Gold nanoparticles, which are also referred to as colloidal gold, have distinctive properties that benefit numerous applications. Usually, they are synthesized by controlled decrease of aqueous HAuCl4 solution employing a reducing agent like citrate under differing conditions.
There are numerous varieties of gold nanoparticles established on the size, shape, and physical properties (Figure 1). Drug delivery,1 carriers for drugs such as Paclitaxel,2 tumor detection,3 and biosensors are some of the applications for gold nanoparticles, along with many others.
Figure 1. Types of gold nanoparticles (stylized). Image Credit: Entegris
The size of gold nanoparticles is a crucial physical parameter4 needing precise measurement. The particle size impacts properties including absorbance wavelength (larger size = longer wavelength), intracellular uptake, surface plasmon resonance (SPR) peak, blood half-life, and biodistribution profile (decreasing size = increased blood half-life). The particle size and width may be employed as an indication of suspension stability. Additionally, surface charge (zeta potential) measurements are employed as a sign of suspension stability.
The most frequently favored method for particle size and zeta potential analysis is dynamic light scattering (DLS) and electrophoretic light scattering (ELS). The Nicomp® Z3000 system (Figure 2) is ideal for ascertaining the size and zeta potential of gold nanoparticles.

Figure 2. Nicomp DLS system. Image Credit: Entegris
Materials
The size and zeta potential of numerous gold nanoparticle samples were examined with the Nicomp DLS system. Three separate gold nanoparticle samples were bought from Sigma Aldrich with nominal sizes of 50, 20, and 5 nm. Also examined was NIST reference material 8012 with a nominal size of 30 nm.
In Table 1, the reference physical properties of these samples can be seen. Sizes analysis results by DLS are presented as the intensity mean of the hydrodynamic diameter. Width of the delivery is presented as the polydispersity index (PDI). The NIST 8012 Report of Investigation offers mean hydrodynamic diameter at two angles, backscatter at 173° and 90°. The reference zeta potential value for NIST 8012 is also supplied, contrasting with the samples bought from Sigma Aldrich.
Table 1. Reference physical properties. Source: Entegris
| Sigma Aldrich part number |
Polydispersity Index (PDI) |
Core size |
Mean hydrodynamic diameter (Z) |
| 742007 |
≤0.2 |
47 – 53 nm |
58 – 66 nm |
| 741965 |
≤0.2 |
18 – 22 nm |
28 – 36 nm |
| 741949 |
≤0.2 |
4 – 7 nm |
14 – 25 nm |
| NIST RM |
Polydispersity Index (PDI) |
DLS, 173° |
DLS, 90° |
Zeta potential |
| 8012 |
N/A |
28.6 ±0.9 nm |
26.5 ±3.6 nm |
-33.6 ±6.9 mV |
Method
Each sample was analyzed on a Nicomp Model Z3000 (Figure 2) for particle size and zeta potential. Prior to the beginning of these studies, both size and zeta potential specifications were examined to substantiate system performance. A concentration study was carried out on sample 742007 to determine if established particle size fluctuated with concentration.
The outcome of this investigation showed that the results were insensitive to concentration, and as such, all measurements were taken at the initial sample concentration without dilution. A time of analysis study was completed on sample 741949 to ascertain a suitable measurement duration built on reaching stability, as demonstrated in the Time History plot in Figure 3. This investigation showed that an analysis time of five minutes was adequate.
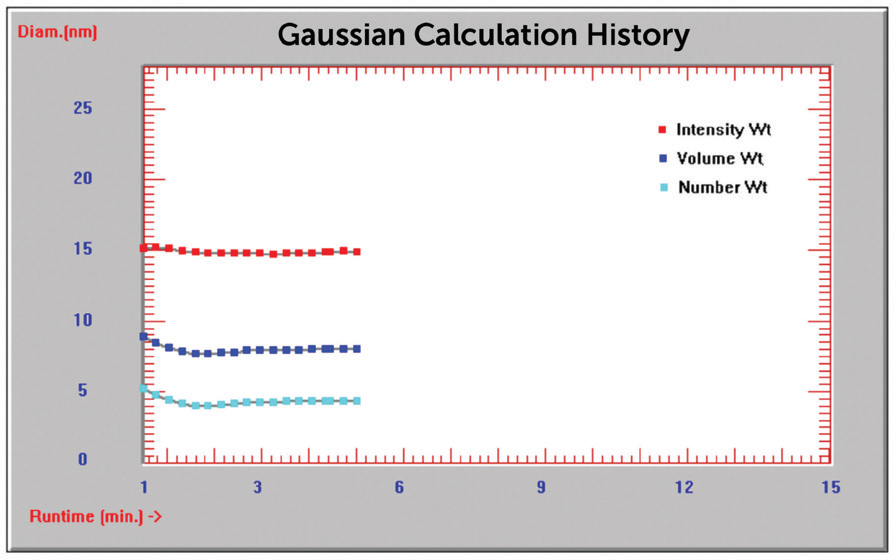
Figure 3. Time history plot diameter (nm) vs. time (min). Image Credit: Entegris
The Nicomp size and zeta potential analysis settings for every measurement can be seen in Figures 4 and 5.
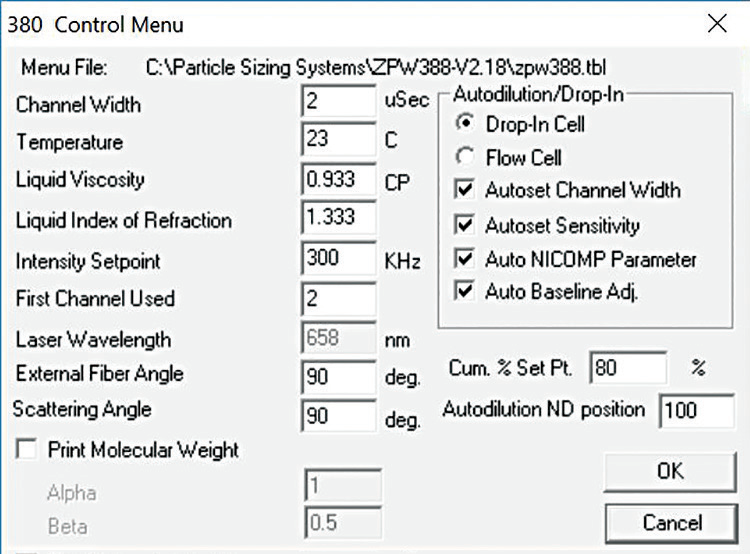
Figure 4. Nicomp size settings. Image Credit: Entegris
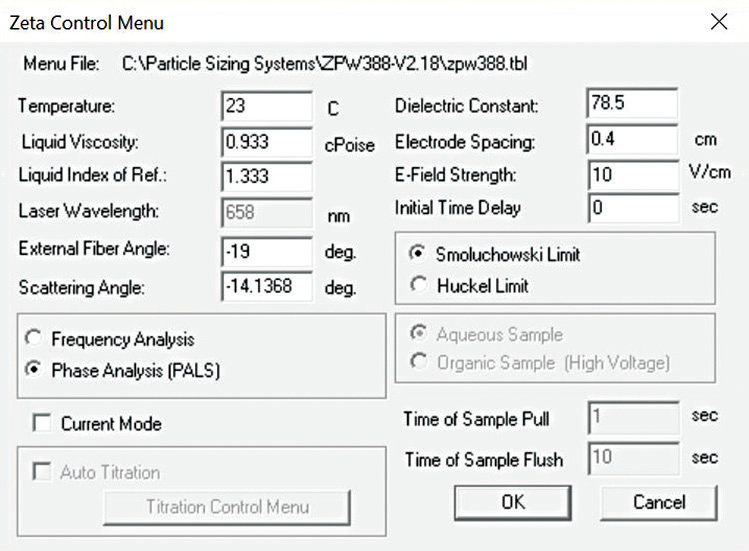
Figure 5. Nicomp zeta potential settings. Image Credit: Entegris
Results - Particle Size
In Figures 6-10, typical graphical results for two analyses for each sample can be seen.
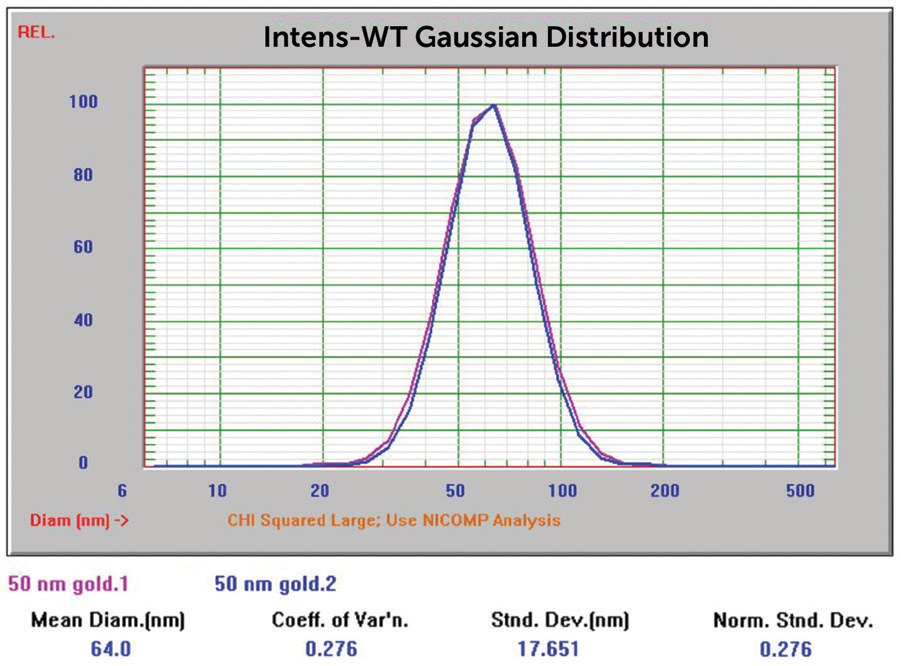
Figure 6. 50 nm Au overlay of two size results. Image Credit: Entegris
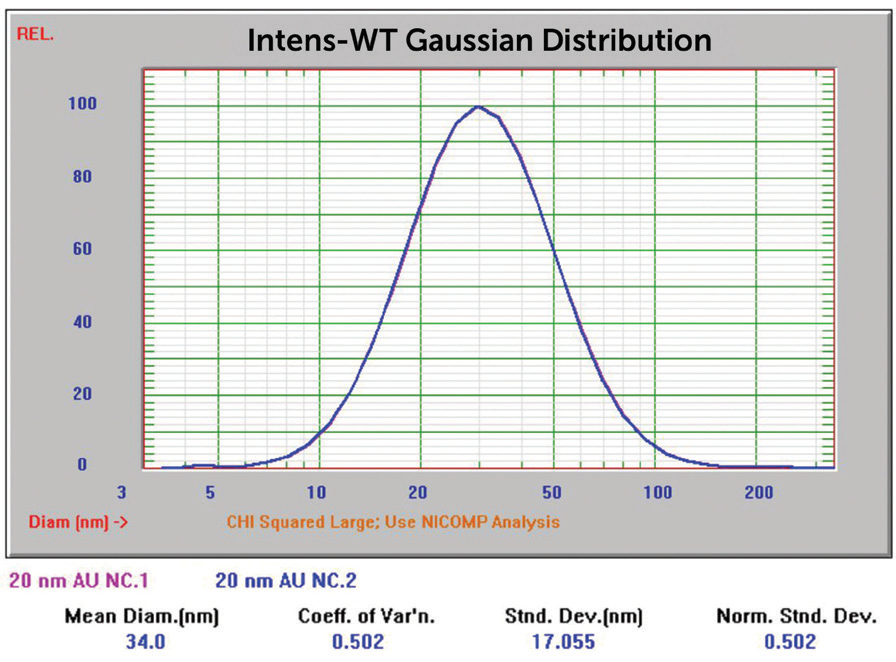
Figure 7. 20 nm Au overlay of two size results. Image Credit: Entegris
Sample 741949 (nominal size 5 nm) made generating results close to anticipated values more difficult. The Gaussian result for the sample, when analyzed with no extra sample preparation, can be seen in Figure 8. Gaussian calculations push the result into one single peak.
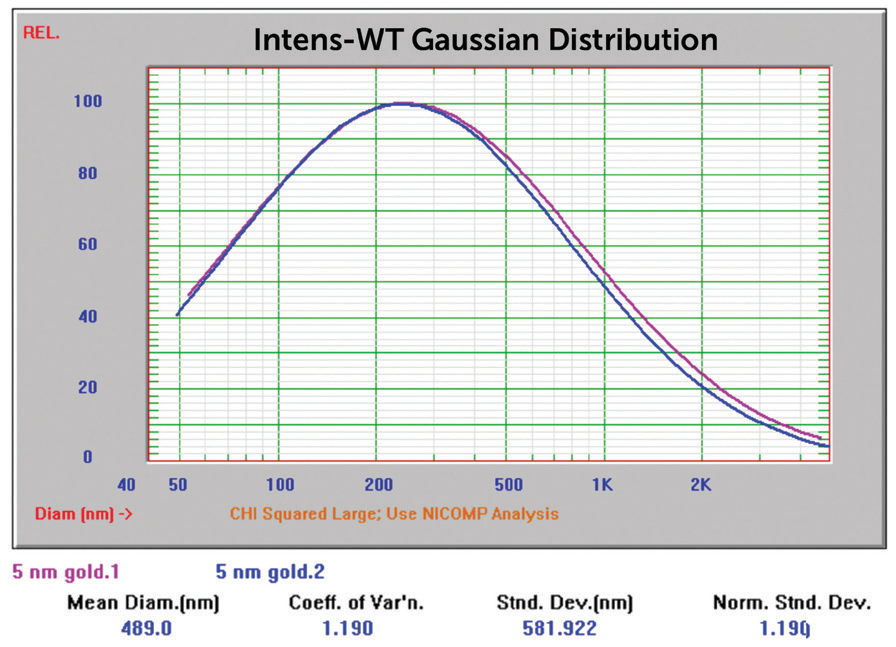
Figure 8. 5 nm Au overlay of two size Gaussian results. Image Credit: Entegris
The Nicomp Z3000 system can also produce multi-modal results by applying the proprietary Nicomp algorithm. The result shown in Figure 8 was recalculated applying the Nicomp algorithm, and in Figure 9, the resultant multi-modal size distribution can be observed.
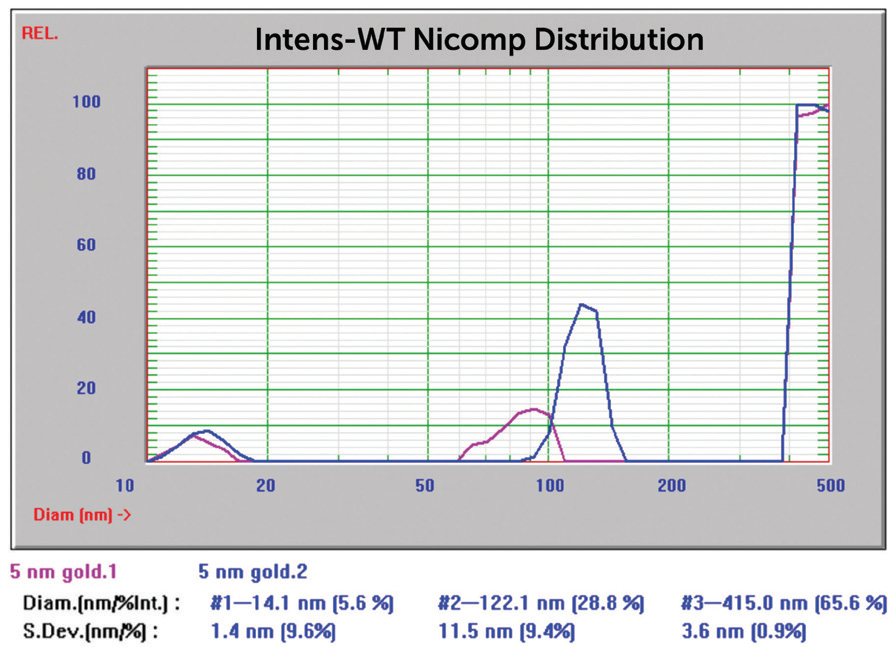
Figure 9. 5 nm Au overlay of two size Nicomp results. Image Credit: Entegris
Following this, the sample tube was centrifuged for eight minutes to try to divide the bigger particles from the measurement zone. The same sample tube was examined following centrifugation, and the result can be seen in Figure 10.
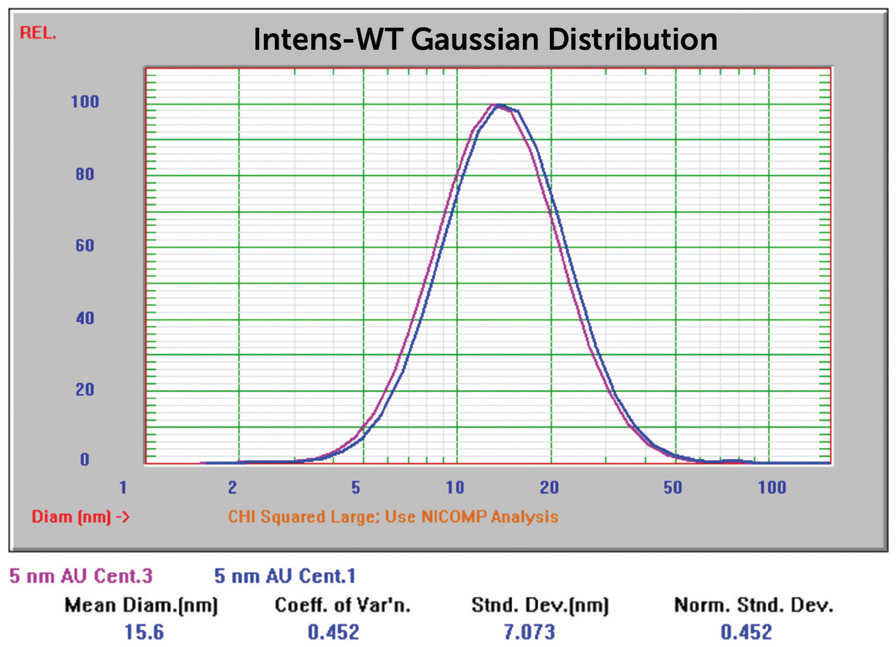
Figure 10. 5 nm Au overlay of two size Nicomp results, centrifuged. Image Credit: Entegris
Sample NIST 8012 was examined at three angles: backscatter @ 170° (pink), 90° (green), and forward angle at 15° (blue). The results are recorded in Figure11.
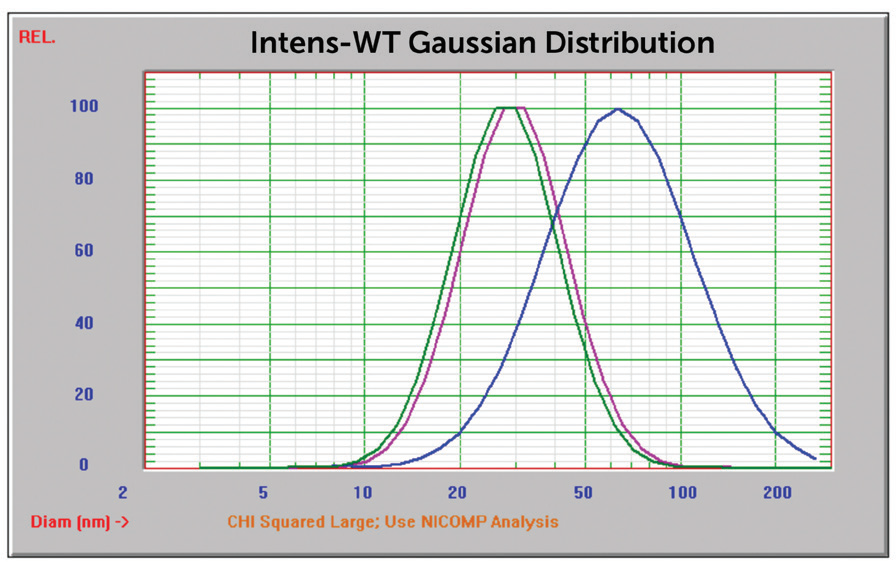
Figure 11. NIST 8012 Au overlay of three size results, 170°, 90°, and 15°. Image Credit: Entegris
These findings and zeta potential values are shown in Table 2.
Table 2. Reported size and zeta potential results. Source: Entegris
| Sample |
Expected result |
Reported size |
Zeta potential |
| 742007 |
58 – 66 nm |
64 nm |
-34.58 mV |
| 741965 |
28 – 36 nm |
34 nm |
-27.95 mV |
| 741949 |
14 – 25 nm |
15.6 nm |
-42.43 mV |
| 8012, 170° |
28.6 ±0.9 nm |
31.9 nm |
— |
| 8012, 90° |
26.5 ±3.6 nm |
30.8 nm |
— |
| 8012, 15° |
— |
66.6 nm |
— |
| 8012 zeta |
-33.6 ±6.9 mV |
— |
-30.49 mV |
Results – Zeta Potential
Additionally, the samples were evaluated for zeta potential applying the phase analysis electrophoretic light scattering technique (PALS). The zeta potential is a measurement of surface charge applied in formulation and suspension stability studies. In Figures 12 to 15, the zeta potential for these samples can be seen.
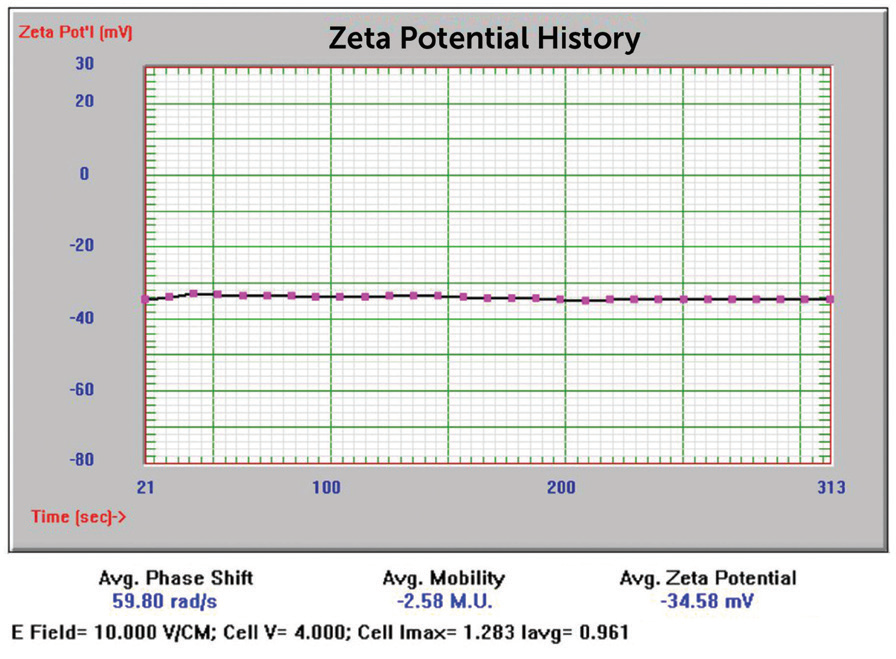
Figure 12. 50 nm Au zeta potential results. Image Credit: Entegris
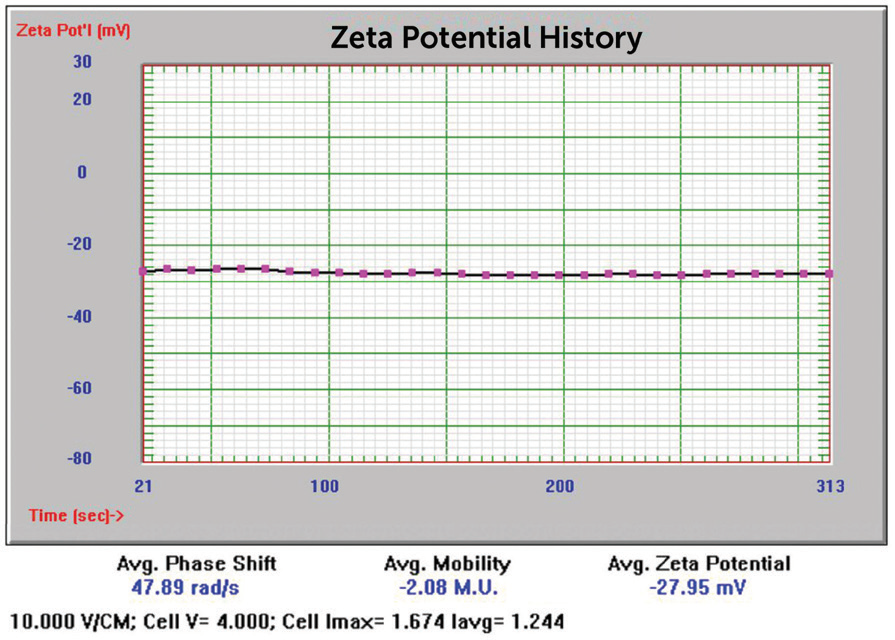
Figure 13. 20 nm Au zeta potential results. Image Credit: Entegris
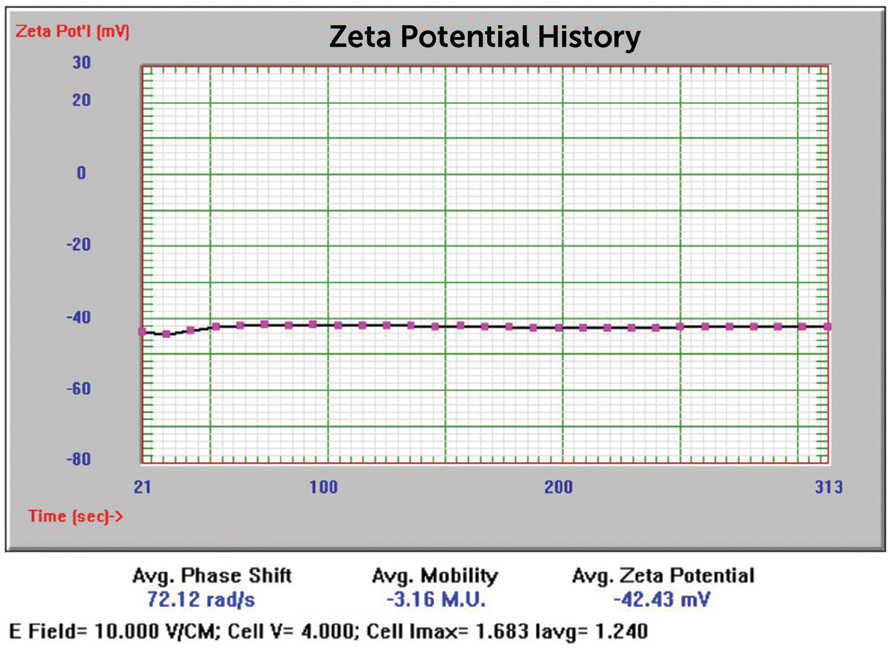
Figure 14. 5 nm Au zeta potential results. Image Credit: Entegris
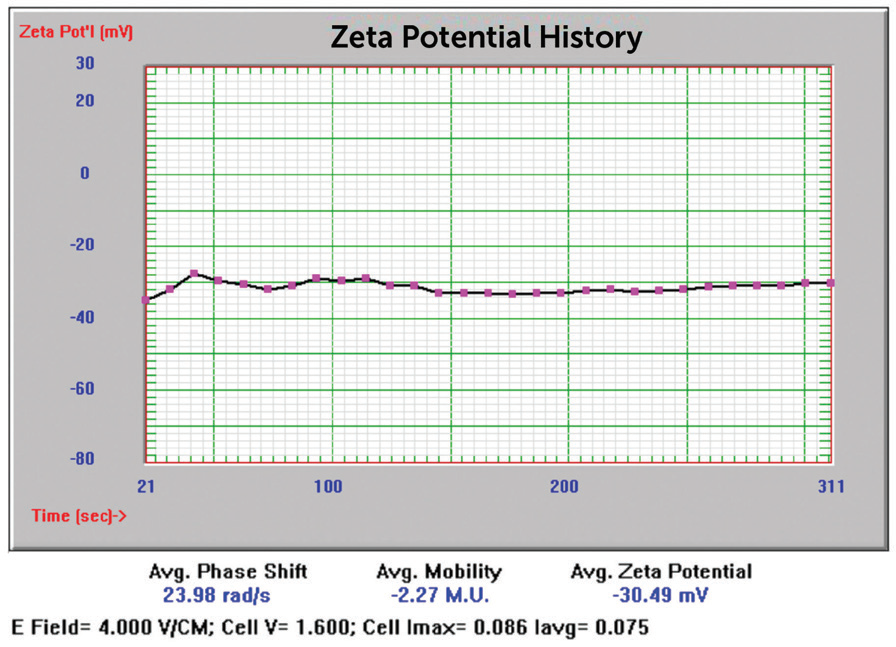
Figure 15. NIST 8012 Au zeta potential results. Image Credit: Entegris
Discussion
The results for Sigma Aldrich 50 and 20 nm nominal size samples were inside the anticipated value range. The Sigma Aldrich 5 nm nominal size results originally reported particle size considerably larger than anticipated, which could show aggregation. This was not unexpected due to the color of this sample displayed in Figure 16, far left.

Figure 16. Left to right; 5, 20, and 50 nm Au sample bottles. Image Credit: Entegris
A darker, bluer color usually suggests a significantly bigger particle size distribution. The result prior to centrifugation (Figure 9) suggests the occurrence of primary particles near 14 nm and aggregate peaks at 122 and 417 nm. This outcome shows the strength of the Nicomp algorithm to determine both primary particles and multiple peaks of aggregates. Following centrifugation, the reported mean size was inside anticipated values.
The NIST 8012 sample was examined at three separate angles: 15°, 90°, and 170°. The marginally smaller result at 90° vs. 170° implies 90° is a superior measurement condition vs. backscatter – consistent with other previously stated results.5 Measurement in the forward angle direction (15°) enhanced sensitivity to bigger particles, shown in Figure 11.
These outcomes indicate that a multi-angle system (a basic and cheap Nicomp option) may be better for certain applications. The zeta potential results were all very repeatable applying the PALS technique. The Nicomp dip cell can record thousands of samples with cheaper ownership than single-use zeta potential cells.
Stability Study
A stability study was done on the NIST 8012 sample to study the impact of salt concentration on size and look. The initial size result can be seen in Figure 11, with a reported size at 90° of 30.8 nm. Firstly, 200 µL of NIST 8102 was pipetted into 200 mL filtered DI water. Following this, KCl at 3 M was added to the diluted NIST 8012 suspension in increments of 500 µL while monitoring the color of the sample.
A color change was observed following the addition of 2 mL of 3 M KCl. Figure 17 displays, from left to right, the NIST 8012 sample after adding the KCL, original sample in cell, and the original sample bottle.

Figure 17. After KCL added, before, original bottle. Image Credit: Entegris
The particle size distribution of the NIST 8012 after adding KCL is displayed in Figure 18. The size grew as a function of time over the 15 minute analysis time. After 24 hours, the sample had entirely aggregated and settled to the base of the cuvette, shown in Figure 19.
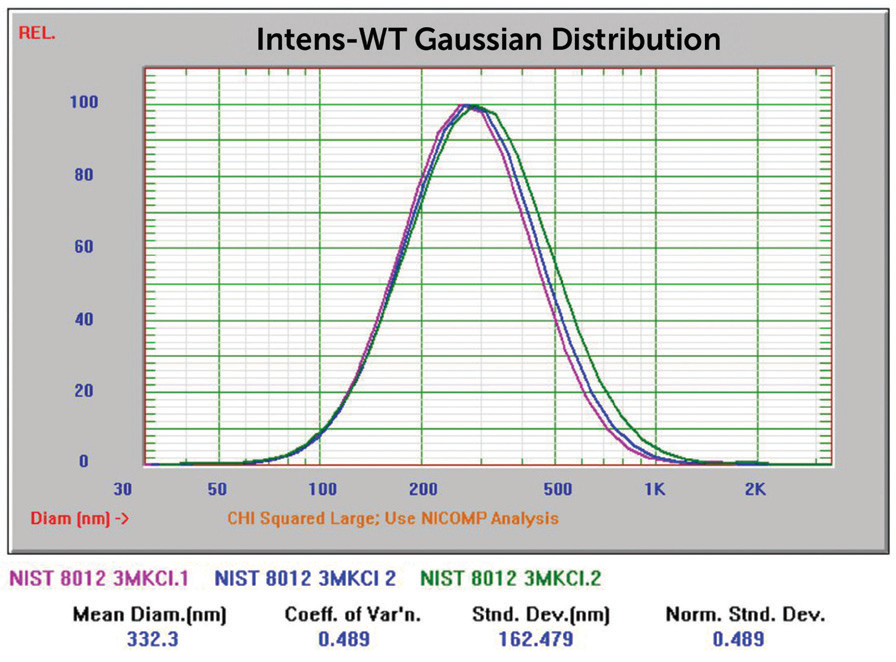
Figure 18. NIST 8012 Au overlay of three size results after KCl addition. Image Credit: Entegris

Figure 19. NIST 8012 with settled aggregates. Image Credit: Entegris
Conclusions
Dynamic light scattering is the favored method for particle size and zeta potential analysis of nanogold particles. The Nicomp Z3000 demonstrated that it is an accurate, high-resolution technique producing results close to anticipated values. The Nicomp algorithm detected the multi-modal mix of primary and aggregated particles for the aggregated samples.
References
- Han, G., Ghosh, P., and Rotello, V.M., Functionalized gold nanoparticles for drug delivery, Nanomedicine, (February 2007), (1): 113–23
- Gibson, J.D., Khanal, B.P., and Zubarev, E.R., Paclitaxel-functionalized gold nanoparticles, Journal of the American Chemical Society, (September 2007), 129 (37): 11653–61
- Sajjadi, A.Y., Suratkar, A.A., Mitra, K.K., and Grace, M.S., Short-Pulse Laser-Based System for Detection of Tumors: Administration of Gold Nanoparticles Enhances Contrast, J. Nanotechnol, Eng., (2012), Med. 3 (2): 021002
- Dreaden, E.C., Austin, L.A., Mackey, M.A., and El-Sayed, M.A., Size matters: gold nanoparticles in targeted cancer drug delivery, Ther Deliv, 2012;3(4):457-78
- PSS Technical Note 724 – Multiangle DLS Measurements

This information has been sourced, reviewed and adapted from materials provided by Entegris.
For more information on this source, please visit Entegris.