Engineers Week – an event first celebrated in 1951 by the National Society of Professional Engineers – took place on February 20th-26th, 2022. The theme of this year’s event was “Reimagining the Possible,” which highlights the ways engineers are making a difference in the world.
The aim was to engage children with engineering and STEM topics and activities. To mark the event, this article discusses the significant impact of materials science engineers and outlines some of the most recent advancements in the field.
Materials Science Underlies Pretty Much Everything
The universe does not simply split itself up into clearly defined silos. Thus, the field of materials science is comprised of an extensive range of applications, from the crystal structures crucial to semiconductors to titanium alloys, from nanotech breakthroughs to the development of sustainable bioplastics, and from metalenses to color-tunable LEDs. Materials science has helped shape the modern world.
This interdisciplinary field - also known as “materials science and engineering” - combines elements of both chemistry and physics and concentrates on designing and discovering new materials (especially solids). It has been described as “using the periodic table as its grocery store and the laws of physics as its cookbook.”1
One of the earliest examples of materials science – a long time before the term was known – was the manufacture of bronze about 3,500 years ago. Comprising copper and lead, this metal alloy is stronger than copper, thus allowing Bronze Age craftsmen to hammer and smelt it into practical tools and objects and helping advance the development of human civilization.

Examples of materials science from different eras: Bronze-age axe head (left), and a breakthrough ultra-thin “fishnet” achromatic metalens recently developed at UC Berkeley (right, scale bar represents 5 micrometers). Image Credit: Radiant Vision Systems
The Foundation of Information Technology
Modern materials science effectively helped create the computer and consumer electronics industry. It all started with the development of an ultra-pure form of silicon (now called electronic-grade silicon) which is made by eliminating impurities such as boron, phosphorous, and carbon. To produce integrated circuits, silicon must contain <0.1 part per trillion of these materials.
Between 1955 and 1990, enhancements and innovations in semiconductors “increased the performance and decreased the cost of electronic materials and devices by a factor of one million - an achievement unparalleled in the history of human technology.”2
To put this into perspective, Applied Materials CTO Omkaram Nalamasu explains, “To build today’s smartphone in the 1980s, it would cost about $110 million, require nearly 200 kilowatts of energy (compared to 2 kW per year today), and the device would be 14 meters tall.3
Today telecommunications infrastructure and systems are dependent on “various crystalline semiconductors; metalized film conductors; dielectric films; solders; ceramics, and polymers formed into substrates on which circuits are assembled or printed; and gold or copper wiring and cabling.”4
The core material in a significant number of LEDs and display devices is indium gallium nitride (InGaN) – a manufactured mixture of gallium nitride and indium nitride. When grown into crystals, InGaN facilitates the bandgap tuning required to emit light across the infrared, visible, and ultraviolet spectra.
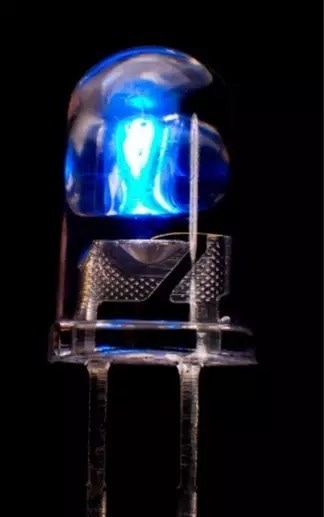
A blue LED, made possible by the alloy indium gallium nitride. Image Credit: Radiant Vision Systems
Materials scientists are persistent in their pursuit of engineering novel smart materials with properties not typically found in nature. For instance, graphene is a two-dimensional version of diamond or graphite, a one-atom-thick layer of carbon in a hexagonal lattice structure.
Graphene is a remarkable material as “it is 200x stronger than steel by weight. Over the last 10 years, we’ve been able to use graphene to make new kinds of electronics, very high-performance transistors, new kinds of sensors, and new kinds of composites based on its unique properties.”5
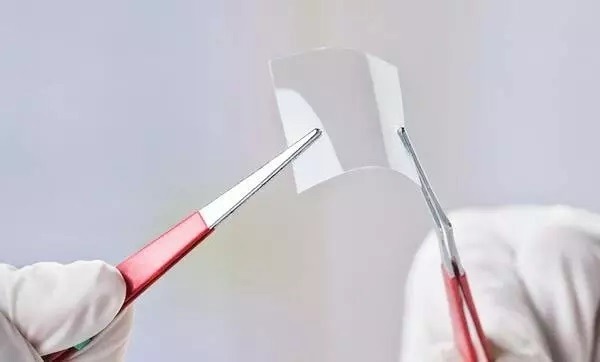
A flexible, transparent sheet of graphene in the lab. Image Credit: Radiant Vision Systems
Recent Materials Science Advancements
Next, we present a number of examples of emerging applications and recent technology innovations where materials science has played a key role.
Safer than Lithium Batteries
Lithium-ion batteries are the primary energy source of most of today’s electronic devices and vehicles. However, they have two major drawbacks: firstly, they contain a liquid electrolyte that can be extremely flammable, and secondly, the global lithium supply chain can lead to heightened geopolitical tensions.
Recently, a group of researchers at the University of Geneva have developed a solid electrolyte material that shows the conductive properties required for batteries. The material (carbo-hydridoborate) is produced using sodium, an element that is less expensive than lithium and abundantly available almost anywhere on earth.
“Frozen Smoke” aka Aerogels
A diverse group of porous materials, aerogels are considered the lightest solids in the world, comprised of up to 99.98% air by volume and harboring uniquely special properties.
For instance, “Transparent super-insulating silica aerogels exhibit the lowest thermal conductivity of any solid known. Ultrahigh surface area carbon aerogels power today’s fast-charging supercapacitors. And ultra-strong, bendable x-aerogels are the lowest-density structural materials ever developed.”6
Unlike garden-variety gels, aerogels are dry and are made up of the low-density solid framework of a gel that remains after extracting the moisture.
NASA made use of silica-based aerogels for insulation on the Mars rover, and they have been utilized as an absorbent in chemical spill clean-ups.
Additionally, “carbon aerogels are used in the construction of small electrochemical double layer supercapacitors. Due to the high surface area of the aerogel, these capacitors can be 2,000 to 5,000 times smaller than similarly rated electrolytic capacitors. Aerogel supercapacitors can have a very low impedance compared to normal supercapacitors and can absorb/produce very high peak currents.”7

A block of silica aerogel. (Image source).
Folding Displays
LG Chem (sister company of LG Display) has created a new cover window for foldable-screen devices such as smartphones and laptops. Known as “Real Folding Window,” it is manufactured by applying special coating materials to PET film (a thin type of plastic) to produce a surface as hard as glass yet with the flexibility of plastic.
The material is intended to overcome device-related issues such as fold impressions or cracks at the hinged part of a screen. It also improves the cover’s heat resistance and other mechanical properties.
Making the Impossible Possible
MIT-based engineers have applied a novel polymerization process to produce an entirely new material that is stronger than steel yet lighter than plastic. It can be manufactured easily in significantly large quantities and can potentially revolutionize the industrial landscape.
Uses include lightweight, durable coatings on everything from bridges and infrastructure to car parts and cell phones. Effectively a new form of plastic, the polymer has the ability to self-assemble into 2D sheets - a process scientists had thought to be impossible, hence its nickname “Impossible Plastic.”
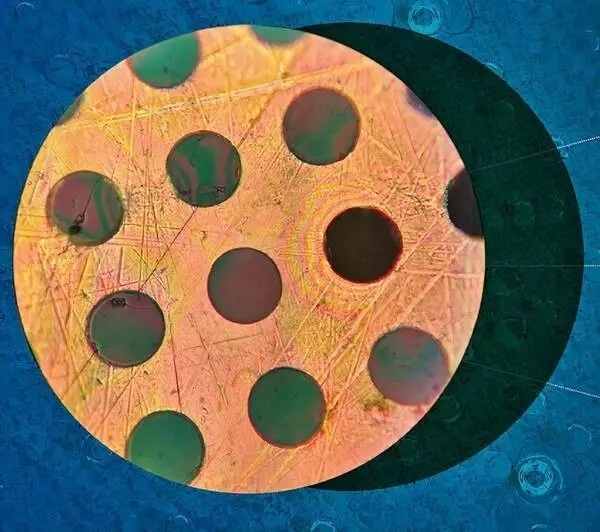
“Impossible” two-dimensional polymeric film that is twice as strong as steel but as light as plastic. (Image source)
This article lists just a few of the latest developments materials scientists and engineers are working on in laboratories and research facilities around the world. Amazing new substances are being developed every day with the potential to revolutionize medicine, technology, and industry and help create a more sustainable planet.
References
- Diamandis, P., “3 Major Materials Science Breakthroughs - and Why They Matter for the Future,” SingularityHub, May 21, 2020.
- Materials for computers and communications, Britannica. (Accessed February 16, 2022)
- Ibid.
- Diamandis, P., “3 Major Materials Science Breakthroughs - and Why They Matter for the Future,” SingularityHub, May 21, 2020.
- Diamandis, P., “Materials Science and Technology Convergence.” Blog post, March 23, 2016. (Accessed February 16, 2022)
- Aerogel.org. (Accessed February 16, 2022)
- “Aerogel.” New World Encyclopedia. (Accessed February 16, 2022)

This information has been sourced, reviewed and adapted from materials provided by Konica Minolta Sensing Americas.
For more information on this source, please visit Konica Minolta Sensing Americas.