Molar mass plays a critical part in the potency, solubility and stability of polysaccharide-based vaccines. The production process involves an important depolymerization stage to decrease the polysaccharide’s initial weight-average molar mass, Mw, from more than 1800 kDa down to less than 350 kDa.
Present methods to monitor molar mass during polysaccharide production utilize off-line SEC-MALS analysis, during which a single run can last 30 minutes. With a normal depolymerization time of approximately 90 minutes, off-line analytics are not able to supply timely feedback on reaching the endpoint. Real-time multi-angle light scattering (RT-MALS) fulfills this requirement and makes sure that the process ends as soon as the endpoint criterion is attained.

Image Credit: Shutterstock.com
Materials and Methods
A polysaccharide solution was depolymerized by ultrasonication at 20 kHz. Almost in real time, molar mass was monitored online by using an Agilent HPLC pump to constantly drive a fraction of the solution from the reactor through an ultraDAWN™ RT-MALS instrument. The solution was continuously diluted by a factor of 10 in order to lessen viscosity for flow through capillary tubing.
The flow rate was 5 mL/min, which led to a lag time (RTD, residence time delay) of only three minutes between the time product was drawn from the reactor and time of measurement. OBSERVER™ software was configured to control the HPLC pump, gather data from the ultraDAWN™, calculate Mw 30 times per minute, and activate a trigger to halt the sonication when Mw < 350 kDa, as laid out by the release specifications.
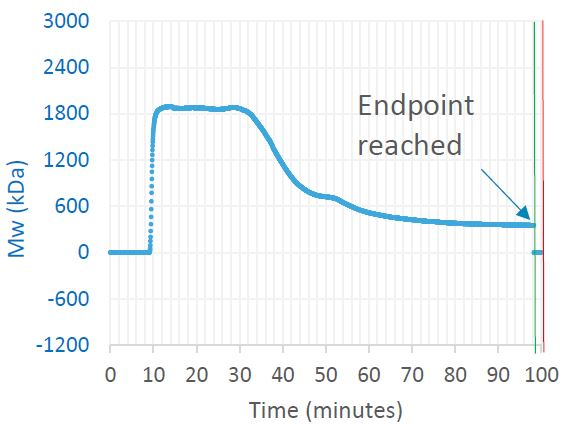
Figure 1. Trace in OBSERVER software of weight-average molar mass measured by ultraDAWN, indicating the achievement of the desired process endpoint. Image Credit: Wyatt Technology
Results and Discussion
RT-MALS clearly tracked reduction of the polysaccharide’s Mw and triggered reaction shutdown once it dropped beneath 350 kDa. SEC-MALS analysis of the final product verified the desired critical quality attribute (CQA) value. Many off-line analyses were removed, the time spent by the drug substance in this part of the process was reduced by a quarter, and total person-hours devoted to this portion of the process was reduced by at least 50%.
Conclusions
As they are high-value-add products, applying PAT to polysaccharide antigens offer significant cost benefits which are gained by in-process determination of the primary attribute impacted by the specific process. Here, the depolymerization process aim is to alter the polymer’s molecular weight, so being able to determine molecular weight almost in real time via RT-MALS reduces uncertainty caused by process drift or differences in raw material properties. RT-MALS control leads to perfect depolymerization with each run, and clear cost savings.
Image Credit: Wyatt Technology

This information has been sourced, reviewed and adapted from materials provided by Wyatt Technology.
For more information on this source, please visit Wyatt Technology.