An alternative, biological way of xylose conversion was examined within the European Valor Plus research project. A Candida yeast strain was used to convert xylose from a hemicellulose hydrolysate into xylitol.
HPLC analysis of the fermentation mash showed successful conversion of xylose to xylitol. Earlier batch HPLC experiments suggested the possibility to apply SMBC for this purification task. The separation was carried out in isocratic mode on polymer-based Eurokat columns, with the target substance xylitol eluting towards the end of the chromatogram, all factors allowing a SMB process.
The continuous chromatography method, SMB chromatography separates binary or pseudo-binary mixtures into pure fractions or substances. This process results in higher yields of purified substances and at the same time consumes less packing material and eluent, when compared to conventional batch chromatography.
Results
To set up a SMB process, a number of parameters of the separation process had to be established first. Separation at three varying temperatures (40 °C, 50 °C and 60 °C) was first tested.
It was observed that separation at 50 °C provided the most favorable results and hence this was selected for the purification of xylitol. Overload studies with a 1:2 dilution of the fermentation mash demonstrated an almost baseline separation of mannitol and xylitol. The chromatogram was divided in the raffinate fraction (except for xylitol) and the extract fraction (xylitol), as shown in Figure 1.
A substance eluting with the dead time of the system was established and as a result an open-loop set-up was selected with a waste outlet. Next, the retention times of the column porosity and substances were established and used for the set-up of the SMB process. These values and the PurityChrom® MCC software were used to obtain the flow rates of the pumps as well as different zones in the process (Figure 2). Following six cycles, samples from the raffinate, extract and waste were collected and examined.
Then, using a rapid analytical method (additional results, Figure A1), the process was evaluated quickly. A more comprehensive analysis showed pure and contamination-free xylitol in the extract (Figure 3, red line). Moreover, no xylitol was detected in the waste or raffinate (Figure 3 blue/green lines).
This SMB process helped to purify 1.8 g/h xylitol with 100% purity and recovery. Compared to the batch process, the yield of the SMB process is greater by the factor of seven.
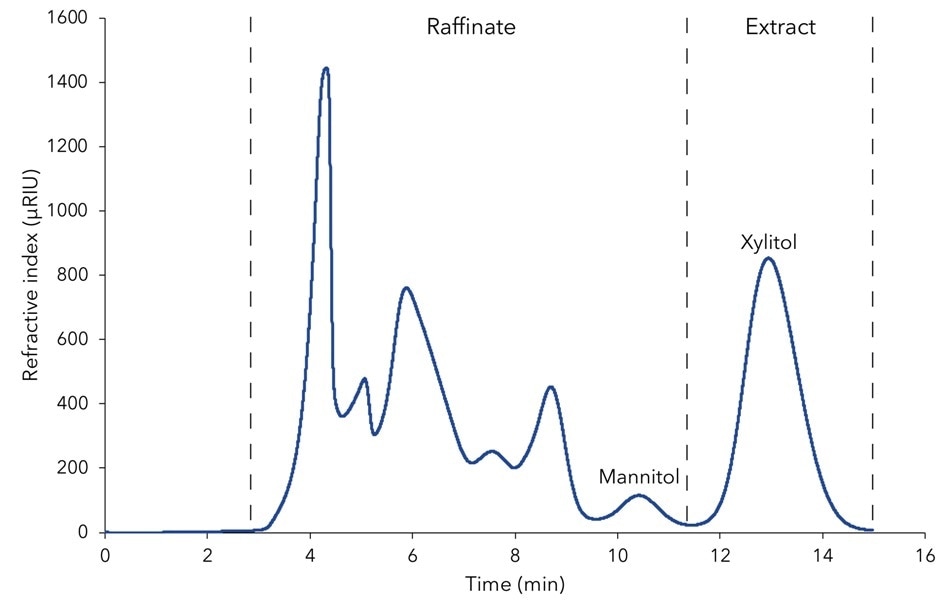
Figure 1. Design of SMB process on chromatogram of fermentation mash with indication of the two fractions "raffinate" and "extract"; 1 mL injection; Eurokat Ca 150 x 20 mm, 25 -56 µm particles; 4 mL/min; 50 °C.
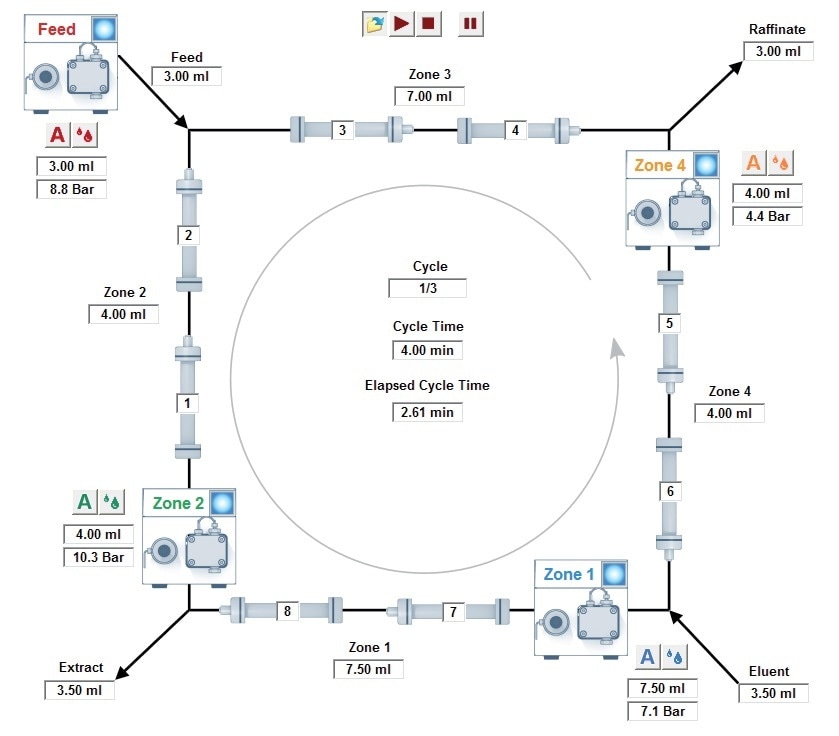
Figure 2. Example scheme of the SMB process set-up with the four pumps, the out – and inlets, the 8 columns, four zones, indication of flow rates, pressure and cycle time; PurityChrom MCC software.
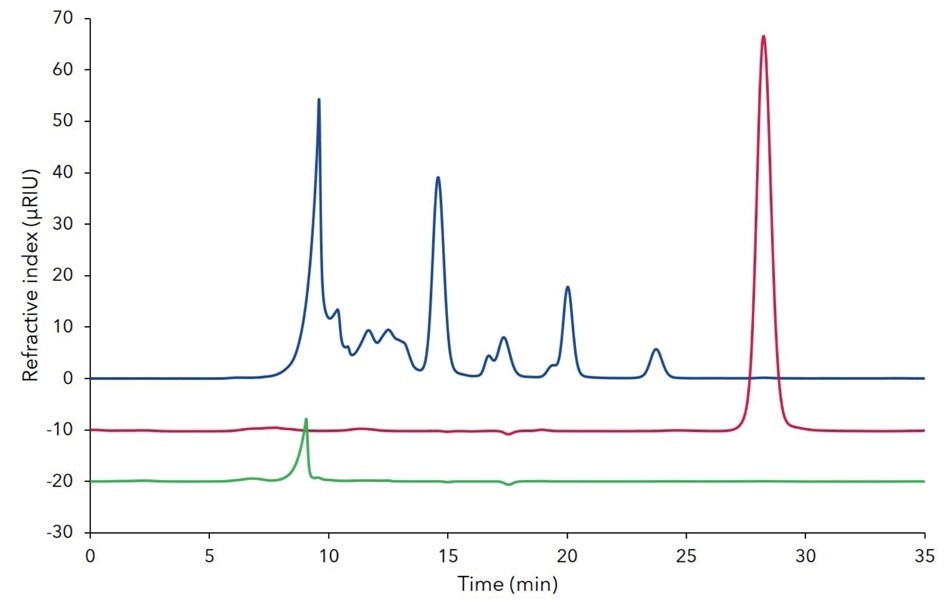
Figure 3. Overlay of analytical chromatograms of raffinate/all but xylitol (blue), extract/xylitol (red) and waste (green) from the SMB process at the 6th cycle; 20 µL injection; Eurokat Ca 300 x 8 mm + pre-column; 10 µm particles, 0.5 mL/min; 75 °C.
Materials and Method
The SMB standard configuration includes four AZURA® Assistants ASM 2.1L with four P 4.1S Pumps and seven multiposition valves (10/50 mL/minute). Temperature was controlled with the SMB oven and flow was regulated with two CORI-Flow M13 flow meters.
For purification, 8 identical Eurokat Ca 150 x 20 mm columns (sulfonated cross-linked styrene-divinylbenzene copolymer) with 25-56 µm particles were used. Eurokat Ca columns 300 x 8 mm, 10 µm particles and dedicated analytical sugar system (VFD0151) were used to perform the analysis.
Conclusion
Using the AZURA® SMB system, xylitol was purified with high purity and yield from the fermentation mash. The purification process promotes a significant higher throughput and thus higher yield of xylitol as traditional batch chromatography.
The xylitol concentration in the original mash limits the actual throughput. Since the developed approach is extremely robust, separation of two to four times more concentrated mash should provide the same separation results.
Additional Results
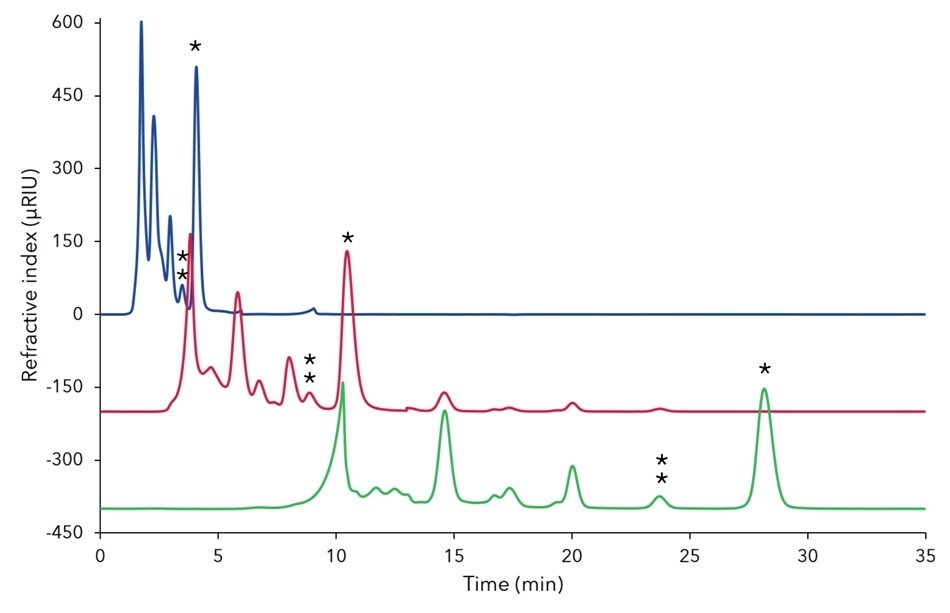
Figure A1. Comparison of separation profiles of fermentation mash using Eurokat Ca columns with different length for a fast, analytical method; blue - 2 x 30 x 8 mm (0.7 mL/ minute), red (offset=-200) -120 x 8 mm (0.7 mL/minute), green (offset=-400) - 300 x 8 mm (0.5 mL/minute); 20 µL injection; 75 °C;
* - xylitol, ** - mannitol
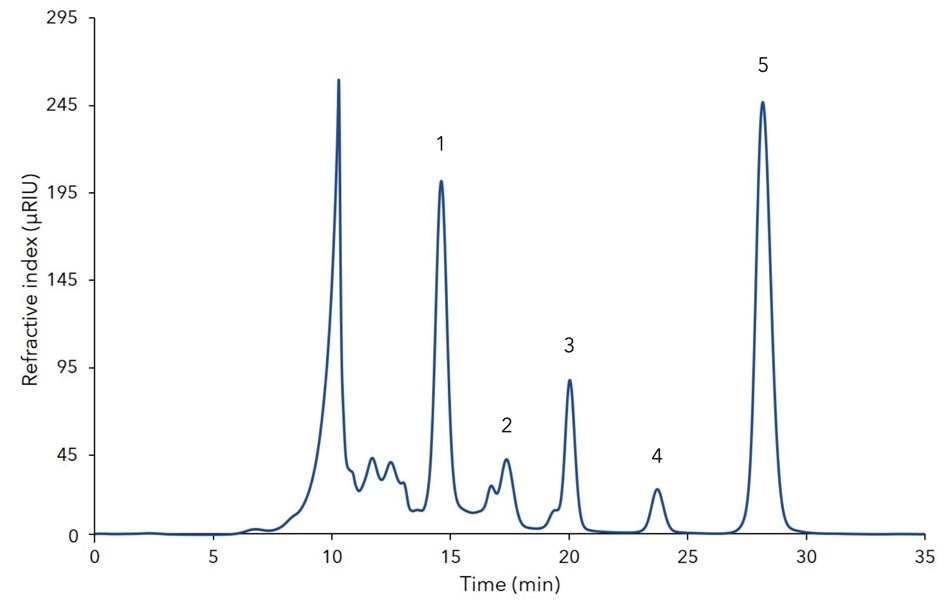
Figure A2. Analytical chromatogram of fermentation mash showing five identified substances; 20 µL injection; Eurokat Ca 300 x 8 mm; 10 µm particles; 0.5 mL/min; 75 °C;
1 - xylose, 2 - arabinose, 3 - glycerol, 4 - mannitol, 5 – xylitol
Table A1. Results of data feed analysis presented as mean of four replicates with standard deviations indicated.
| |
Concentration (mg/mL) |
| Xylose |
38.44 ± 0.13 |
| Arabinose |
8.67 ± 0.04 |
| Glycerol |
18.63 ± 0.13 |
| Mannitol |
5.59 ± 0.08 |
| Xylitol |
61.91 ± 0.34 |

 Download the White Paper for More Information
Download the White Paper for More Information

This information has been sourced, reviewed and adapted from materials provided by KNAUER Wissenschaftliche Geräte GmbH.
For more information on this source, please visit KNAUER Wissenschaftliche Geräte GmbH.