Analog and digital connections allow compatibility between several Size Exclusion Chromatography (SEC) and High Performance Liquid Chromatography (HPLC) systems and detectors by enabling exchange of injection trigger signals and exchange raw detector signals.
Malvern Panalytical’s OmniSEC software (version 5.1) can now make communication with Waters Empower® software employed for SEC measurements. As a result, users can now directly copy sample sequences from Empower® for synchronization of measurements with the help of injection trigger signals.
This article discuses the method of connecting Viscotek detectors to Waters systems, especially the Waters Alliance® system, as well as the procedure of using the OmniSEC software in conjunction with Empower® software to run separation sequences without any problem.
Fluidic Connections
In order to reduce the number of connections and dead volumes, it is necessary to make fluidic connections of the HPLC tubing as normal. The last comes in the flow sequence is typically refractive index (RI) detectors in order to protect the easily breakable RI flow cell from any additional back pressure created by successive tubing and detector cells.
It is necessary to plumb viscometers last in line as the sample is split in half due to their design. Plumbing the modular RI detectors and modular viscometers in parallel has to be done for using them in the same system. The flow rate to each detector is reduced due to the flow split needed for this setup, negatively affecting the sensitivity of the viscometer. The Triple Detector Array (TDA) features a powerful RI flow cell that allows plumbing the RI detector and viscometer in series. As a result, sensitivity of all of the detectors is maintained at the optimum level.
Electrical Connections
It is necessary to make electrical connections between the instruments for exchanging analog signals and injection trigger signals from modular detectors. The connections are used on the back of different instruments for exchange of signals. Figure 1 shows connections to the e2414 RI detector and Waters Alliance autosampler.
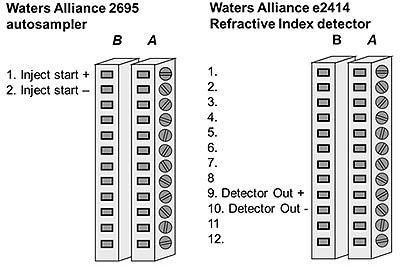
Figure 1. Diagramatic representation of the electrical connections on the Waters Alliance® autosampler and RI detector.
The Viscotek detectors can accept and process these signals in the OmniSEC software. Figure 2A shows a phoenix connection shared by the 270 Dual Detector, Zetasizer µV, and TDA through the OmniFACE A/ D board. The SEC-MALS 20 accepts a trigger signal via a phoenix connector designated as "DIGITAL IN" and analog signals via gold RCA connectors designated as "ANALOG IN."

Figure 2. A: Phoenix connection on the 270, TDA and OmniFACE. B: Connections on the rear of the SEC-MALS 20.
The appropriate connections are joined to connect analog detector signals. The Waters RI detector is connected to the 270, Zetasizer µV, and TDA using the cable (part number CAB2966) with loose wires at both ends. Cable CAB2966 must be connected to the "RI in LOW" and "RI in HI" connections. The RI detector is connected to the SEC-MALS 20 detector using the cable (part number SEC0001) that has loose wires and an RCA plug. Correct polarity needs to be ensured in both cases.
The appropriate connections are joined for exchange of injection trigger signals. Cable CAB2966 is recommended to connect the Waters autosampler to all Viscotek detectors. The Waters autosampler is independent of polarity as it provides a contact closure signal.
Sample Sequence Setup
It is possible to read and import sequences (sample set methods) generated in the Waters Empower® software using the OmniSEC software. A ‘sample set method’ is created in Empower®, followed by opening up the sequence editor in the OmniSEC software.
OmniSEC is logged into Empower® by keying in the username and password of the Empower® user. The relevant project with the desired sequence is selected from the Projects” drop-down menu. This is followed by populating the Sequences” drop-down menu with all of the “sample set methods” present in that Empower® project. A sample set method’ is selected and the load’ button is clicked to populate the sequence with the injections in the method by ignoring non-injection events such as delays. The sequence remains synchronized due to the trigger signals (Figure 3).
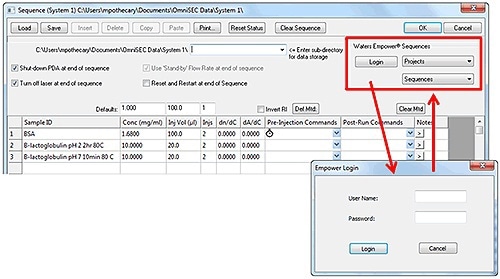
Figure 3. The sequence editor window in OmniSEC and Empower login window.
The run length in the “Acquire – Configure” menu is set prior to commencing the sequence, shortening the runs in OmniSEC by one minute when compared to those in Empower®. As a result, data will be saved by the OmniSEC software, which then moves to the subsequent run and wait for the subsequent injection. As a result, none of the injections triggers are missed during this period.
Data Acquisition
After importing the sequence, “ok” button is clicked to commence the sequence. The detectors are initialized by the OmniSEC software, which then waits for an injection trigger. The sequence is then started in Empower®. The Waters system now performs injections and the OmniSEC software records the injection triggers and performs data collection when the samples are injected by the Waters system.
Conclusion
A Waters chromatography system can be equipped with advanced SEC detection capability such as light scattering by integrating the Viscotek range of detectors. OmniSEC 5.1 can avoid creating duplicate sequences, thanks to its ability to query Empower®. This, in turn, reduces time and errors.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information on this source, please visit Malvern Panalytical.