Octane number ratings describe the way that gasoline will perform under different conditions – the Motor Octane Number (MON) and the Research Octane Number (RON).
Gasoline needs to adhere to set Octane number requirements, meaning that refineries must be able to measure this parameter during processing to ensure that their end product can be sold. In addition to this the Octane number of some components used in gasoline blending, such as reformate, should also be monitored.
The conventional method of Octane number measurement is called the knock engine method. In this method the fuel is combusted and its combustion is compared to that of known standards. This method is lab-based, is time and labor inefficient and cannot be used for process control.
Process Insights’ NIR spectrometers can be used to provide real-time Octane number measurement in refinery environments. Real-time NIR analysis saves both time and money when compared to conventional analysis and the systems built by Process Insights are Designed for process monitoring. Octane number measurement using NIR is well established in the literature, having first been carried out in 1989.1
Measurement Background
The near IR portion of the electromagnetic spectrum includes absorptions for C-H, N-H and O-H stretches. This information can be directly linked to the composition of samples being analyzed in both qualitative and quantitative terms.2
By collecting the NIR spectra of fuel samples with a known Octane number it is possible to develop a model that accurately predicts a samples Octane number from information in its NIR spectrum. This process can be carried out remotely because the NIR systems provided by Process Insights use fiber optics to transmit information between the spectrometer and the sample.
Process Insights’ NIR spectrometer can use multiple channels, meaning up to 12 different parameters (each on a different stream) can be measured simultaneously. This multi-parameter measurement improves cost efficiency.
Experimental
The NIR spectrum, between 1,000 and 1,600 nm, of several different gasoline samples of known Octane number were recorded using an NIR spectrometer from Process Insights. The data from these measurements, taken with a 1 cm path length, is shown in Figure 1 with their Octane numbers shown in Figure 2.
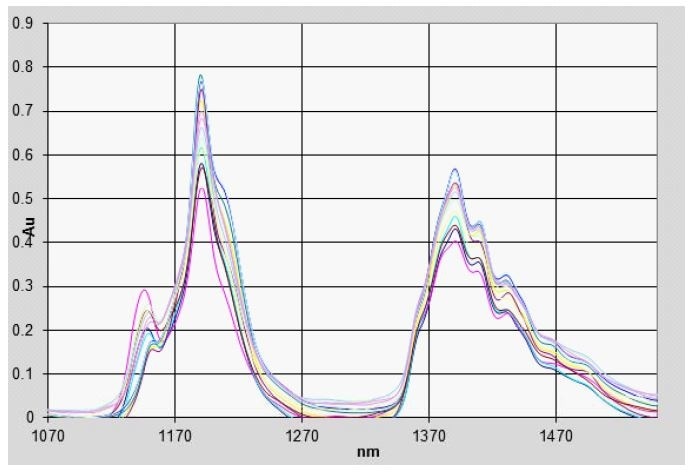
Figure 1. Gasoline Spectra (NIR)
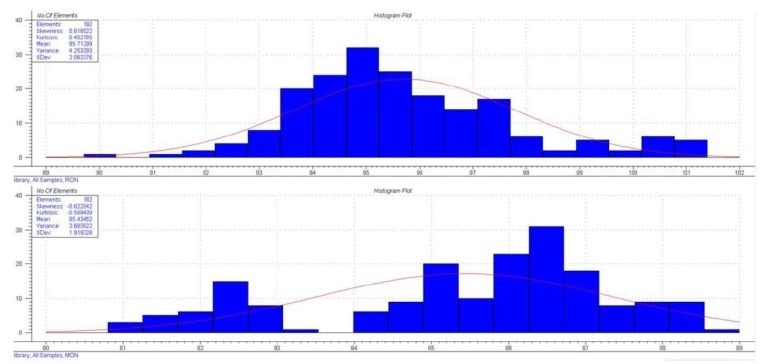
Figure 2. Octane Distribution
The samples show MON values between 80.0 – 89.0 and RON values between 89.7 – 101.4 – standard ranges for blended gas. Using this data and the collected spectra a calibration curve was generated via a PLS regression with the Unscrambler™ software.
Results
The calibration model was used to calculate the Octane number (MON and RON) of gasoline in process streams using an in-situ probe. Figures 3 and 4 show the predictions of RON and MON respectively.
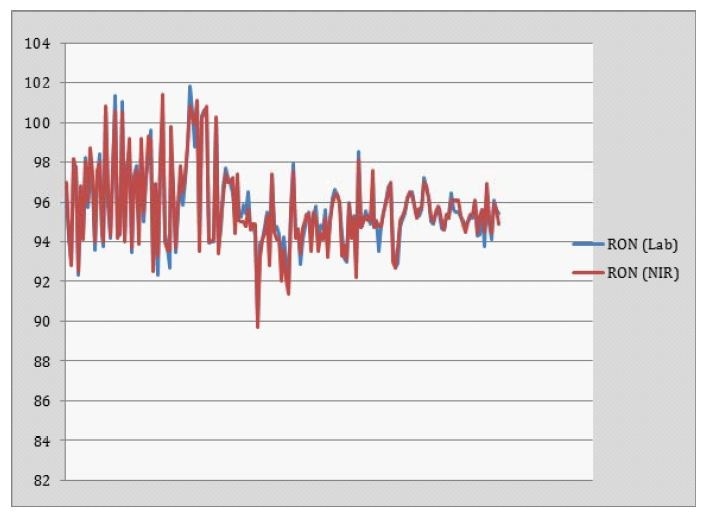
Figure 3. Ron Prediction
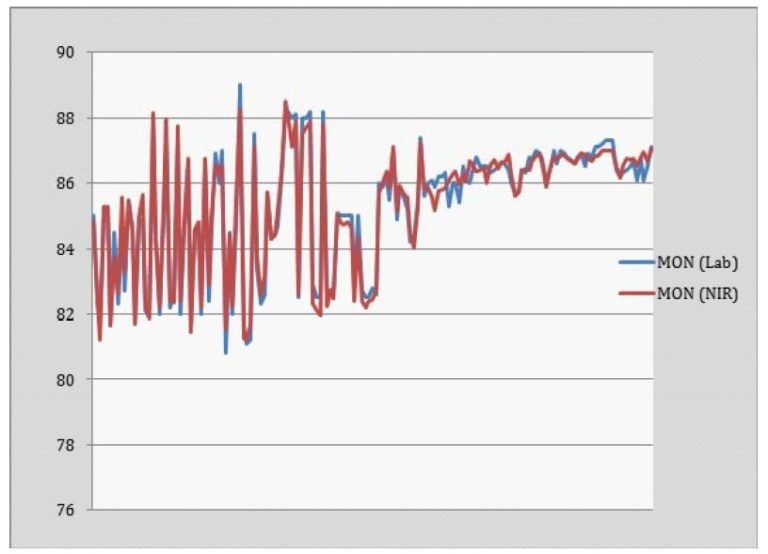
Figure 4. MON Prediction
It was found that the calculated values were in agreement with lab-based determination. Calculation of both RON and MON using this method is simple for different types of gasoline and can be used to clearly differentiate between samples with high, middle and low Octane numbers.
Conclusion
Process Insights NIR spectrometers can be used to rapidly and accurately determine the Octane number of gasoline samples.
The results from the analysis are provided almost instantaneously and can be taken in-situ, avoiding the need for lab analysis and enabling process control. The NIR systems used can determine the RON and MON as well as other parameters such as density, benzene concentration, distillation points and more, for both blended fuels and individual fuel components.
References
- Kelly, Jeffrey J., et al. “Prediction of gasoline octane numbers from near-infrared spectral features in the range 660-1215 nm.” Analytical Chemistry (1989): 313-320.
- D. Burns, E. Ciurczak. Handbook of Near-Infrared Analysis. Marcel Dekker, Inc., 1992.
- E1655, ASTM. “Standard Practices for Infrared, Multivariate, Quantitative Analysis.” n.d.
- H. Martens, T. Naes. Multivariate Calibration. John Wiley & Sons, 1989.

This information has been sourced, reviewed and adapted from materials provided by Process Insights – Optical Absorption Spectroscopy.
For more information on this source, please visit Process Insights – Optical Absorption Spectroscopy.