Ensuring reliable density measurements requires a structured approach that extends beyond individual tests. This section introduces Good Density and Refractometry Practice (GDRP™), a lifecycle-based framework designed to support compliance, optimize performance, and maintain long-term measurement confidence across laboratory workflows.
Good Density and Refractometry Practice (GDRP™) is a five-step framework for managing density and refractive index systems across their full working life, from instrument selection to day-to-day use. The aim is straightforward: more dependable results, clearer compliance, and fewer avoidable costs and disruptions.
GDRP™: Five Steps to Excellence
Accurate measurements are rarely “fixed” at the point of analysis. They come from choosing the right system, installing it correctly, and making sure the people using it know exactly what to do. GDRP puts that lifecycle thinking into a practical structure, supporting:
-
regulatory compliance
-
operator training and qualification
-
higher productivity and lower operating costs
-
sustained accuracy and precision over time
GDRP also sits comfortably alongside established quality frameworks such as Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP). Within METTLER TOLEDO’s broader Good Measuring Practices approach, it turns high-level expectations into instrument-focused guidance that teams can apply on the bench.
A Lifecycle Approach to Compliance and Performance
In pharmaceutical R&D and manufacturing, instruments must deliver consistent results without slipping outside internal quality systems or external regulatory requirements. That balance is easiest to maintain when decisions are made with risk in mind, starting early and continuing throughout the instrument’s life.
METTLER TOLEDO’s Good Measuring Practices framework reflects this through four qualification stages:
-
Design Qualification (DQ): confirming the system meets the intended requirements
-
Installation Qualification (IQ): verifying correct installation
-
Operational Qualification (OQ): confirming the system operates as expected
-
Performance Qualification (PQ): demonstrating consistent performance over time
These stages help teams define appropriate tests, documentation, and maintenance plans
based on what the application actually demands. GDRP builds on the same logic, but presents it as a clear, five-step workflow.
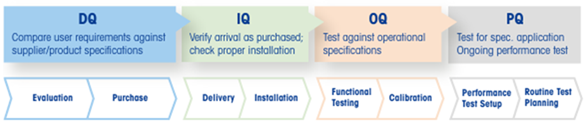
Figure 1. Good Measuring Practices. Image Credit: Mettler-Toledo International Inc.
The Five Steps of GDRP™

Figure 2. Good Measuring Practices outlines the five steps in the lifecycle of an instrument. Image Credit: Mettler-Toledo International Inc.
Step 1: Evaluation
Start by pinning down the measurement needs, regulatory expectations, and practical constraints (throughput, sample types, data handling, and so on). This is also the point where it pays to think ahead, not just about today’s method but about likely future requirements.
Step 2: Selection

Image Credit: Mettler-Toledo International Inc.
With requirements defined, choose the instrument and the supporting pieces that make it usable in a regulated workflow, including software, automation options, and data management tools where relevant.
Step 3: Installation

Image Credit: Mettler-Toledo International Inc.
Performance depends heavily on setup. Environmental conditions such as temperature stability, vibration, and drafts matter for precision instruments, as do correct connections to peripherals and data systems. Getting the installation right reduces troubleshooting later and helps protect data quality from the start.
Step 4: Qualification
After installation, qualification confirms the system meets defined specifications and any relevant regulatory expectations. Documentation is part of the deliverable here, but so is practical operator training so the system is used correctly and consistently.
Step 5: Routine Operation

Image Credit: Mettler-Toledo International Inc.
Long-term reliability comes down to habits and upkeep: regular calibration, planned maintenance, and periodic refreshers for users. These steps reduce measurement drift, prevent deviations, and keep the system performing as intended.
Ensuring Long-Term Confidence in Measurement
GDRP brings lifecycle management and risk-based thinking into a format that is easy to follow in the lab. For teams working with density and refractometry, it provides a structured way to protect instrument performance and data integrity while staying aligned with pharmaceutical quality requirements.

This information has been sourced, reviewed, and adapted from materials provided by Mettler-Toledo International Inc.
For more information on this source, please visit Mettler-Toledo International Inc.