Apr 18 2017
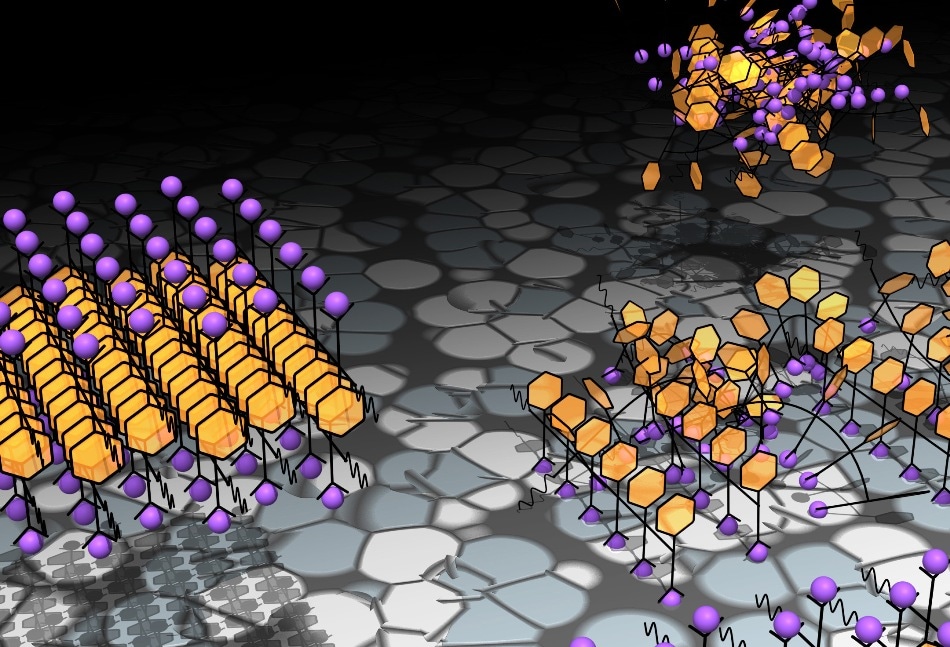 A small change to a peptoid that crystallizes in one step (left) sends the modified peptoid down a more complicated path from disordered clump to crystal (right) (Credit: PNNL)
A small change to a peptoid that crystallizes in one step (left) sends the modified peptoid down a more complicated path from disordered clump to crystal (right) (Credit: PNNL)
Solar panels, a better medical drug, or smooth chocolate all require just the right amount of crystals to make up the material. Now, researchers trying to understand the paths crystals take as they develop have been able to influence that path by adjusting the starting ingredient.
The findings from the research are reported in the April 17 issue of Nature Materials, and could ultimately help scientists to have better control of the design of various products for medical or energy technologies.
The findings address an ongoing debate about crystallization pathways. They imply you can control the various stages of materials assembly by carefully choosing the structure of your starting molecules.
Jim De Yoreo, Materials Scientist, Department of Energy's Pacific Northwest National Laboratory
From Floppy to Stiff
One of the simplest crystals, diamonds, are made up of a single atom - carbon.
However, in the living world, crystals, like the ones formed by cocoa butter in chocolate or ill-formed ones that result in sickle cell anemia, are formed from molecules that are long and floppy and comprise of a lengthy distinct sequence of many atoms.
They can crystallize in numerous ways, but only one way is the best. In pharmaceuticals, the difference can mean a drug that functions effectively versus one that does not.
Chemists do not yet have enough control over crystallization to guarantee the best form, partially because chemists are not sure how the initial steps in crystallization occur.
A specific debate has concentrated on whether complex molecules can assemble directly, with one molecule attaching to another, like adding one playing card at a time to a deck. They refer to this as a one-step process, the mathematical rules for which researchers have long understood.
The other side of the debate argues that crystals need two steps to form. Experiments propose that the initial molecules first form a disorderly clump and then, from within that group, start rearranging into a crystal, as if the cards have to be shuffled first before they could form a deck.
De Yoreo and his colleagues wanted to establish if crystallization always required the disordered step, and if not, why not.
Clump, Snap and ...
To achieve that, the researchers formed crystals from a slightly simplified version of the sequence-defined molecules found in nature, a version they refer as a peptoid.
The peptoid was not intricate - just a string of two repeating chemical subunits (think "ABABAB") - yet complex because it was a 12-subunits long. Based on its symmetrical chemical nature, the team expected a number of molecules to join into a larger structure, like Lego blocks snapping together.
In a second series of experiments, they wanted to test how a somewhat more complicated molecule assembled. So, the researchers incorporated a molecule onto the original ABABAB sequence that stuck out like a tail.
The tails attracted each other, and the team anticipated their link would cause the new molecules to clump. But they were not sure what would happen later.
The researchers placed the peptoid molecules into solutions to allow them crystallize. Then the team used various analytical methods to see what shapes the peptoids formed and how fast. They discovered that the two peptoids created crystals in extremely different fashions.
A Tail of Two Steps
As the researchers mostly anticipated, the simpler peptoid developed the initial crystals a few nanometers in size that grew longer and taller as more of the peptoid molecules snapped into place. The simple peptoid followed all the rules of a single-step crystallization process.
However, introducing the tail into the mix disturbed the calm, resulting in a complex set of events to occur before the crystals appeared. Overall, the team demonstrated that this more complicated peptoid first clumped together into small clusters unnoticed with the simpler molecules.
Some of these clusters settled onto the existing surface, where they sat static before unexpectedly converting into crystals and finally growing into the same crystals seen with the simple peptoid. This behavior was rather new and required a different mathematical model to define it, according to the researchers. Understanding the new rules will permit the researchers to establish the ideal way to crystallize molecules.
We were not expecting that such a minor change would make the peptoids behave this way. The results are making us think about the system in a new way, which we believe will lead to more predictive control over the design and assembly of biomimetic materials.
Jim De Yoreo, Materials Scientist, Department of Energy's Pacific Northwest National Laboratory
This research was supported by the Department of Energy Office of Science and PNNL's Laboratory Directed Research and Development program.