|
Well they mean different things to different people:
• To a chemist they are polymers which contain urethane groups (-NH-CO-O-) produced by the reaction of a polyol with an isocyanate, of which will be expanded upon later in this article.
• To an engineer they are materials which offer a combination of unique properties which allow products to meet a range of demanding applications.
• To an accountant they are materials which are very cost effective both to process and in use.
A PU may be either thermosetting or thermoplastic. These different characteristics, and their extraordinary physical properties, are explained by the different chemical structures which can exist in PUs.
What Forms can PU’s take?
PUs can be processed into elastomers, coatings, adhesives, binders, sealants and foams for a host of applications in, amongst others, the shoe, clothing, furniture, domestic appliance, building, marine, electrical engineering and transport industries. Indeed PUs can be formulated for a bewildering array of applications for an increasing number of industries.
PUs are perhaps the most versatile polymeric materials. Whilst their major use is still in foams; utilising their strength, insulation, resilience and comfort properties, PUs also fulfil the need when a component needs to exhibit both the strength of plastics and the flexibility of rubbers. They are employed in a diverse range of industries such as furniture, transport, construction, appliances and footwear to name just a few.
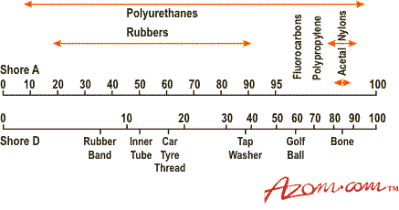
Since their discovery over 60 years ago PU chemistry has been developed and adapted to make the diverse range of products available today. The versatility of PUs means that they can be formulated into a very hard solid or a soft elastomer, or something in between, which allows the material to be custom tailored to its' end use. Figure I shows the hardness spread, i.e. from 10 Shore A (softer than an eraser) to 95 Shore D (harder than a golf ball or bone). The Shore A equates, for all practical purposes to IHRD (International Rubber Hardness Degrees), see Figure 1.
|
|
|
Figure 1. Hardness spread of polyurethanes.
|
Uniquely PUs can be tailored, by both chemistry and processing, to yield products in a wide variety of forms and which allow the processor to control the nature and the properties of the end product. For example thermoplastic polyurethanes (TPUs) are formulated for use in applications ranging through hose and cable sheathing, films and sheet, catheters, gears, bushes and bearings and such specialised areas as mining cables and optical fibres.
|