Achieving accurate results with digital density meters requires more than high resolution - it depends on careful control of key variables and consistent handling throughout the workflow. This article highlights the critical factors influencing measurement accuracy, along with practical guidance to improve repeatability and minimize errors in routine density analysis.
Ensuring Accuracy and Repeatability When Using Density Meters
Temperature, viscosity, air pressure as well as sampling and cleaning, are the key factors influencing measurement accuracy.
Temperature
Density is largely temperature-dependent. The density of a sample is defined as its mass divided by its volume. When the temperature of a sample rises, its volume increases, and so its density decreases.
A temperature change of 0.01 °C can alter a sample's measured density by 0.00011 g/cm3. Temperature must be accurately controlled and measured (up to ±0.01 °C using a six-place density meter).
Viscosity and Homogeneity
The accuracy of measurements is also influenced by sample viscosity and homogeneity. Using an oscillating U-tube approach, higher viscosities will yield measurements slightly greater than the true density.
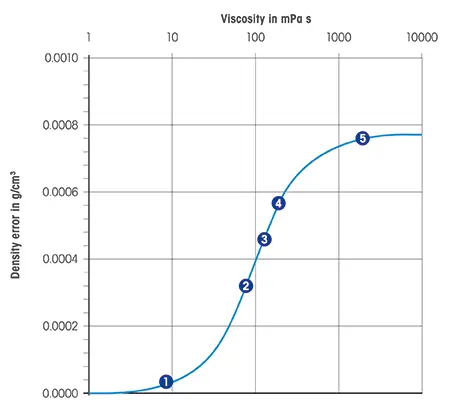
Image Credit: Mettler-Toledo International Inc.
The above chart data indicates that higher viscosity levels can lead to greater deviations in density measurements. In other words, as a liquid becomes more resistant to flow, the likelihood of measurement error increases.
That said, modern digital density meters incorporate automatic viscosity compensation, allowing for precise results while maintaining rapid analysis times.
As a result, most benchtop digital density meters, including METTLER TOLEDO density meters, feature automated viscosity adjustment. Sample homogeneity also influences measurement accuracy. To guarantee sample homogeneity, stir the mixture manually or automatically.
Air Pressure
Air pressure affects the nominal density value of air, which is used as a reference point in standard adjustments with air and water. Modern digital density meters include a built-in pressure sensor that is employed during the adjustment phase to ensure optimal performance.
Sampling and Measurement
When manually loading a cell with a syringe, fill it gently and evenly to avoid air bubbles or a thin air film between the sample and the measuring cell, which must be completely filled. Do not remove the syringe until the measurement is complete.

Image Credit: Mettler-Toledo International Inc.
Using a peristaltic pump or a sample automation unit will result in a controlled sampling speed and complete fill. When the sample comes out a few centimeters from the draining tube, the measuring cell is full.
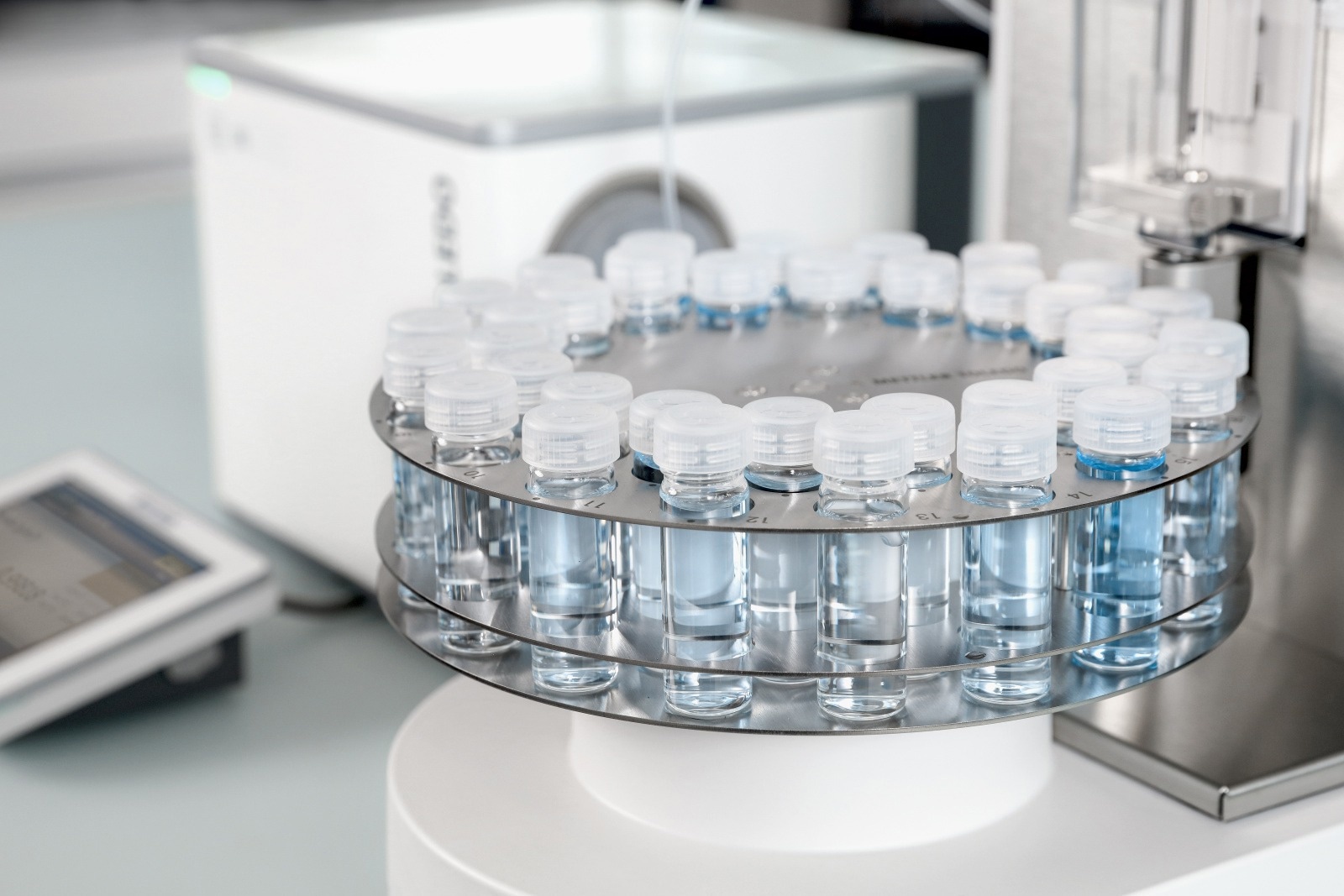
Image Credit: Mettler-Toledo International Inc.
For measurement, the U-tube containing the sample is heated to the desired temperature, the device waits until the oscillation frequency stabilizes, and the frequency is converted to density or specific gravity. Built-in conversion tables display results in various units, such as propylene glycol concentration. This avoids the potential of errors during manual conversion.
Specific limits for each sample should be established for quality control, and measurement results should be automatically compared against these limits to display pass/fail information.This eliminates the need to look up tolerances in a product table, saving time and reducing human error.
Cleaning and Drying
Because the majority of inaccurate readings are caused by insufficient or incorrect cell cleaning, this phase is perhaps the most critical in the measurement process. After completing a measurement, the sample must be withdrawn from the cell and deposited in a waste container or returned to the vial if an automation unit is used.
An appropriate cleaning solution is also required for thorough cleaning and accurate measurement.
Table 1. Recommended cleaning solutions by product type for density meter cells. Source: Mettler-Toledo International Inc.
|
Sample
|
Solvent 1
|
Solvent 2
|
|
Water, water-based
|
Deionized water
|
Acetone, ethanol (100 %)
|
|
Acids (concentrated)
|
Water (flush the measuring cell with plenty of water to remove the heat from the reaction of water and acid)
|
Acetone, ethanol (100 %)
|
|
Alkaline solutions (concentrated)
|
0.3...0.5 % deconex solution
|
Acetone, ethanol (100 %)
|
|
Samples with fats or oily components
|
0.3...0.5 % deconex solution
|
Acetone, ethanol (100 %)
|
|
Petrochemical samples, edible oils, and fats
|
Toluene, xylene, or petrol ether mixtures
|
Room temperature: low-boiling petrol ether mixture or acetone. Temperature > 30 °C: hexane or similar organic solvents
|
The final step is to dry the measurement cell using an air pump that draws ambient air through a bed of silica gel drying agent. After allowing an adequate drying period, verify that the cell is thoroughly dried by measuring the air density and comparing it to the known reference value. With METTLER TOLEDO density meters, you can conveniently perform this verification by simply executing a 'Cell Test' with a single click™.

This information has been sourced, reviewed, and adapted from materials provided by Mettler-Toledo International Inc.
For more information on this source, please visit Mettler-Toledo International Inc.