Tetracycline antibiotics (TCs) are among the most commonly utilized antibiotics in the animal production sector. They are employed to avoid infection in animals, treat diseases and safeguard animal growth.
Overusing antibiotics in animal-originated food products (for example, milk and meat) may lead to residue violations, resulting in risks to consumers’ health. Tetracycline residues present in food products may lead to a range of problems, including:
- Resistance to bacteria, which could transfer to humans
- Risk of toxicity
- Side-effects
- Allergic reactions
To reduce the risks and safeguard consumer health, regulatory authorities worldwide have set out guidelines for acceptable levels of veterinary drugs in food products. In the US, these levels are called ‘tolerances,’ while China, Canada and the European Union refer to them as ‘maximum residue levels’ (MRLs).1,2
Utilizing techniques that are highly sensitive and selective to provide accurate monitoring of tetracycline levels as specified regulatory requirements is a key element in protecting consumer health.
This article outlines a UHPLC/MS/MS method that offers the speed and sensitivity required for carrying out a quantitative analysis of tetracyclines in meat products.
To improve the testing lab’s efficiency, a QS-Works high-throughput autosampler was used for the following reasons:
- It offers large capacity
- It has temperature-controlled trays
- It utilizes robotic arms, allowing for long-term unattended usage
- It allows for as many as 600 samples to be analyzed in one sequence
The method outlined showed excellent sensitivity, recovery, precision, linearity and selectivity needed to analyze tetracyclines in meat products at the low tolerance levels set out by various regulatory authorities around the globe.
The Experiment
A range of hardware was utilized to carry out the analyses. Chromatographic separation was performed on a PerkinElmer QSight® LX50 UHPLC system. The detection process was completed using a PerkinElmer QSight® 420 MS/MS detector.
A QS Works autosampler with a high capacity was utilized to increase the lab’s productivity. The Simplicity 3Q™ software platform was employed to carry out data acquisition and processing and all instrument control.
LC Method and MS Source Conditions
The MS source parameters and LC method can be seen in Table 1. Multiple reaction monitoring mode (MRM) transitions of the tetracyclines are displayed in Table 2.
Table 1. LC Method and MS Source Parameters. Source: PerkinElmer Food Safety and Quality
| LC and MS Condition |
| Mobile phase A |
Water (contains 0.1% Oxalic acid) |
| Mobile phase B |
Acetonitrile (contains 0.1% formic acid) |
| Column |
PerkinElmer Quasar SPP Pesticides, 100 × 4.6 mm; 2.7 μm, (N9306880) mm |
| Column oven temperature |
20 °C |
| Autosampler temperature |
20 °C |
| Injection volume |
5 μL |
| ESI voltage (positive) |
5100 V |
| Drying gas |
120 |
| Nebulizer gas |
250 |
| Source temperature |
350 °C |
| HSID temperature |
250 °C |
Table 2. MRM Transitions. Source: PerkinElmer Food Safety and Quality
| Compound name |
Precursor ion/Da |
Product ion/Da |
CE/V |
EV/V |
CCL2/V |
| Oxytetracyline |
461.3 |
426.3
201.2 |
-21
-49 |
31
25 |
-136
-180 |
| Tetracycline |
445.3 |
410.3
427.3 |
-22
-15 |
29
27 |
-144
-124 |
| Chlortetracycline |
479.2 |
154.0
444.2 |
-34
-25 |
26
29 |
-136
-164 |
| Doxytetracycline |
445.3 |
154.1
154.0 |
-35
-34 |
29
26 |
-140
-136 |
A minimum of two MRM transitions were monitored for each analyte. This was to lower the number of false positives and negatives in the outlined method.
The optimizing of MS/MS parameters, which included the choice of parent ions and product ions, entrance voltages (EV/CCL2) and collision energies (CE) was done by infusion of standards.
The optimization of source conditions was achieved via t-infusion of neat standards with LC flow, with the acquisition MS method established based on those optimized conditions.
Standards and Sample Preparation
All reagents, solvents and diluents utilized were LC/MS grade. All tetracycline standards were sourced from Sigma-Aldrich® Inc. These were stored at - 20 °C in a freezer to prevent their degradation.
All stock and mixed drug solutions utilized in the spiking and calibration procedures for all the tetracyclines were prepared in methanol. To decrease the risks of degradation of standards, all working and stock standards were stored in a freezer until needed.
A homogenized sample for use in the procedure is created as follows:
- A representative sample of meat is cut into small pieces and then blended.
- A 1 g aliquot of the homogenized meat sample and 8 ml of 0.1 M aqueous EDTA solution are placed in a 50 ml centrifuge tube and vortexed.
- A 17 ml measure of acetonitrile is subsequently placed in the same tube and vortexed.
- The centrifuge tube is placed into the freezer, which is maintained at -20 °C for 30 minutes.
- The tube then undergoes centrifuging for 10 minutes at 4000 rpm.
- An 8 ml measure of supernatant is transferred into a 50 ml clean centrifuge tube.
- A 5 ml measure of hexane is added to de-fat the sample.
- The tube is vortexed for 3 minutes.
- The solution is then centrifuged for one minute at 4000 rpm.
- The 3 ml of lower phase is transferred into a new 15 mL centrifuge tube.
- It is then evaporated to dryness by blowing nitrogen over it at a temperature of 50 °C.
- The dried extract is reconstituted by dissolving it in 1 ml of methanol:water (30:70, v/v) solution, which contains 0.01 M oxalic acid.
- Sample extracts are vortexed and filtered by means of a 0.2 µm PTFE syringe filter prior to injection onto the LC/MS/MS system.3
Results and Discussion
This article aims to evaluate the performance of the high-capacity QS-Works autosampler with a validated sample preparation procedure for the determination of tetracyclines in a meat sample.
Utilizing a high throughput autosampler, for example, the QS-Works, increases testing lab productivity and efficiencies.
The QS-Works allows food testing labs with a large number of samples to process samples more rapidly while giving enhanced data quality and consistency due to its robotic sampling unit (Figure 1).

Figure 1. QS-Works autosampler. Image Credit: PerkinElmer Food Safety and Quality
Reference target levels were determined by utilizing the maximum residue levels (MRLs) established for tetracyclines in foods by Health Canada or tolerances by the US FDA (X in Table 3).
Table 3. Results of retention time, matrix effect (ME), linearity, recovery, and reproducibility (%RSD). Source: PerkinElmer Food Safety and Quality
| Compound name |
Level(ng/g) Tolerance (x) |
RT(min) |
ME(%) |
Linearity(R2) |
Recovery(%) |
RSD with oxalic acid (%)(n=5) |
RSD without oxalic acid (%)(n=5) |
| Oxytetracyline |
200 |
3.09 |
86 |
0.9984 |
70.0 |
4.6 |
11.0 |
| Tetracycline |
200 |
3.26 |
80 |
0.9997 |
116.0 |
4.9 |
16.0 |
| Chlortetracycline |
200 |
3.65 |
96 |
0.9991 |
87.3 |
1.9 |
9.5 |
| Doxytetracycline |
200 |
3.73 |
94 |
0.9980 |
71.6 |
1.5 |
8.0 |
The detection threshold, or ‘yes/no’ screening level, should be at or below 0.5X. The chromatograms (XIC) of four tetracyclines demonstrated good peak resolution (Figure 2).
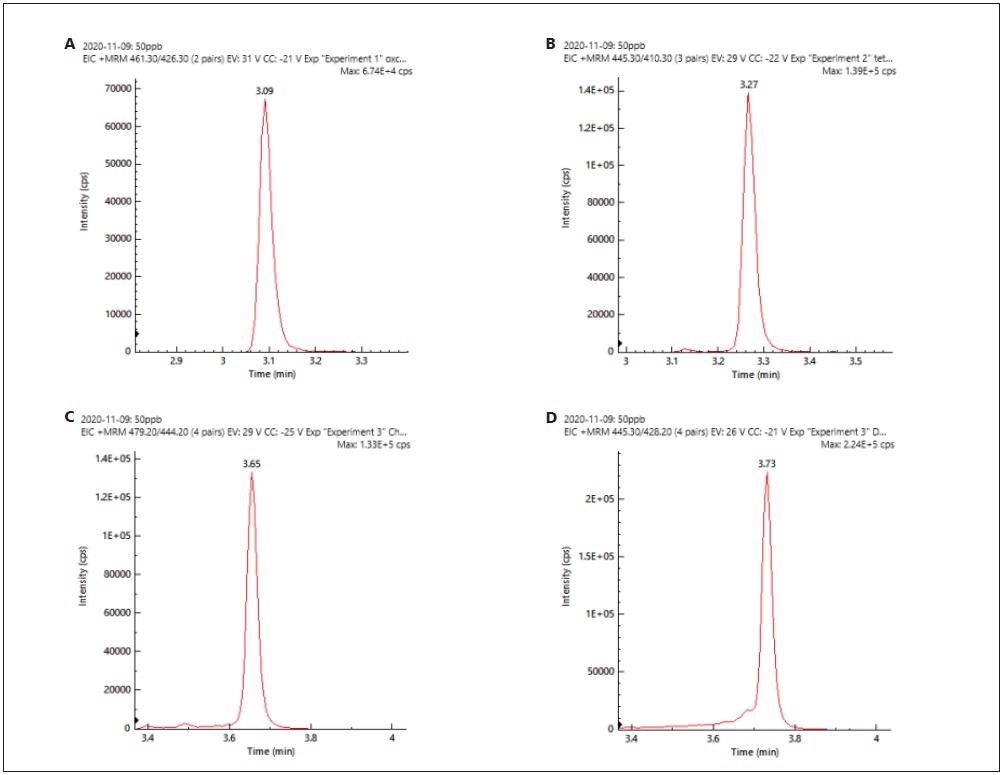
Figure 2. XIC chromatograms of a) oxytetracycline, b) tetracycline, c) chlortetracycline, d) doxytetracycline. Image Credit: PerkinElmer Food Safety and Quality
Two ion transitions were observed for confirmation and identification purposes, resulting in greater selectivity and specificity in identification. Every ion ratio was determined by calculating the peak area for less intense ion to peak area for more intense ion in order to generate ion ratios.
Among the main concerns for LC/MS/MS method development are sample matrix effects (MEs). This is especially the case when analyzing food due to the complex and diverse nature of food sample matrices.
ESI is vulnerable to ionization suppression of analytes where charge-competing matrix components are present. Matrix-induced enhancement effects can occur in ESI, which may lead to a large bias in quantification.
To surmount sample matrix effects, a number of approaches have been utilized, including:
- Standard addition
- Sample clean-up
- Utilizing isotope internal standards
- Sample dilution
- Matrix-matched (MM) calibration
- Using high efficiency columns for improved separation
Sample matrix effects were calculated by the comparison of the slopes of calibration curves acquired from the meat sample matrix to slopes acquired from reagent-only (RO).
The sample matrix effect (expressed as a percentage) for each analyte was then determined by the percentage difference between the slopes being compared.
The matrix effects for most of the compounds were less than 20% (Table 3).
Matrix-matched calibrations were utilized in this analysis for the quantification of all analytes. This was to control matrix effects and decrease the level of variations in analytical results.
Calibration was carried out utilizing both reagents-only (RO) and matrix-matched (MM) standards.
Every calibration curve that was constructed from both RO and meat sample matrix demonstrated good linearity with a correlation coefficient (R2) larger than 0.99. For typical examples of calibration curves, see Figures 3 and 4.
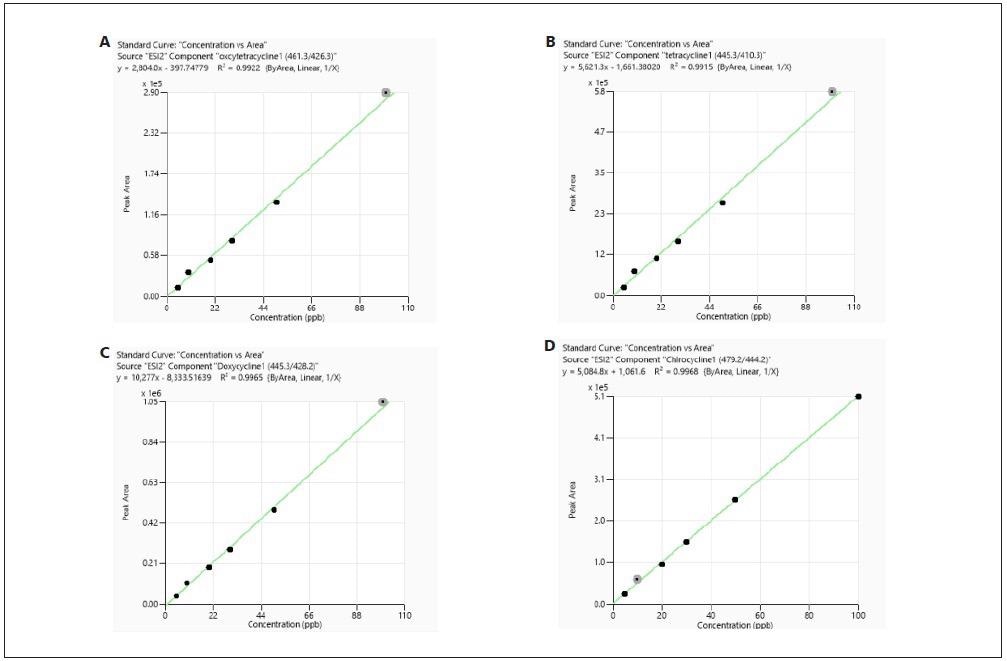
Figure 3. Calibration curves of tetracyclines obtained from standards prepared in reagent only. Image Credit: PerkinElmer Food Safety and Quality
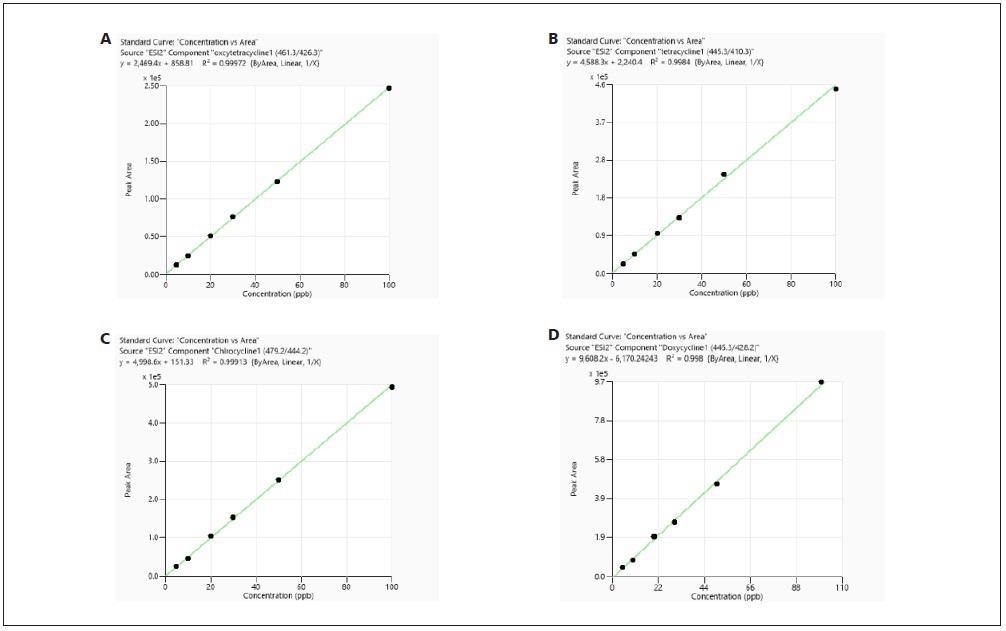
Figure 4. Calibration curves of tetracyclines obtained from standards prepared in meat sample matrix. Image Credit: PerkinElmer Food Safety and Quality
The degree of carryover was evaluated by injecting the reagent blank after a high concentration standard. No carryover was noted in any of the tests.
Oxalic acid was added into the aqueous mobile phase3 to prevent chelation of tetracyclines by metal ions of LC parts or column body. This also helped sustain good peak shape and reproducibility over the run.
The method displays good precision with RSD at less than 5% for five replicates. For the effect of using oxalic acid in mobile phase and reconstitute solvent on the reproducibility, see Table 3.
The reproducibility and peak shape of all analytes was improved by the addition of oxalic acid into mobile phase and reconstitute solvent. The limits of quantification (LOQ) for each tetracycline analyzed were lower than the permitted tolerance level in meat samples.
This indicates that the technique meets the levels of sensitivity needed to analyze tolerance levels for tetracycline in meat.
The LOQs for the tetracyclines were estimated based on signal to noise (S/N) ratio of 10. Every LOQ for the drugs studied was 1.5 ± 1.0 µg/L.
These results show this method can be employed for the rapid screening and quantification of tetracyclines in meat samples, while the absolute recoveries of tetracyclines were in the range of 70-120% (Table 3). Utilization of a deuterated tetracycline can compensate for losses of tetracyclines with this extraction process.
Conclusion
A PerkinElmer high throughput QS-Works autosampler and LX 50 UHPLC coupled to a QSight 420 MS/MS system were utilized to demonstrate a UHPLC/MS/MS procedure.
The instrument displays good sensitivity for identifying and quantifying tetracyclines in a homogenized meat sample. The low detection levels attained in the method can help support low regulatory restrictions for everyday screening and quantitation analysis.
References
- List of Maximum Residue Limits (MRLs) for Veterinary Drugs in Foods, Health Canada, August, 2017. https://www.canada.ca/en/health-canada/services/drugs-health-products/veterinary-drugs/maximum-residue-limits-mrls/list-maximum-residue-limits-mrls-veterinary-drugs-foods. Accessed November, 2020.
- Standard Method Performance Requirements (SMPRs®) for Screening and Identification Method for Regulated Veterinary Drug Residues in Food. SMPR2018_010.pdf (aoac.org). Accessed November, 2020.
- Aurélien Desmarchelier, Sébastien Anizan, Mai Minh Tien, Marie- Claude Savoy & Cindy Bion (2018) Determination of five tetracyclines and their epimers by LC-MS/ MS based on a liquid-liquid extraction with low temperature partitioning, Food Additives & Contaminants: Part A, 35:4, 687-695, DOI: 10.1080/19440049.2018.1427894
Acknowledgments
Produced from materials originally authored by Saba Hariri, Abir Khaled, Avinash Dalmia, and Feng Qin from PerkinElmer, Inc.

This information has been sourced, reviewed and adapted from materials provided by PerkinElmer Food Safety and Quality.
For more information on this source, please visit PerkinElmer Food Safety and Quality.