Ethylene vinyl acetate (EVA) copolymer (Figure 1), based on the fraction of two monomers, can be used in a myriad of applications. The polymer melt needs to be monitored during manufacture to have a polymeric product with consistent quality. The sample manipulation in the case of conventional at-line and on-line techniques makes the sample unrepresentative of the large population.
Moreover, the polymer melt’s measured properties are influenced by the history of the sample pertaining to temperature, strain, stress, and strain rate. Therefore, the polymer melt requires a real-time, in-line sampling in a non-destructive manner.
Figure 1. EVA copolymer
Instrumentation
There are several methods (electrical, ultrasonic, optical, rheometric and much more) available for gaining insights into bulk physical properties of a polymer melt. Today’s spectroscopic methods can provide data on the molecular scale and are compatible to in-line analysis, of which Raman spectroscopy is more suitable for inline analysis of history-dependent materials.
The requirement for short path lengths in the case of MIR spectroscopy compels the removal of the sample from the process by a side stream and subsequent analysis in a temperature- and pressure-controlled cell.
Combination bands and overtones are intrinsic to NIR spectroscopy, thus providing less qualitative chemical data when compared to either MIR or Raman spectroscopy. Correlating these features to specific chemical species is a challenging task and the analyst finds it difficult to gain knowledge during process development. In addition, transmission probes used in the NIR system affected the flow of the polymer melt, causing problems with the shear heating and rheology of the polymer.
Furthermore, the process mixing can be changed due to the size of an in-line transmission probe, triggering turbulent flow or behaving as a fouling mechanism. Moreover, the NIR probes are not a standard component and therefore it is difficult to deploy them onto a range of processes.
Experimental Procedure
A single-screw extruder was used to extrude EVA copolymer samples with different compositions of vinyl acetate (VA) from 2 to 43.1wt%, at a set screw speed of 15rpm and melt temperature of 180°C. Raman spectra were collected every minute using Kaiser Optical Systems’ fiber-coupled HoloProbe™ Raman analyzer (current version is RamanRxn3™ analyzer) integrated with a CCD detector and a 785-nm laser.
The Raman analyzer used a proprietary high-pressure inline optic to interface with the extruder barrel. The acquisition time was 28s. The spectra’s first derivative was calculated to remove the fluorescence background, followed by mean-centering of the resulting first-derivative spectra. The ensuing first-derivative spectra were then analyzed using a one-factor partial least squares (PLS) regression model. NIR spectroscopy was also used to analyze the samples.
Experimental Results
Figure 2 illustrates the Raman spectra of EVA copolymer melts with 2% and 34.2% vinyl acetate. Characteristic vinyl acetate bands are observed at 630cm–1 (O–C=O deformation) and 1740cm–1 (C=O stretch). Raman spectroscopy enabled real-time monitoring of the sequential extrusion of each copolymer with increasing vinyl acetate content.
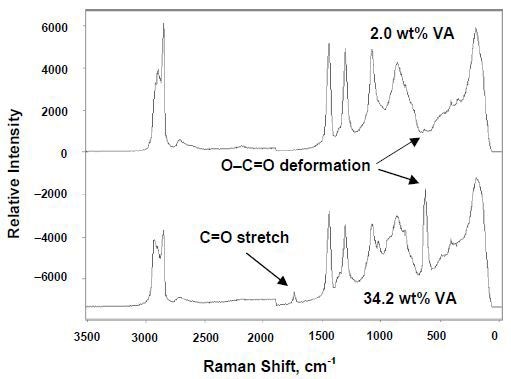
Figure 2. In-line Raman spectra of EVA copolymer melts showing characteristic VA bands.
Figure 3 shows the stepwise increment in the area of the O–C=O deformation band at 630cm–1 for increasing VA content.
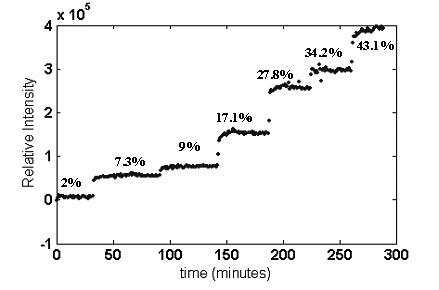
Figure 3. Stepwise increase in 630cm–1 band intensity as EVA copolymers with increasing VA content are extruded.
Figure 4 provides the comparison of the vinyl acetate content obtained from thermogravimetric analysis to the predicted values obtained from the one-factor PLS model.
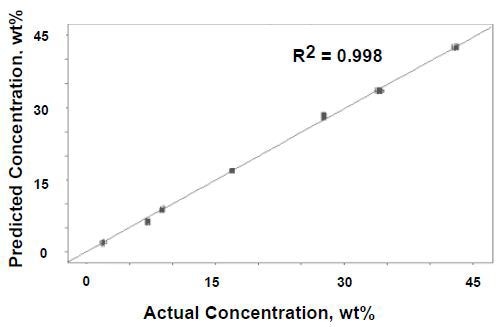
Figure 4. One-factor PLS results for Raman data versus actual concentration.
Conclusion
The results demonstrate the ability of Raman spectroscopy to provide in-line analysis during copolymer production. As a scattering method, Raman spectroscopy requires only a single optical probe instead of a destructive dual transmission probe. It was interfaced well with the process apparatus, thus monitoring the process non-invasively.
The use of visible and NIR excitation radiation makes Raman spectroscopy amenable for fiber optics. This allowed the integration of the Raman probe into the extruder and the remote operation of the spectrograph. Besides simple, flexible and non-invasive sampling, Raman spectroscopy has high sensitivity and provides information-rich content.
About Kaiser Optical Systems
Kaiser Optical Systems, Inc. is a world leader in spectrographic instrumentation and applied holographic technology. Principal products include Raman sensors and instrumentation, advanced holographic components for spectroscopy, and astronomy and ultra-fast sciences. Principal offices and the manufacturing facility are located in Ann Arbor, Michigan.
Their products and services are deployed throughout the world in such diverse applications as pharmaceutical and chemical manufacturing, nanotechnology, telecommunications, education, forensic science, deep-sea exploration, and astronomy. From particles smaller than a human hair to objects as large as planets, their products are providing their customers unique insights into both today’s as well as “age-old” questions.
Kaiser was founded in 1979 to meet the need for diffractive or holographic optics for the avionics market. Kaiser entered the spectroscopy market in 1990 with the introduction of the holographic notch filter. In 1993 Kaiser released their first Raman analyzer, the HoloProbe. In 2013, the company became part of the Endress+Hauser Group.
To better serve the European community, Kaiser opened a new subsidiary in Europe in 1998. Kaiser Optical Systems SARL is located in Lyon, France. Kaiser SARL supervises their distributor network within Europe.

This information has been sourced, reviewed and adapted from materials provided by Kaiser Optical Systems.
For more information on this source, please visit Kaiser Optical Systems.