Sponsored by InterpowerReviewed by Ify IsiborApr 17 2026
Global exports of medical power cords must meet a range of national and international requirements. This article explains key guidance, with sections organised by country and region.
North America
Hospital-grade cords and cord sets destined for use in North America must meet UL 817 standards and CSA C22.1 no. 21. These standards apply to the NEMA 5-15, 5-20, 6-15, and 6-20 cord sets.
Guidance states that “The blades on the plug shall be of solid brass material and the grounding pin cannot be easily bent or removed,” and it is important both the plug and connector are a straight or right-angle type, and these are molded onto a flexible cord.
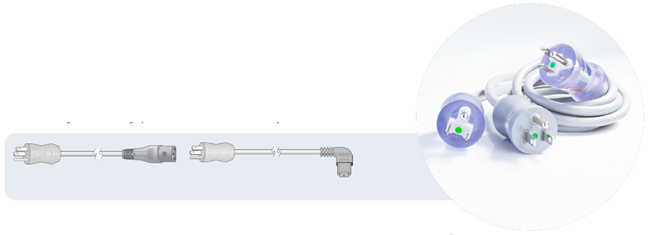
Image Credit: Interpower

NEMA 5-15. Image Credit: Interpower

NEMA 5-20. Image Credit: Interpower

NEMA 6-15. Image Credit: Interpower

NEMA 6-20. Image Credit: Interpower
Image Credit: Interpower
Testing
All North American hospital-grade cords are subjected to the UL 817 Abrupt Removal Test. This test involves plugging hospital-grade plugs into a socket before attaching them to a 10-lb weight.
The weight is then abruptly dropped to determine how far the blades have bent and to see if this test pulls the plug wires away from their connections.
Other standard tests outlined in the UL 817 guidance include:
- Abrupt Pull Test
- Rotary Pull Test
- Insulation Resistance Test
- Cord Jacket Retention Test
- Conductor Secureness Test
- Security of ‘Blades and Pins’ Test
North American hospital-grade cords are marked with a green dot indicating they are hospital-grade.
Design

Image Credit: Interpower
Interpower’s range of hospital-grade plugs features larger plugs than standard cords, helping protect internal components from abrupt disconnects. A retaining ring is used to clamp the ground, line, and neutral wires firmly in place.

Left to Right: Standard NEMA 5-15, Hospital-Grade NEMA 5-15. Image Credit: Interpower
These plugs can also be molded or hand-wired and are available in standard gray, black, or clear. Customized colors are also available where required for most specialist applications.
Australia
Medical equipment destined for use in Australia is subject to the Australian/New Zealand Standards (AS/NZS 3200 and 3112). No special requirements are stipulated for plugs used in a medical setting, but all Australian plugs must be approved in line with the AS/NZS standard.
Some hospitals in Australia prefer to utilize a clear plug with an orange, flexible cable.

Image Credit: Interpower
When replacing an original plug, however, it is important that the new plug be a clear rewirable plug, which complies with AS 3112 for three-pin plugs. This guidance states that “the plug shall be clear-backed to facilitate inspection of the core colors and the condition of the terminations.”

Image Credit: Interpower
This approach allows staff or technicians to visibly verify whether any conductors or other components have been pulled loose.
Denmark
Hospital-grade cords, plugs, and sockets used in medical applications in Denmark are subject to the Danish Safety Technology Authority (SB 107-2-D1) requirements.
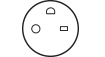
Medical Socket. Image Credit: Interpower
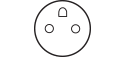
Standard Socket. Image Credit: Interpower
These requirements stipulate that the socket is designed to prevent “normal equipment” from being connected, because this may disrupt the main circuit in specific medical settings.
Red is the preferred color for hospital-grade cord sets in Denmark, but this is not a specific requirement of the Danish standard.
Design

Image Credit: Interpower
Interpower introduced red hospital-grade cord sets for use in Denmark in February 2017. The standard assembly of these cord sets features:
- A Danish hospital-grade plug
- A straight IEC 60320 C13 connector
- H05VV-F 1.00 mm2 cable
Power cords and cable sizes can also be provided in 0.75 mm2 and 1.50 mm2 versions. The available 10 A, 13 A, and 16 A 250 V cord sets are provided at 8.2 feet long unless the customer requests a custom length.
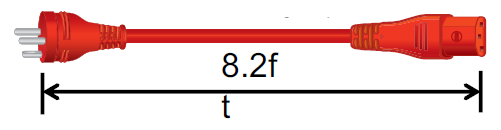
Image Credit: Interpower
It is also important to note that some part numbers contain a CE marking, meaning they are also marketable in Europe.
Japan
Japanese hospital-grade plugs and outlet sockets must be approved in line with the Japanese Industrial Standards Committee’s JIS T 1021 standard.
Japanese hospital-grade plugs may feature a green dot, with each plug indelibly marked with the letter H.

Image Credit: Interpower
Cords, plugs, and sockets used in Japan must carry the PSE approval mark under DENAN, a national law of Japan’s Ministry of Economy, Trade and Industry (METI).
The PSE mark is the minimum legal requirement for plugs, meaning that any plugs marketed as hospital grade must undergo in-house tests defined by JIS T 1021 and be self-declared as conforming to those requirements.
A JIS mark can also be awarded by a Japanese testing laboratory accredited by METI when products and the factory comply with JIS requirements. This service is not offered by UL.
Power Mains
Japan’s plugs are not compatible with a 250 V power mains, unlike their North American counterparts, NEMA’s 5-15, 6-15, and 6-20 hospital-grade plugs.
Mains power is 125 V in America and 100 V in Japan, and while this voltage difference can be managed with a step-down converter, there is also an Alternate Current (AC) cycle difference.
In America, the AC cycle is 60 Hz, while in Japan it is either 50 Hz or 60 Hz, depending on the region. Plugging a 50 Hz device with a heating element or motor into a 60 Hz main could cause the motor to burn out, become a fire hazard, or cause the user to receive an electrical shock.
The Rest of the World
EN 60601 (European Norms) Medical Equipment Standards for European Countries
EN 60601 is a medical equipment standard developed by the Committee for Electrotechnical Standardization (CENELEC), an organization with 34 member countries.
The EN designation is a codified standard in each of these countries, with an EN designation carrying with it “the obligation to be implemented at a national level by being given the status of a national standard and by withdrawing any conflicting national standard.”
The table below lists all 34 members of CENELEC.
Source: Interpower
| Members of CENELEC (Committee for Electrotechnical Standardization) |
1. Austria
2. Belgium
3. Bulgaria
4. Croatia
5. Cyprus
6. Czech Republic
7. Denmark
8. Estonia
9. Finland
10. France
11. Germany
12. Greece |
13. Hungary
14. Iceland
15. Ireland
16. Italy
17. Latvia
18. Lithuania
19. Luxembourg
20. Malta
21. Netherlands
22. North Macedonia
23. Norway
24. Poland |
25. Portugal
26. Romania
27. Serbia
28. Spain
29. Slovakia
30. Slovenia
31. Sweden
32. Switzerland
33. Turkey
34. United Kingdom |
India and many other non-European countries outside of China, Korea, and Japan use the IEC 60601 standards, which incorporate over 100 medical equipment standards.
IEC 60601-1 is a general standard, while the standards from IEC 60601-2 through IEC 60601-118 focus on specific pieces of medical equipment.
Other Countries
Countries that do not use testing agency standards, such as UL or CSA, for hospital-grade cord sets and components typically differ in their approach. For example, Germany, Spain, Italy, Switzerland, Israel, and many other countries use existing household or industrial cords and cord sets in medical facilities.
Common Problems
The most frequent problem encountered with medical power cords worldwide is the risk of urgent or abrupt disconnects, where plugs are rapidly pulled out of the wall socket, damaging or bending plug blades.

Image Credit: Interpower
Straightening bent plug blades can result in further damage, such as loosening blades or ground pins, or cracking brass blades. This can lead to arcing, which can cause a fire.
It is important to properly connect or disconnect the plugs by grasping the plug body, not the cord.
Sourcing Power Cords and Supplies from Interpower
Sourcing reliable, affordable products from Interpower lets customers lock in a specific quantity price break with a blanket or scheduled order.
Blanket Orders
Interpower customers can order a specified quantity (e.g., 1,000 cord sets), lock in a price, and request shipments as needed over a 1-year period. This allows the company’s needs to dictate product quantities and delivery dates.
Scheduled Orders
It is also possible to place orders with pre-determined quantities and delivery dates over a 1-year period. For instance, a customer may opt to place an order for 1,000 cord sets, with 250 delivered on January 15, April 15, July 15, and October 15.
No Minimum Order Requirements
Interpower’s same-day shipment service on in-stock products also features no minimum order requirements, meaning that every part the company manufactures in Iowa, USA, or sells can be purchased individually.
The company’s comprehensive product line of power system components is complemented by an extensive inventory, meaning that any part ordered can often be supplemented by another part from inventory or manufactured on demand.
Acknowledgments
Produced from materials originally authored by Interpower.

This information has been sourced, reviewed, and adapted from materials provided by Interpower.
For more information on this source, please visit Interpower.