Appliance designers and manufacturers commonly rely on plastic parts as essential components. To ensure these parts meet high-quality standards, it’s important to take both functionality and the intended environmental conditions into account.

Figure 1. Thermo Scientific™ Nicolet™ Summit™ X FTIR Spectrometer with Thermo Scientific™ Everest™ Diamond ATR Accessory. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
Infrared spectroscopy (IR) has proven to be an incredibly useful analytical tool for engineers and scientists, due to its ability to characterize the molecular makeup of polymers.
IR can be applied across all phases of the product lifecycle, including design, manufacturing, and failure analysis during use.
This article aims to prove IR’s utility, both when used by itself and in conjunction with other methods, to assist in the design, manufacture, and support of products that use plastic components. It will focus on IR analysis of failed parts and describe other analytical techniques that can help pinpoint failure modes.
Identifying Failures and Ensuring Quality of Plastic Materials
Anyone familiar with polymer chemistry undoubtedly recognizes the value of IR spectroscopy for characterizing plastic materials. It can assess the mostly organic make-up of polymer molecules, rendering IR a vital tool for product development, quality control, and problem solving. Some key sectors in which infrared analysis has proven to be crucial tool include:
- Material identification and verification
- Copolymer and blend assessment
- Additive identification and quantification
- Contaminant identification, both in bulk and at surfaces
- Molecular degradation assessment
For the identification of unknown materials, an important component of failure analysis, the most common approach is performing a quick IR analysis followed by a simple search in which the sample spectrum is compared to a spectral database of plasticizers, polymers, or other compounds.
When used for the quality control of incoming materials, comparisons are made against the spectra of known, high-quality materials.
Case Studies
Incorrect Material: Plastic Fastener Cracking
Plastic fasteners are used to hold appliance assemblies together. Therefore, they must be strong - but not too brittle - to achieve the desired behavior over the product’s lifespan. In this case study, fasteners cracked over time while in service.
The observed failure features suggested slow crack growth (or creep rupture) under the load stress to which the parts were subjected. Further investigation determined that the failed parts appeared to be from a single production run.
The parts are injection molded from polypropylene copolymer resin. Known control parts were available for comparison from previous production runs. A quick infrared scan of both the failed parts and the control was conducted using the Everest Diamond ATR Accessory, yielding the top two spectra in Figure 1.
Comparison to the reference spectra of polypropylene and polyethylene confirms that both the failed and control parts are copolymers of these two materials.
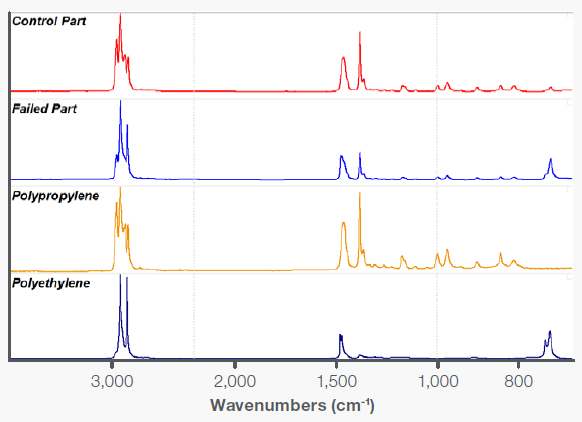
Figure 2. Spectra of a control and a failed plastic fastener, along with reference spectra of pure polyethylene and polypropylene. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
However, closer examination of the expanded hydrocarbon stretching region of both the failed and control parts reveals a more complete story (Figure 4).
In the failed part, there is a significantly higher ratio of polyethylene to polypropylene. This resulted in the piece being less stiff than needed, making it more prone to deformation and cracking under long-term stress.
This failure could have been prevented if quality control verification of the incoming polymer beads had been conducted prior to molding. FTIR with the Everest Diamond ATR Accessory provides a fast and simple method for copolymer blend ratio analysis on plastic parts and polymer beads.
The cost of failure, in terms of resolution and potential product recalls, could easily offset the investment in running this routine QC test.
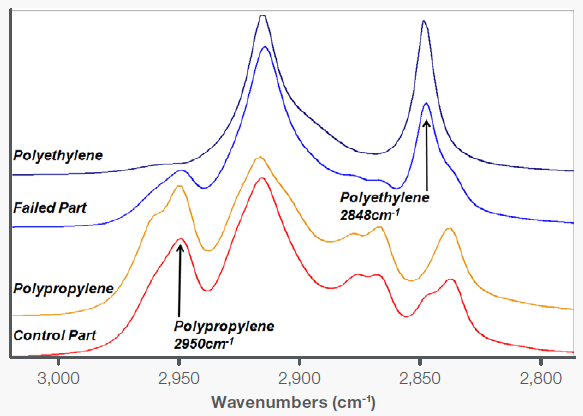
Figure 3. Expanded hydrocarbon stretching showing stronger polyethylene characteristics in the failed part. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
Useful information can be obtained about a sample using many sample analysis techniques. The easiest and most popular technique currently available in modern Fourier transform infrared (FTIR) spectrometers is attenuated total reflectance (ATR), using diamond crystals. The nature of the ATR technique ensures that spectral intensities remain within a linear range.
Sample analysis is often finished in less than one minute, making it an incredibly fast screening technique. What is more, the diamond ATR crystal cannot be scratched. The sample is positioned in close proximity to the diamond crystal, so solid samples must be pressed into the crystal surface using a pressure device (Figure 2).
It should be noted that ATR is a surface analysis technique that penetrates approximately 2-4 micrometers into the sample. Therefore, in many cases, the sample surface must be cleaned or excised if identification of the bulk plastic is desired.
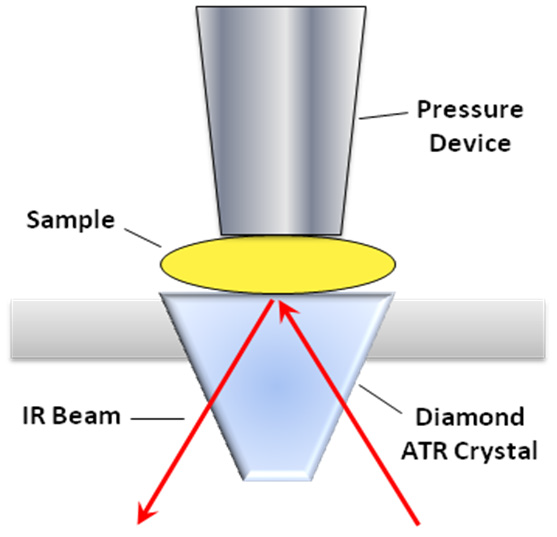
Figure 4. Schematic representation of a diamond ATR accessory. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
Inclusions: Valve Failure
Several valves had failed during installation with their mating components, so they were all evaluated. The failures took place within the retention arm, which functions as a snap during the installation of the valve assembly. The valves are made from a lubricated, unfilled, injection-molding grade of poly(butylene terephthalate) (PBT).
Microscopic examination of the fracture surfaces on the valves generally demonstrated similar features, typical of brittle fractures, across all the failed parts. The failures exhibited crack initiation at an inclusion present within the molded retention arm, as illustrated in Figure 5.

Figure 5. The fracture surface is shown with crack initiation at an inclusion. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
The valves were first analyzed using energy dispersive X-ray spectroscopy (EDS). Table 1 shows these results. Analysis of the base fracture surface exclusively showed carbon and oxygen, which aligns with expectations of an unfilled PBT resin. Analysis of the included particle, meanwhile, showed substantial amounts of sulfur and trace levels of sodium, potassium, and chlorine.
Table 1. EDS analysis results. Source: Thermo Fisher Scientific - Vibrational Spectroscopy
| Element |
Base material (relative weight %) |
Inclusion (relative weight %) |
| Carbon |
66.5 |
73.9 |
| Oxygen |
33.5 |
15.4 |
| Nitrogen |
--- |
4.8 |
| Chlorine |
--- |
0.2 |
| Potassium |
--- |
0.1 |
| Sodium |
--- |
0.1 |
| Sulfur |
--- |
5.5 |
The valve samples were analyzed directly, employing the Everest ATR Accessory to perform FTIR. A base-material sample was analyzed, producing a spectrum that was characteristic of a thermoplastic polyester like PBT. The fracture surface inclusion was excised and then analyzed via FTIR.
These results were directly compared with those obtained on the base material, showing the presence of additional absorption bands (Figure 6). A spectral subtraction was performed to remove the bands associated with the PBT base material. The resulting spectrum exhibited absorption bands attributed to poly(phenylene sulfide) (PPS), as shown in Figure 7.
PPS’s melting temperature is roughly 285 °C, and as such, is consistent with its presence as a non-dispersed particle within the significantly lower melting PBT resin. The included contaminant’s presence acted as a source of strong stress concentration by focusing and multiplying the stress applied through the snap installation.
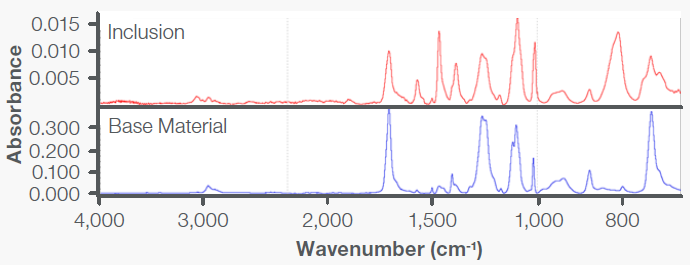
Figure 6. FTIR spectrum showing that the inclusion in the valve sample contained additional absorption bands in comparison with the base material. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
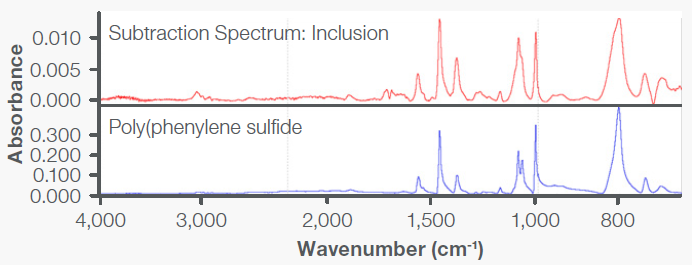
Figure 7. Spectral subtraction of the inclusion minus base material resulted in a spectrum characteristic of PPS. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
Environmental Stress Cracking: Brackets
A failure analysis was conducted on a set of brackets that had cracked while in service. Sporadic failures had been reported on these parts. The brackets were injection molded from a flame-retardant grade of polycarbonate + poly(acrylonitrile butadiene styrene) (PC+ABS).
The brackets were microscopically examined, which identified catastrophic transverse cracking at the base of the cylindrical boss, congruent with bending or tensile stresses. Upon further inspection of the fracture surfaces, the characteristics of a brittle fracture were evident, with multiple apparent crack origins along the inner diameter of the boss wall (Figure 8).
The perceived features were suggestive of a slow crack initiation and growth mechanism, which was transitioning into more rapid crack extension. Ultimately, the observed features were indicative of environmental stress cracking (ESC), a failure mechanism in which a plastic material cracks due to contact with an incompatible chemical agent under tensile stress. Multiple cracks had a liquid residue leaching from the fractures.

Figure 8. The failed bracket fracture surfaces exhibited features that are characteristic of environmental stress cracking (ESC). Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
Specimens that represented the bracket material were analyzed using FTIR with the Everest ATR Accessory. This analysis of bracket core specimens demonstrated absorption bands characteristic of a PC+ABS resin, as seen in Figure 9. Residue was removed from the fracture surfaces, which were analyzed and seen to be indicative of organic esters, which are known to act as ESC agents in combination with PC+ABS resins. This can be seen in Figure 10.
Melt flow rate testing was used as well, which determined that the part material had undergone significant molecular degradation during the injection molding process. This degradation made the parts susceptible to environmental stress cracking and early failure.
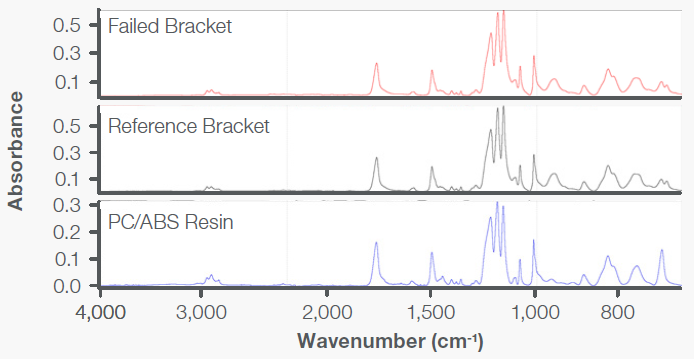
Figure 9. The FTIR spectra representing the failed and reference brackets contained absorption bands indicative of a PC+ABS resin. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
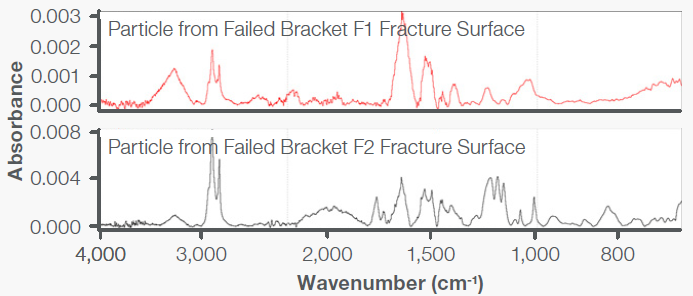
Figure 10. FTIR spectra obtained on the bracket fracture surfaces contained absorbances associated with organic esters. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
Degradation: Water Tubing Leakage
A section of plastic tubing was analyzed, as it had leaked while being used. This tubing had been used to supply water to a refrigerator, and the leak resulted in substantial damage to the property. This type of tubing is often extruded from a type of low-density polyethylene (LDPE).
The tubing was examined and found to have a single through-crack and multiple partial cracks that were transversely oriented. The cracks exhibited features that are characteristic of brittle fracture, indicating crack initiation along the inner diameter wall of the tubing. This is shown in Figure 11.
There was also some discoloration present, all in the area of cracking. These observations suggested a slow crack growth mechanism associated with localized bending loads and the internal pressure within the system.

Figure 11. Broken tubing, showing a series of parallel transverse cracks with brittle fracture features. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
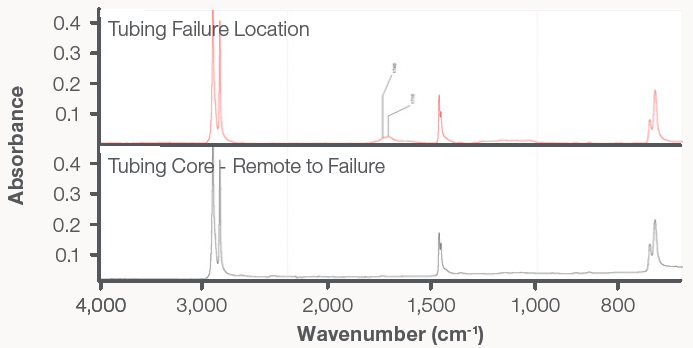
Figure 12. The FTIR spectrum of the failure location in the broken tubing exhibited carbonyl bands associated with the degradation of polyethylene resins. Image Credit: Thermo Fisher Scientific - Vibrational Spectroscopy
An area of the tubing further away from the crack was analyzed using FTIR with the Everest ATR Accessory. The gained results suggested polyethylene resin. Analysis of the inner diameter surface that was adjacent to the primary throughcrack also demonstrated characteristics of polyethylene. However, extra absorption bands were also present, which were indicative of carbonyl functionality.
Specifically distinct bands were apparent at 1715 cm-1 and 1740 cm-1, which are associated with esters and carboxylic acids (Figure 12). These materials, and similar ones, are often formed as byproducts of the polyolefin resin oxidation process: one example is polyethylene. Polyethylene can undergo oxidation when it is exposed to chlorinated municipal water. Oxidation causes molecular degradation, which results in a significant reduction in the tubing material’s mechanical integrity. Alongside the nominal internal pressure, this produced cracking within the tubing.
Conclusion
Infrared spectroscopy is a valuable tool for polymer characterization, which has effective uses in manufacturing, product design, and, as this article has proven, failure analysis. Thermo Scientific Summit FTIR Spectrometers can provide rapid analysis of samples with very little effort, equipped with diamond ATR accessories. The provided case studies can be used to influence future decisions concerning product improvements and determine the best control measures to ensure quality.
Acknowledgments
Created using materials originally produced by Jeffrey Janse from The Madison Group and Mike Garry from Thermo Fisher Scientific.

This information has been sourced, reviewed, and adapted from materials provided by Thermo Fisher Scientific - Vibrational Spectroscopy.
For more information on this source, please visit Thermo Fisher Scientific - Vibrational Spectroscopy.