In the early stage of pharmaceutical product development, the rate-determining step has shifted from the synthesis phase to the analysis phase. There is a need for innovative techniques that allow for rapid screening of drug candidates and also enable data processing. The necessity to process a large number of drug candidates has resulted in the development of the field of high-throughput screening (HTS).
Screening of Drug Candidates
Polymorphic form is one of the most critical characteristics of solid-state drug candidates. A large number of molecular solids, for instance, organic drug candidates undergo crystallization in varied polymorphic forms. These solids vary only in terms of the space group, which the extended solid belongs to or the manner in which the constituent molecules conform to each other.
Pseudopolymorphic versions of compounds like hydrates and solvates are often considered along with polymorphs. Variations in solvation or hydration and in polymorphic form could considerably impact the drug’s stability, dissolution, and bioavailability properties. Therefore, in drug manufacture it is important to characterize these forms or versions early in the lifetime of a candidate and create only the preferred polymorphic version.
Analytical Methods
X-ray diffraction (XRD) along with infrared (IR) spectroscopy is generally utilized for differentiating polymorphs; however, IR and the subsequent data collection require sample preparation which is difficult to automate, a key requisite in a screening tool.
Nevertheless, technological advancements have made Raman spectroscopy open to rapid and automated data analysis. This technique is a form of vibrational spectroscopy that provides contact-free analysis and requires no sample preparation.

Figure 1. RamanRxn1 HTS with a wellplate
Kaiser Optical Systems’ RamanRxn1™ High Throughput Screener (HTS) is designed to allow automated and rapid data collection and analysis on little amounts of solid in a wellplate (Figure 1).
In automated experiments, large amounts of information are produced that can be studied by means of chemometric techniques. Raman spectroscopy can track a candidate form from the discovery phase through to the manufacturing phase, thanks to its instrumentational and analytical capability. This capability improves the existing knowledge for transfer at every step of scale-up while a candidate API is converted into a drug product.
Identification of Polymorphs
In an evaporation experiment, three products were used in which one drug candidate was crystallized by gradual evaporation from three different solvents (Figure 2). At the left side of the figure, the crystals’ white-light images indicate that three different crystalline versions have been created, which are validated in situ and sans any sample preparation by the Raman spectra of three exclusive polymorphs illustrated at the right of the figure.
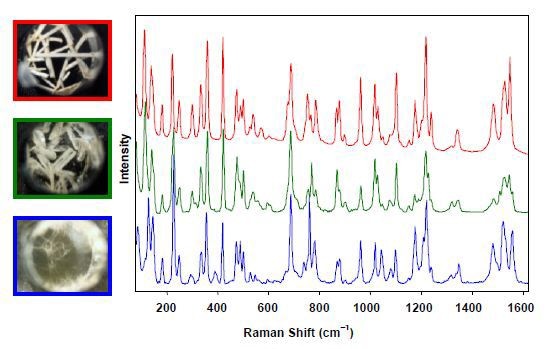
Figure 2. Raman spectra of three unique polymorphs formed by crystallizing a drug candidate by evaporation from three different solvents.
Following Raman analysis, these polymorphs can be assessed individually for their suitability in the preferred dosage form. The application of Raman HTS is a paradigm of PAT viable methods in research and development.
Solvates
Other than hydrates, solvates are not often used in drug development, but they are used as intermediates in the development of the end drug product. Therefore, it is important to distinguish the solvates to better understand the formation of the end product. While In these experiments, similar solvents were used, the Raman spectra of numerous solvates (Figure 3) are satisfactorily different, making it easy to differentiate them.
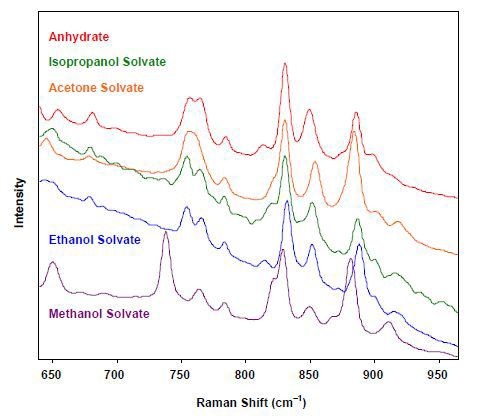
Figure 3. Raman spectra of solvates
Channel Solvates
In other solvates, the solvent molecules are part of the crystal lattice. In contrast, the solvent molecules in channel solvates are found in channel-like interstices between the drug molecules. Channel solvates pose unique challenges as they could integrate different range of solvents and also water from the atmosphere. They can be easily identified by the variations in a slurry’s Raman spectra along with the solvent and crystals separated from the slurry (Figure 4).
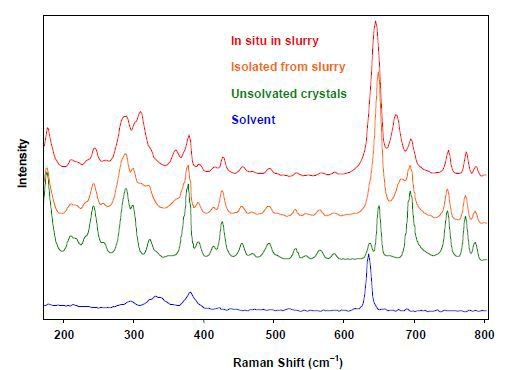
Figure 4. Raman spectra of channel solvates showing the change in position of the solvent peak between crystals in slurry, dry solvated crystals, dry unsolvated crystals, and neat solvent
Conclusion
Raman spectroscopy is a suitable technique for HTS screening of polymorphs during the early stage of product development. Unlike XRD, the technique is just as effective for analyzing slurries and solids and offers the required flexibility to manage a wide range of drug candidates.
Using Kaiser’s RamanRxn1 HTS, each well in these experiments can be examined in a few seconds. In spite of varying well fills, each well can be automatically focused to provide data of high quality. In contrast to a traditional Raman microscope, the entire wellplate and data collection is completed automatically without staff intervention.
About Kaiser Optical Systems
Kaiser Optical Systems, Inc. is a world leader in spectrographic instrumentation and applied holographic technology. Principal products include Raman sensors and instrumentation, advanced holographic components for spectroscopy, and astronomy and ultra-fast sciences. Principal offices and the manufacturing facility are located in Ann Arbor, Michigan.
Their products and services are deployed throughout the world in such diverse applications as pharmaceutical and chemical manufacturing, nanotechnology, telecommunications, education, forensic science, deep-sea exploration, and astronomy. From particles smaller than a human hair to objects as large as planets, their products are providing our customers unique insights into both today’s as well as “age-old” questions.
Kaiser was founded in 1979 to meet the need for diffractive or holographic optics for the avionics market. Kaiser entered the spectroscopy market in 1990 with the introduction of the holographic notch filter. In 1993 Kaiser released their first Raman analyzer, the HoloProbe. In 2013, the company became part of the Endress+Hauser Group.
To better serve the European community, Kaiser opened a new subsidiary in Europe in 1998. Kaiser Optical Systems SARL is located in Lyon, France. Kaiser SARL supervises their distributor network within Europe.

This information has been sourced, reviewed and adapted from materials provided by Kaiser Optical Systems.
For more information on this source, please visit Kaiser Optical Systems.