Per- and polyfluoroalkyl substances (PFAS) are integral to multiple aspects of contemporary life and find extensive use across a variety of sectors.1 An area of significant PFA usage is semiconductor manufacturing, where PFAS are instrumental in producing components essential for fabrication processes.
Given the escalating concerns and regulations surrounding PFAS emissions as well as their environmental and health ramifications, the routine monitoring of PFAS sources is anticipated to gain increasing importance as efforts aim to reduce the global PFAS burden.2
Experimental Setup
Figure 1 illustrates the experimental apparatus designed for the thermal release of PFAS from PFAS tubing, a material commonly used due to its distinctive chemical attributes. Approximately 5 g of PFAS tubing was positioned within a heated stainless-steel oven, continuously purged with UHP nitrogen gas at a flow rate of two liters per minute.
The oven was directly coupled to the input of a chemical ionization time-of-flight mass spectrometer (CI-TOFMS). The oven’s temperature was gradually increased from 100 to 330 °C at approximately 2 °C per minute.
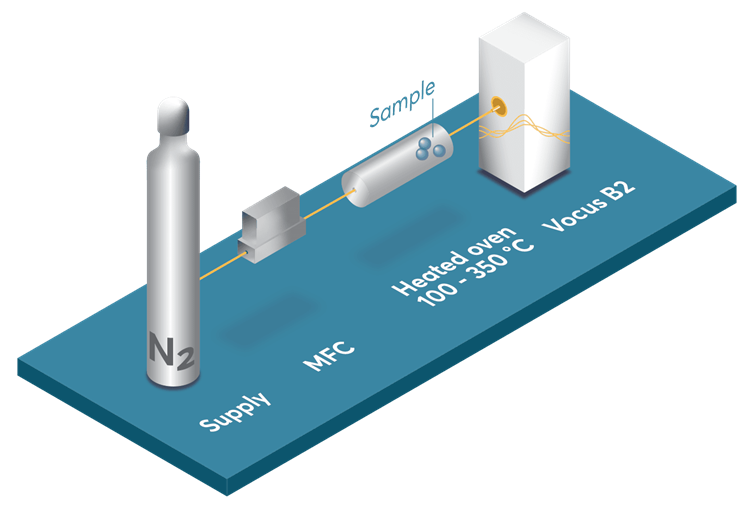
Figure 1. Experimental setup used in this study. Image Credit: TOFWERK
The resulting emissions were detected using the rapid polarity switching capabilities of the TOFWERK Aim Reactor, linked to the Vocus B2, to capture emissions through online measurement of both positive and negative analyte ions.
Results
All pertinent PFAS compounds were identified as the parent molecule clustered with iodide reagent ions or as their deprotonated anion.3 Compounds were characterized based on their chemical formula through precise measurement of the mass-to-charge ratio and isotopic patterns, facilitated by fragmentation-free ionization.
The top of Figure 2 presents a time series of representative PFAS compounds, such as PFBA, PFHxA, and PFOA (indicated by the blue trace), where the observed signal commenced increasing at 150 °C and continued to rise until approximately 250 °C. Beyond this temperature, the signal diminished, suggesting that decomposition mechanisms were beginning to play a crucial role.
As the emitted PFAS compounds degraded, the emergence of various known fluorinated breakdown products was observed, including hydrofluoric acid and TFA (red trace), and non-fluorinated compounds such as fulminic acid (HCNO), as depicted in black (Figure 2). Similar findings have been documented in previous literature.4
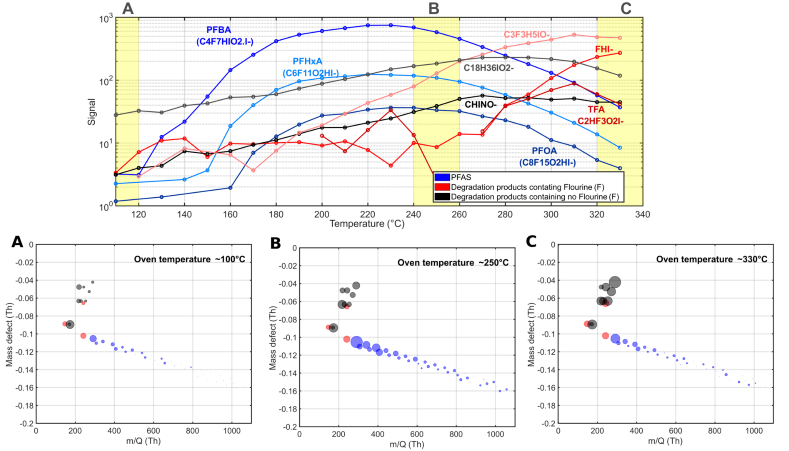
Figure 2. Time series of some compounds emitted from the material as a function of oven temperature (top) and mass defect plots showing total emissions at three temperature points: (A) 100 °C, (B) 250 °C, and (C) 330 °C. Image Credit: TOFWERK
The mass defect plot at the bottom of Figure 2 displays the total material emissions at three distinct temperature intervals. At approximately 100 °C, marking the initiation of the heating process, minimal PFAS emissions were detected from the sample. At around 250 °C, elevated PFAS emissions were observed, encompassing long-chain perfluoroalkyl carboxylic acids (PFCAs) with carbon chain lengths ranging from nine to 18.
As temperatures exceeded 250 °C, a variety of fluorinated and non-fluorinated compounds were detected at increased levels. These non-fluorinated compounds are indicative of various plasticizer products. Volatile hydrocarbon emissions, as measured by benzene cations, remained low across all investigated temperatures.
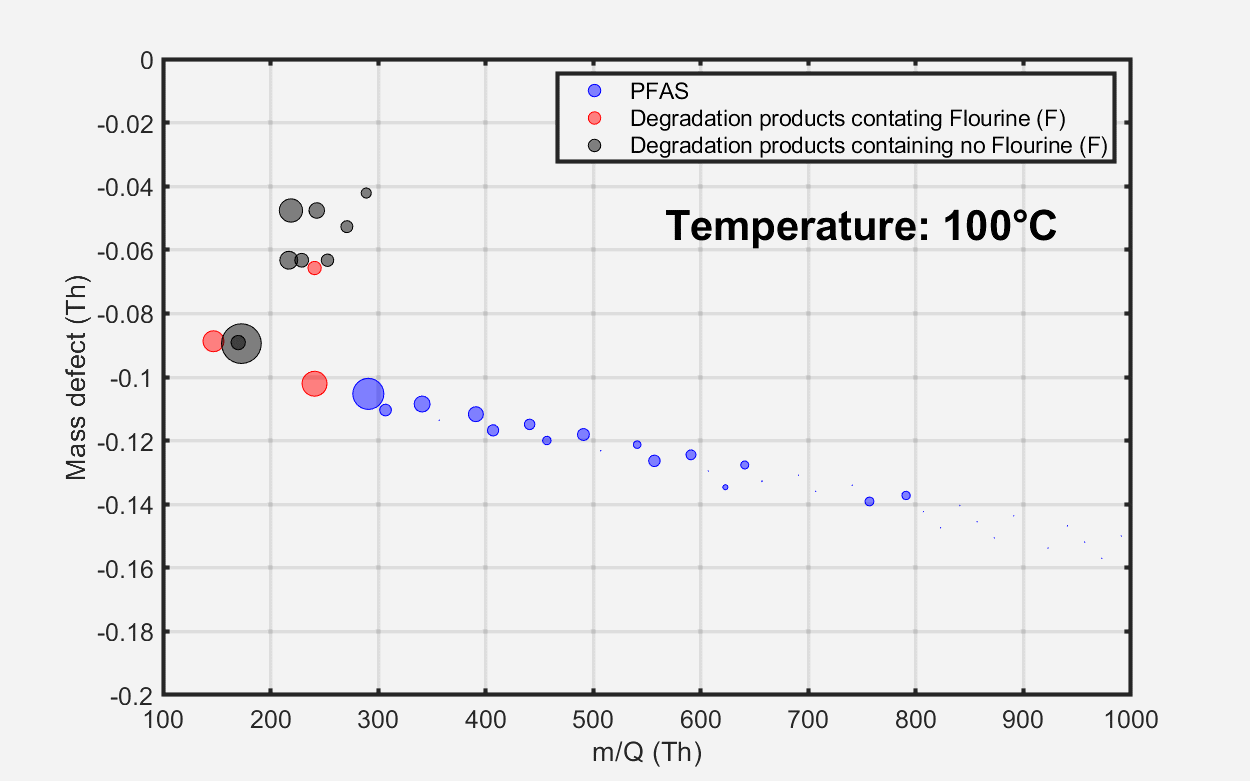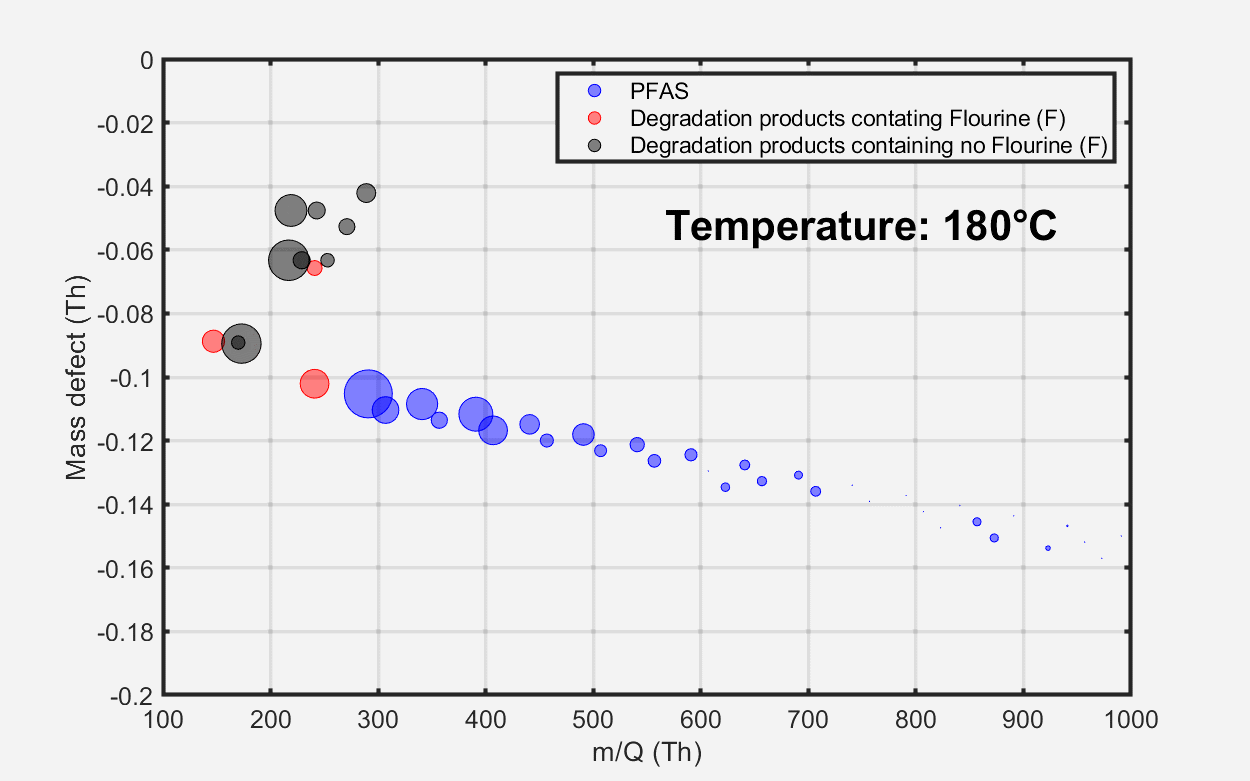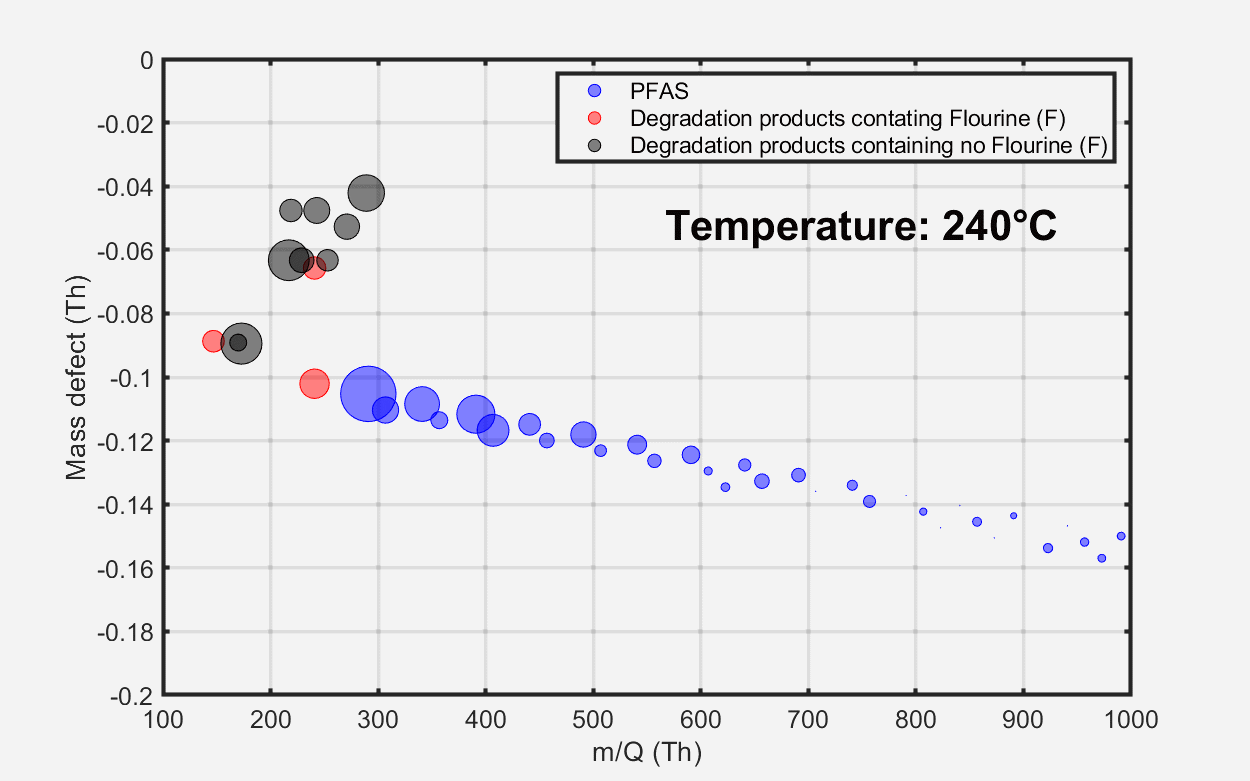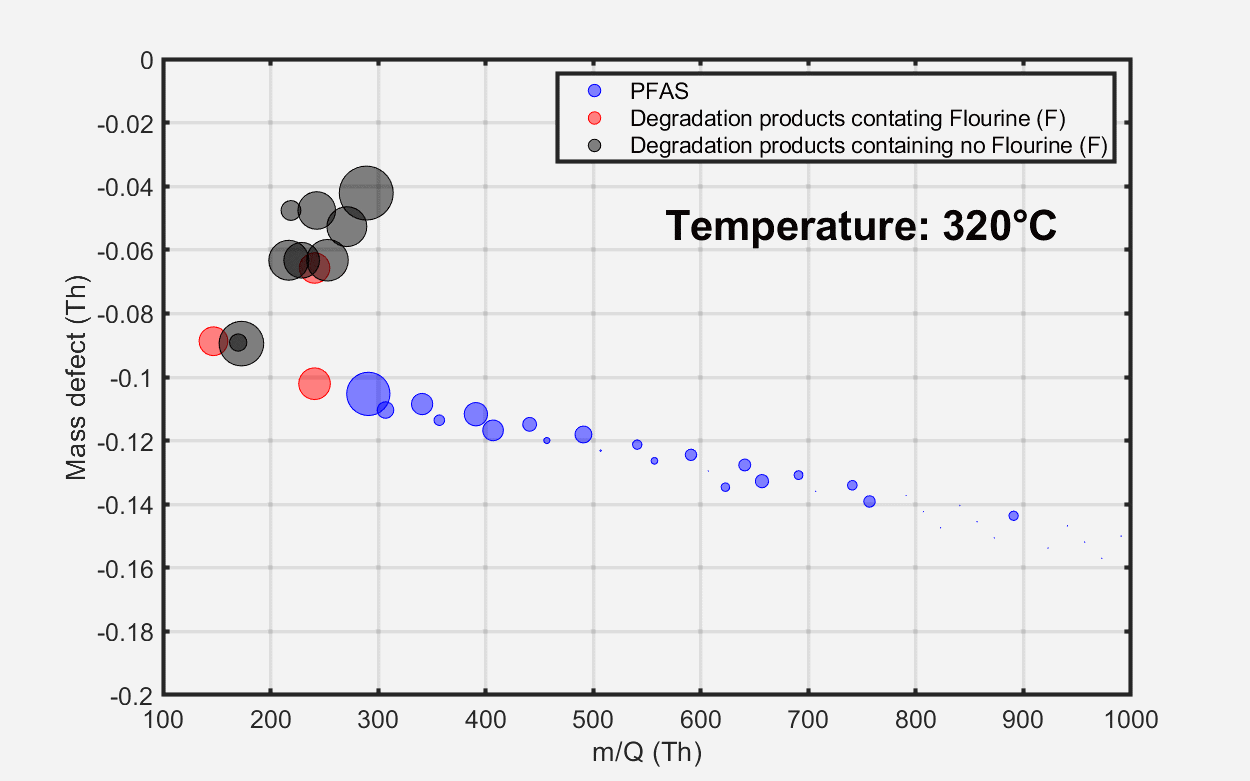
Figure 3. Evolution of material emissions depicted as a function of temperature. Image Credit: TOFWERK
Conclusion
The Vocus B Series, using iodide reagent ions, facilitates real-time PFAS tubing emissions monitoring in the gas phase. While the hydrocarbon emissions from PFA are minimal, the comprehensive capabilities of the Vocus B series are likely crucial for other material off-gassing applications.
Detailed information on emissions and thermal degradation is crucial for optimizing processes and ensuring adherence to regulatory standards, particularly for high-performance materials subjected to a broad range of process and environmental conditions.
References
- Gaines, L.G.T. (2022). Historical and current usage of per- and polyfluoroalkyl substances (PFAS): A literature review. American Journal of Industrial Medicine, 66(5). DOI: 10.1002/ajim.23362. https://onlinelibrary.wiley.com/doi/10.1002/ajim.23362.
- Panieri, E., et al. (2022). PFAS molecules: A major concern for the human health and the environment. Toxics, 10(2), p.44. DOI: 10.3390/toxics10020044. https://www.mdpi.com/2305-6304/10/2/44.
- Bowers, B.B., Thornton, J.A. and Ryan Christopher Sullivan (2023). Evaluation of iodide chemical ionization mass spectrometry for gas and aerosol-phase per- and polyfluoroalkyl substances (PFAS) analysis. Environmental science. Processes & impacts, 25(2), pp.277–287. DOI: 10.1039/d2em00275b. https://pubs.rsc.org/en/content/articlelanding/2023/em/d2em00275b.
- Mattila, J.M., et al. (2024). Characterizing Volatile Emissions and Combustion Byproducts from Aqueous Film-Forming Foams Using Online Chemical Ionization Mass Spectrometry. Environmental Science & Technology. DOI: 10.1021/acs.est.3c09255. https://pubs.acs.org/doi/10.1021/acs.est.3c09255.
Acknowledgments
Produced using materials originally authored by Priyanka Bansal, Felipe Lopez-Hilfiker, and Katie Schmidt from TOFWERK.

This information has been sourced, reviewed, and adapted from materials provided by TOFWERK.
For more information on this source, please visit TOFWERK.