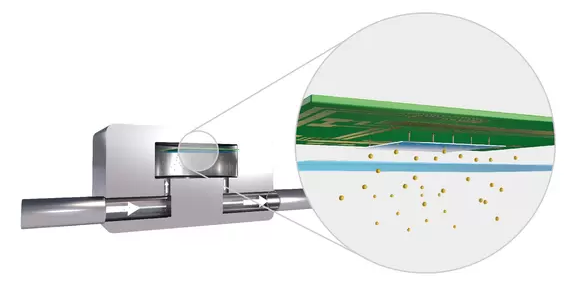
Thermal conductivity cell with high sensitivity. Image Credit: ELTRA GmbH
Integrated Standard Solutions
The filters and chemicals needed to operate the ELEMENTRAC ONH-p 2 elemental analyzer are easily organized on the front panel and these can be hidden behind a removable door at the time of routine operation. A setup like this considerably increases the user-friendliness while decreasing the time needed for maintenance. Novel details significantly enhance measurement reproducibility.
Innovative Sample Port and Pulsed Chamber Flushing
The ELEMENTRAC ONH-p 2 elemental analyzer features a new sample port that ensures easy operation and reproducible quantified values. Different shapes of samples, such as granules, solid pieces, or powder in capsules, can be used up to a weight of 2000 mg.
Such samples are rapidly freed from the surrounding atmosphere using the pulsed carrier gas flushing provided in the sample port. The samples subsequently drop in a vertical manner into the preheated graphite crucible for analysis.
- Capsules do not need to be closed
- Resists dust development
- Minimal wear and maintenance
- Up to 2000 mg granulate can be applied directly
Powerful Catalyst
During elemental analysis, in the graphite crucible, carbon monoxide (CO) gas is produced, which is changed to carbon dioxide (CO2) in the catalyst and then identified in the IR cells. The catalyst with copper oxide filling can be maintained easily and guarantees complete oxidation, thus ensuring consistent oxygen analysis of even challenging materials, like oxides.
Closed Gas Management
The ELEMENTRAC ONH elemental analyzer range utilizes a closed-gas system in overpressure. This system ensures that all the released sample gas is invariably fed to the detectors, guaranteeing excellent reproducibility and low detection limits.
Options
Apart from the incorporated solutions of the ELEMENTRAC ONH-p 2, additional options are also available to boost efficiency and expand the application range of elemental analysis.
Autocleaner
When the sample is melted in a graphite crucible at temperatures as high as 3000°C, deposits are created in the furnace chamber and at the upper electrode, which may affect the reproducibility of ONH measurements adversely.
These deposits are consistently removed by the new Autocleaner, which is provided as an option, allowing accurate elemental analysis even for high throughputs. The elemental analyzer comes with an efficient gas calibration and cleaning furnace for comprehensive pre-cleaning of carrier gas.