Platinum group metals (PGMs) - like platinum (Pt), palladium (Pd), and rhodium (Rh) - have wildly varying compositions and volatile prices, making them more important than ever in the purchase, trading, and recycling of spent catalytic converters. In 2020, the demand for Pt, Pd, and Rh was 215, 308, and 31.2 tons, respectively.

Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
The automotive catalyst industry imbibed approximately 32% of total Pt, 85% of total Pd, and 90% of total Rh.1 In the same year, 33.7 tons of Pt, 41.2 tons of Pd, and 7.3 tons of Rh1 were retrieved from recycling programs, primarily from spent catalytic converters, accounting for approximately $12 billion of the cumulative average price of fine metals in 2020.
Pt, Pd, and Rh prices have fluctuated dramatically over the last 15 years based on supply, demand, and speculation (Figure 1). These fluctuations, as well as the toughening of emission regulations, had a direct impact on the compositions of the catalysts, which in turn had a significant impact on demand. Pd and Rh prices increased due to a supply shortage in 2020-2021.
The value of a single catalytic converter’s PGM composition can range from less than $100 to more than $1000. As these materials are frequently powdered and blended when traded, traders and recyclers must be able to:
- Detect single catalytic converters or blends holding high levels of PGMs
- Catch deceitfully blended and adulterated material
- Pay or get paid a fair price

The physical recycling begins with decanning or the removal of the shell and extraction of the honeycomb-shaped material inside the catalytic converter. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
It is invaluable to accurately identify the components of Pt, Pd, and Rh of spent automotive catalytic converters in real-time to maximize profit and avoid significant financial loss.
![Pt, Pd and Rh prices 2006-2021 (Data collected from [2]).](https://www.azom.com/images/Article_Images/ImageForArticle_21874_16583095814476100.jpg)
Figure 1. Pt, Pd and Rh prices 2006-2021 (Data collected from [2]). Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
Thermo Scientific™ Niton™ Handheld XRF Analyzers
Thermo Scientific provides a complete line of handheld XRF analyzers for accurately measuring Pt, Pd, and Rh in automotive catalytic converters:
- The Niton XL3t 951 and 981 GOLDD+ analyzers are high-end models prepared with a 50kV tube and a large silicon drift detector. The Niton XL3t 951 and 981 GOLDD+ analyzers also identify light elements like magnesium, aluminum, or silicon and have better sensitivity to distinguish low levels of PGMs.
- The Niton XL2 501 analyzer provides great value for money while upholding a high level of efficiency for PGMs.
- The Niton XL5 Plus analyzer is the top-of-the-line model with state-of-the-art hardware comprising an extra-large silicon drift detector and a 5W tube offering ultimate sensitivity. Productivity is further improved due to shorter measurement times.
- The Niton XL3t 501 and 801 analyzers, the mid-range models prepared with 50 kV and a filter changer, permit optimum beam conditions and offer more detailed and precise results for PGMs.
The pre-calibrated Automotive Catalyst Mode employs a fundamental parameter approach (FP), an algorithm that rectifies the impact of all elements in the sample using theoretical constants. FP is appropriate for analyzing spent materials, which now exhibit unparalleled fluctuation in composition.
Assessing rare earth elements, zirconium, lead, and other elements that may be present in high concentrations in spent catalysts is crucial in obtaining precise Pt, Pd, and Rh results.

Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
Source: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
| |
 |
 |
 |
 |
| Models |
XL2 501 |
XL3t 501/801 |
XL3t 951/981 GOLDD+ |
XL5 Plus |
| Tube Max Power1 |
2W |
2W |
2W |
5W |
| Tube max voltage2 |
45 kV |
50 kV |
50 kV |
50 kV |
| Detector technology3 |
Si-PIN |
Si-PIN |
Large SDD |
Extra-Large SDD |
| Number of beams for PGMs / total number of beams4 |
1 for PGMs/1 |
2 for PGMs/3 |
2 for PGMs/4 |
2 for PGMs/4 |
| Typical total measurement time |
80 seconds |
120 seconds |
60 seconds |
30 seconds |
| Limits of detection (3σ) @ typical measurement time 5 |
Pt: 20 ppm
Pd: 13ppm
Rh: 13 ppm |
Pt: 16 ppm
Pd: 7 ppm
Rh: 6 ppm |
Pt: 10 ppm
Pd: 5 ppm
Rh: 5 ppm |
Pt: 9 ppm
Pd: 3 ppm
Rh: 3 ppm |
| Accuracy6 |
?? |
??? |
???? |
???? |
| Precision7 |
? |
?? |
???? |
???? |
| Sensitivity8 |
? |
?? |
???? |
???? |
| Productivity9 |
? |
?? |
???? |
???? |
1. Higher power provides better precision and allows shorter measurements at constant precision. Sensitivity is improved also. 2. Higher voltage obtains better precision and accuracy for elements such as Pd, Rh, and rare earth elements. 3. Silicon drift detector (SDD) technology collects a higher count rate and improves precision, and sensitivity, and enables light element detection vs. Silicon-positive-intrinsic-negative (Si-PIN) technology. 4. More beam conditions with different filters mean better precision and sensitivity for lines with different energies. 5. The limits of detection depend on the testing time, the interferences/matrix, and the level of statistical confidence. 6. Indicates the degree of closeness to the true value. 7. Indicates how repeatable and reproducible measurement is. 8. Indicates the ability to detect small amounts of PGMs. 9. Economic productivity: sample throughput and economic recovery of Pt, Pd, and Rh.
Sample Preparation
The gathered catalysts with ceramic substrate go through a decanning operation, which is the extraction of the honeycomb-structured ceramic from the steel case. The automotive catalyst material is composed of a ceramic substrate, primarily cordierite, which has been coated with a PGM-containing wash coat.
As a result, the entire ceramic is sorted, crushed, milled, and frequently mixed with other catalysts. Converters with a metallic substrate, on the other hand, are crushed initially, and then the metallic parts are distinguished from the wash-coat powder comprising precious metals using magnets and winnowing.
Due to this advancement, the PGM content of wash coats from metallic catalytic converters is significantly higher than that of ceramic converters. In both cases, the materials are pulverized to a particle size of less than 250 µm, loaded into XRF cups for analysis, and placed in a test stand.

Materials are pulverized to a maximum of 250um and loaded in XRF sample cups or sample bags for analysis. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
Table 1. Typical results of analysis for a commercially available automotive catalyst-certified reference material. Source: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
| Materials/Elements |
Niton value (ppm) |
Certified value (ppm) |
| BAM ERM-504a |
| Pt |
1495 ± 41 (2σ) |
1414 ± 15 |
| Pd |
1583 ± 19 (2σ) |
1596 ± 6 |
| Rh |
210 ± 8 (2σ) |
210 ± 4 |
Results
Table 1 shows the results of an analysis of one commonly produced reference material. Pt, Pd, and Rh values measured with the Niton XL5 Plus analyzer agree very well with certified values. Figures 2 to 4 depict the similarity curves between lab results and concentrations evaluated with the Niton XL5 Plus analyzer.
There is an outstanding linear correlation between lab and measured values for all elements de?ned in 120 samples over a range of concentrations: both the slopes and the coefficients of determination R2 are similar to one.
The average disparity between laboratory and measured values for Pt, Pd, and Rh was 5.0%, 3.9%, and 2.9%, respectively. These findings indicate the analytical robustness in the face of significant matrix changes and sometimes very high concentrations of noteworthy concomitant elements such as cerium, lanthanum, zirconium, lead, iron, or chromium observed in the 120 samples.
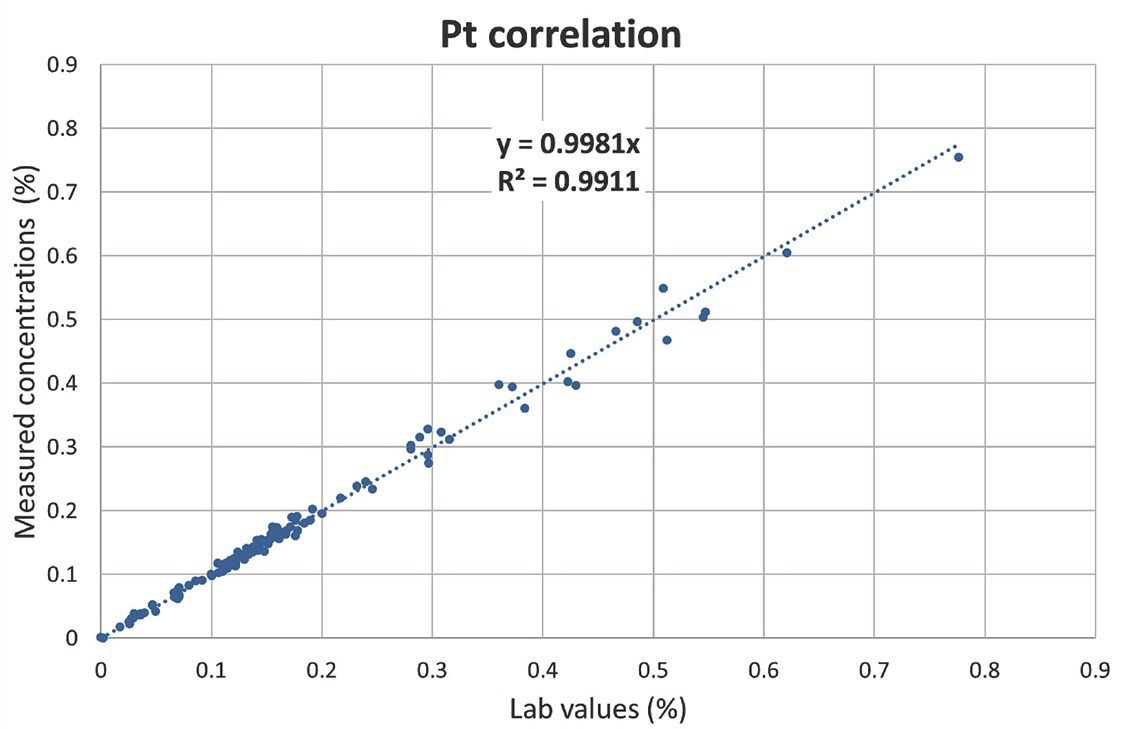
Figure 2. Correlation curve for Pt. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
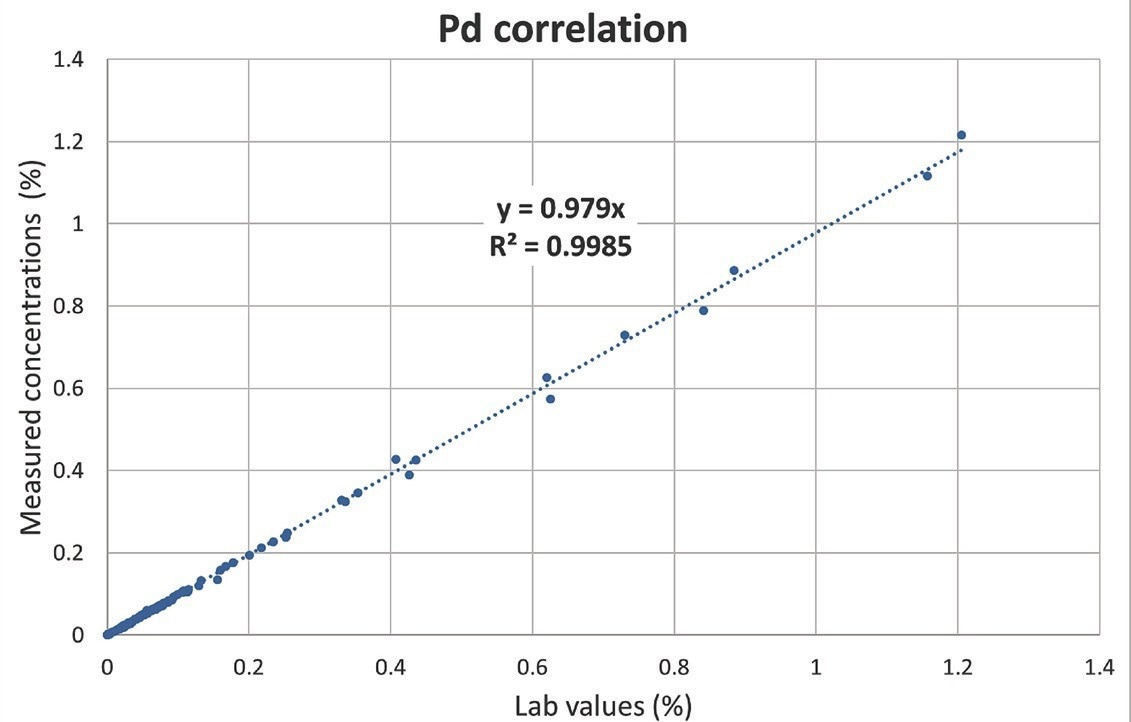
Figure 3. Correlation curve for Pd. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
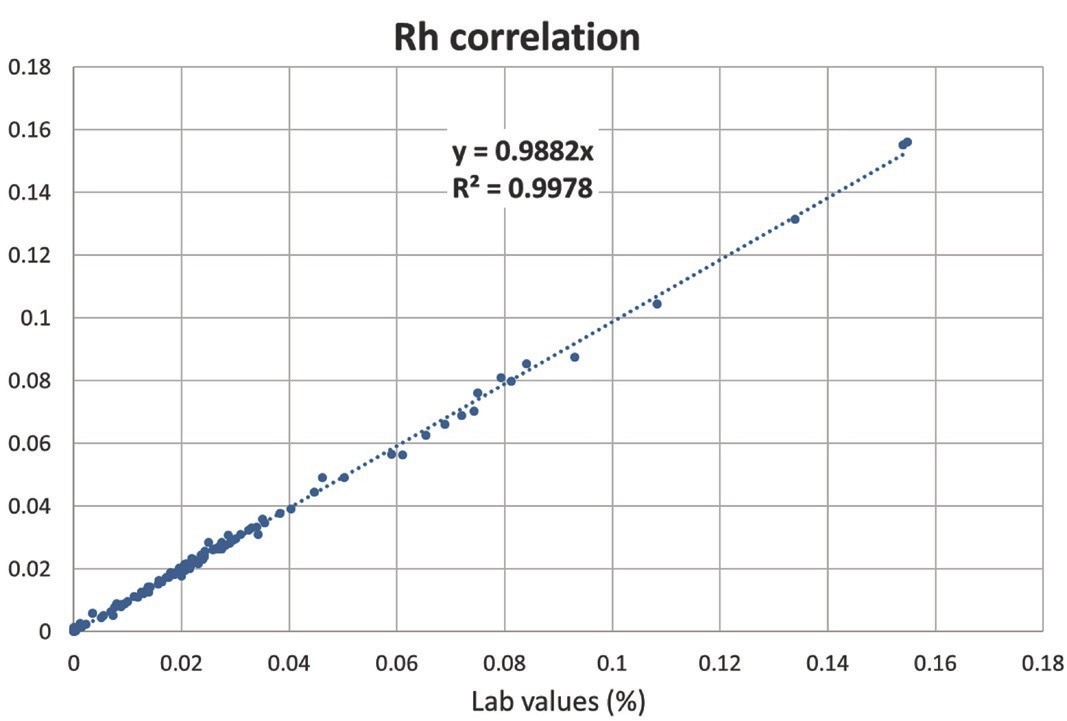
Figure 4. Correlation curve for Rh. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
Proficiency Test
Under lab code #9, Thermo Fisher participated as a laboratory in a proficiency test organized by the well-known XRF company Fluxana3 to analyze two samples FLX-CRM 132 and FLX-CRM 133. Table 2 and the Fluxana report4 present the results, which prove the outstanding precision obtained using the Thermo Fisher Niton XL3t 980 analyzer (same instrument as XL3t 951/981).
Table 2. Results of the proficiency test. Source: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
| |
Mean Niton XL3t 980 Value ± sd (ppm) |
Certified Value ± uncertainty (ppm) |
| FLX-CRM 132 |
| Pd |
1668 ± 18 |
1673 ± 27 |
| Pt |
1758 ± 16 |
1770 ± 73 |
| Rh |
284 ± 6 |
295 ± 12 |
| FLX-CRM 133 |
| Pd |
1062 ± 10 |
1075 ± 40 |
| Pt |
460 ± 4 |
465 ± 28 |
| Rh |
232 ± 4 |
242 ± 18 |
Conclusion
Niton handheld XRF analyzers accurately analyze ceramic catalytic converters from gasoline or diesel engines, wash coats from metallic converters, and material blends.
Niton XRF analyzers are the perfect tool for accurate analysis for individuals and businesses who trade and recycle used automotive catalytic converters. The following are the primary advantages of using Niton XRF analyzers:
- Real-time examination
- Fast return on investment and low cost of ownership
- Ease of use vs. lab analysis needing special know-how
- Outstanding precision demonstrated over many years of operation
References
- Johnson Matthey, Pgm market report February 2021. http://www.platinum.matthey.com/
- http://www.platinum.matthey.com/
- https://www.fluxana.com/service/proficiency-testing
- https://fluxana.com/images/products/Ringversuch/FINAL_ Report_RV_2017_01.pdf

This information has been sourced, reviewed and adapted from materials provided by Thermo Fisher Scientific – Handheld Elemental & Radiation Detection.
For more information on this source, please visit Thermo Fisher Scientific – Handheld Elemental & Radiation Detection.