‘Xylene solubles’ (XS) is an established term indicating the percentage of soluble species in polypropylene homo- and o-polymers. This value is almost proportional to the amorphous content of the materials. In practice, this measure is broadly used for product quality control and monitoring physical properties of the polymer during synthesis and processing.
Method
The xylene extraction method (ASTM D5492, technically equivalent to the standard ISO 16152) is normally used to determine the xylene solubles content by weight. However, this method requires dissolution of the products in a harmful solvent, use of high temperatures (135 and 150 °C), large sample sizes (to guarantee reliable reproducibility), highly skilled analysts and long measurement times. The xylene solubles can also be determined using a Fourier transform infrared technique (FTIR). Although this method significantly brings down the analysis time, it requires delicate sample preparation and a highly skilled operator. In contrast to the standard wet chemistry method and FTIR technique, low resolution nuclear magnetic resonance (NMR) offers a rapid, direct and user friendly method for determination of the xylene solubles content in polypropylene products that can be operated by a non NMR expert user. This method is based on the measurement of the NMR response obtained from the amorphous part of the material and the quantification of the xylene solubles content by calibration.
Advantages of Benchtop NMR
The benefits of Benchtop NMR are:
- NMR is highly stable over the long-term and requires little recalibration.
- The sample measurement time is short, around 20 seconds.
- Minimal sample preparation is needed.
- The NMR technique is non-destructive, hence repeatability measurements can be performed conveniently.
Calibration and Results
The calibration is performed using at least 6 samples of real products with predefined xylene solubles values, spanning across the range of concentrations of interest.
Figure 1 shows a calibration for polypropylenes with xylene solubles content ranging from 0.9 to 4.9 % by weight (wt. -%). As shown in the figure, NMR gives an excellent linear correlation between the NMR response and the concentration of xylene solubles in the products.
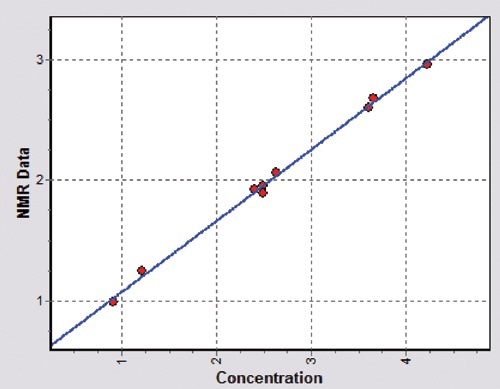
Figure 1. Calibration obtained for xylene solubles in polypropylenes; standard deviation of the linear fit is 0.06 wt. -%, the correlation coefficient is R2 = 1.00. Measurements were made using Oxford Instruments Magnetic Resonance MQC- 23 benchtop NMR analyser fitted with an 18-mm diameter probe.
The instrument repeatability was evaluated by measuring one sample ten times. After every test measurement, the sample was transferred from the magnet bore back to the conditioning block for 20 min to be conditioned at 60 °C and then measured again.
Table 1 shows the repeatability test results.
Table 1. Results of instrument repeatability test.
| Given XS Value, wt. -% |
Results (wt. -%) of Repeat Measurements |
Mean XS Value, wt. -% |
Standard Deviation wt. -% |
| 0.92 |
0.94 |
0.87 |
0.90 |
0.94 |
0.92 |
0.91 |
0.93 |
0.89 |
0.92 |
0.91 |
0.91 |
0.02 |
Instrument Configuration
The MQC-23 with 0.55 Tesla magnet fitted with an 18-mm diameter (8-ml) probe is ideal for this application. The ‘Xylene Solubles in Polypropylene’ package comprises the following:
- The MQC-23, which can be controlled using its own built-in computer using Microsoft Windows or via a stand-alone PC.
- MultiQuant software including RI Calibration, RI Analysis and the EasyCal ‘Xylene Solubles in Polypropylene’ application.
- Test/tuning sample.
- Sample tubes with an outer diameter of 18 mm.
- Polytetrafluoroethylene (PTFE) stoppers.
- Stopper removal tool.
- Installation Manual.
- Method Sheet.
- Dry block heater that can maintain samples at 60 °C.
- Aluminum block with holes for 18-mm diameter tubes.
Advantages of MQC-23
The benefits of the MQC-23 over its counterparts are:
- High-signal sensitivity.
- Small benchtop footprint.
- Specific ‘Xylene Solubles in Polypropylene’ Applications Software.
- Low maintenance.
- Minimal sample preparation.

This information has been sourced, reviewed and adapted from materials provided by Oxford Instruments Magnetic Resonance.
For more information on this source, please visit Oxford Instruments Magnetic Resonance.