Man-made chemicals known as per- and polyfluoroalkyl substances (PFAS) are extensively used in a variety of industrial processes and consumer goods due to their water- and oil-repellent qualities.1 Despite their manufacture dating back to the 1940s, global awareness of these substances has increased only in the last decade, largely attributed to their detrimental health outcomes.

Image Credit: zimmytws/Shutterstock.com
These compounds, frequently termed ‘forever chemicals’ owing to their resistance to natural breakdown and their propensity to accumulate in living organisms, have been discovered in atmospheric samples, surface and ocean waters, terrestrial soils, and even in remote Arctic locations.2 Moreover, elevated PFAS levels have been identified indoors, highlighting a significant route for human exposure.3
CompTox, a chemical information repository managed by the U.S. Environmental Protection Agency, lists close to 15,000 distinct synthetic PFAS. The emergence of novel PFAS alongside the simultaneous introduction of next-generation PFAS-like compounds presents considerable challenges for researchers in environmental science and regulatory bodies.
Keeping pace with novel emission sources, developing specialized analytical methodologies, and understanding their toxicological impacts are complex endeavors. Consequently, the majority of these substances are not presently subject to regulatory scrutiny under existing environmental legislation, particularly concerning their direct quantification in ambient air.
The low concentrations of these compounds make their analysis complex and require highly sensitive analytical detection techniques. Established methodologies for gas-phase sampling have historically relied on indirect analytical procedures. These involve employing passive or active air sampling apparatuses that gather specimens onto filters and/or sorbent materials, with subsequent laboratory examination.
While vital, these approaches encounter significant limitations in directly addressing the problem at its origin. For instance, protracted collection durations, coupled with the labor-intensive nature of sample analysis, impede a thorough comprehension of the sources and behaviors of these atmospheric pollutants.
Furthermore, indirect methods are often specific, identifying only known substances, which restricts their capacity to detect new or emerging contaminants. To gain insight into the release of these contaminants into the atmosphere and to understand their origins and transport pathways, real-time measurement techniques can be used, enabling the identification of a broad spectrum of compounds.
Direct PFAS Air Detection via Chemical Ionization
Chemical ionization (CI) employing iodide as a reagent ion has recently emerged as a promising approach for the real-time identification of PFAS in air.4-6 The adaptable TOFWERK Vocus Aim Reactor (leveraging an adduct ionization mechanism) uses a gentle ionization process, which minimizes fragmentation, thereby preserving the original molecular ion.
When combined with a TOFWERK time-of-flight mass spectrometer, this approach enables the accurate determination of molecular formulae for detected ions with a 1 second temporal resolution and exceptional sensitivity.
This investigation aimed to demonstrate the utility of the Vocus Aim Reactor as a tool for real-time quantitative PFAS Air Detection and to introduce this novel technique to a wider audience beyond the scientific community. This method directly analyzes air without requiring sample collection or prior separation, differentiating it from conventional approaches.
To acquire quantitative data, a dependable calibration strategy for PFAS is fundamental. Generally, chemical ionization can be calibrated using a multicomponent gas cylinder for compounds possessing adequate vapor pressure and limited reactivity. Alternatively, certified permeation tubes with established permeation rates can be employed, but the availability of these standards is restricted.
For less volatile molecules, solutions with a known concentration can be prepared and vaporized using commercially accessible liquid calibration systems (LCS). However, these devices feature extensive surfaces where molecules with very low volatility can adhere, leading to prolonged response times and the risk of persistent contamination from toxic compounds, such as PFAS.
To address these issues, TOFWERK’s objective is to showcase a standardized and reliable calibration method, including calibration factors, detection thresholds, and humidity dependencies for these emerging contaminants, and to contrast this method with a recently described calibration approach.3 Additionally, the team assessed two ion chemistries available within the Vocus Aim Reactor: iodide and nitrate.
This methodology is also extended to other emergent contaminants, including pesticides, which present similar challenges in gas-phase measurement and calibration due to their high toxicity and contamination concerns.
Experimental
Calibration measurements were carried out using a Vocus 2R equipped with an Aim Reactor operating at a measurement frequency of 0.5 Hz. The reactor was operated at 50 mbar and 50 °C for both iodide and nitrate ion chemistries. Liquid calibration standards of PFAS were formulated in methanol, methyl acetate, and dichloromethane at concentrations spanning from 0.2 to 4 mg L-1 to investigate potential solvent-induced variations in calibration outcomes.
A 250 μL glass syringe (Hamilton) and a syringe pump (KD Scientific) were used to evaluate the differences between single and continuous injections of liquid standards. The examined injection rates ranged from 20 μL/hour to 800 μL/hour. Single injections involved the immediate introduction of a specific volume of the liquid standard, while continuous injections maintained a steady flow of the liquid standard over time.
This comparison was intended to determine which method yielded more consistent and precise calibration results by assessing precision, stability, and reproducibility.
The sample was introduced into a 130 °C heated 0.5-inch OD Sulfinert tubing at a 90° angle via a GC septum into the injection port. The tubing was fitted with a heating element, allowing for straightforward sonication or replacement of the tubing if necessary.
The injector was continuously purged with two sLPM of UHP N2, with the surplus flow directed to the exhaust. Figure 1 illustrates the experimental setup employed during the calibrations. Given that the Aim Reactor allows for switching between reagent ions, a selection of the chemicals was calibrated in both iodide and nitrate modes.
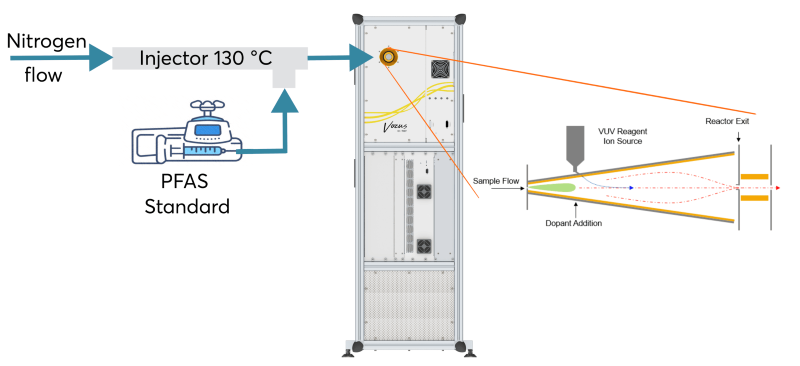
Figure 1. Experimental setup used in the calibration of the PFAS. Image Credit: TOFWERK
Table 1. Iodide CIMS emerging contaminants calibration summary. All calibration factors are reported in counts per second (cps) per part per trillion by volume (pptv) and normalized per million counts of total reagent ion signal. Source: TOFWERK
| Compound |
Calibration factor
(ncps ppt-1) |
LOD 1 s
(ppt) |
LOD 1 min
(ppt) |
LOD 1 min
(ng/m3) |
| TFA |
4.30 |
30.0 |
4.0 |
18 |
| 6:2 FTOH |
5.40 |
1.8 |
0.3 |
6 |
| 8:2 FTOH |
5.50 |
1.1 |
0.2 |
4 |
| PFBA |
5.29 |
1.3 |
0.2 |
2 |
| PFPeA |
5.92 |
1.7 |
0.2 |
2 |
| PFHxA |
5.27 |
0.9 |
0.1 |
1 |
| PFHpA |
4.29 |
1.0 |
0.2 |
3 |
| PFOA |
2.77 |
1.3 |
0.3 |
6 |
| PFNA |
1.86 |
2.0 |
0.3 |
6 |
| PFDA |
0.77 |
3.0 |
0.5 |
11 |
| PFUnA |
0.36 |
3.4 |
0.5 |
13 |
| PFDoDA |
0.16 |
4.7 |
0.7 |
19 |
| PFTriDA |
0.06 |
7.6 |
1.2 |
36 |
| PFTeDA |
0.03 |
6.5 |
1.0 |
32 |
| DDT |
0.29 |
5.0 |
0.7 |
10 |
| Pentachlorophenol |
0.29 |
14.0 |
2.0 |
22 |
Results
Using the calibration setup previously described, Table 1 presents the calibration factors and detection limits (LODs) for two fluorotelomer alcohols (FTOHs), 11 perfluorinated carboxylic acids (PFCAs), pentachlorophenol, and 4,4 DDT. The LODs were measured in both nitrogen and ambient air to ascertain potential interferences arising from the dilution medium of the injected solutions.
The 1 Hz LODs showed variations of less than 10 % between the matrices and were determined to be in the low parts-per-trillion (ppt) range. By increasing the averaging time to one minute, the LODs were reduced by nearly one order of magnitude.
Figure 2 displays the calibration curves from a single experiment for 8:2 FTOH, PFBA, PFHxA, and PFOA using iodide as the reagent ion and the syringe pump methodology. Good linearity (R2 > 0.98) was observed for the calibrated PFAS, even at lower ppt levels, indicating the AIM reactor’s superior capacity to detect PFAS in the low parts-per-trillion range.
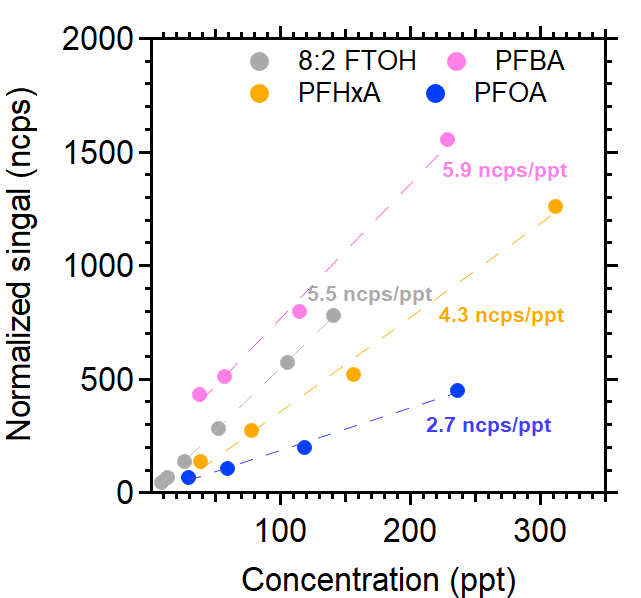
Figure 2. TOFWERK iodide Aim calibration curves for selective PFAS. Image Credit: TOFWERK
The sensitivity for perfluorocarboxylic acids (PFCAs) was also measured using NO3- as the reagent ion. On average, the measured sensitivities were within 20 % of those acquired with the iodide mode (Figure 3), suggesting that the nitrate reagent ion is similarly effective in detecting this subclass of PFAS. For both ion chemistries, the measured sensitivities decrease with increasing PFCA molecular size. This is likely associated with their ability to be efficiently transferred to the gas phase through this calibration approach.
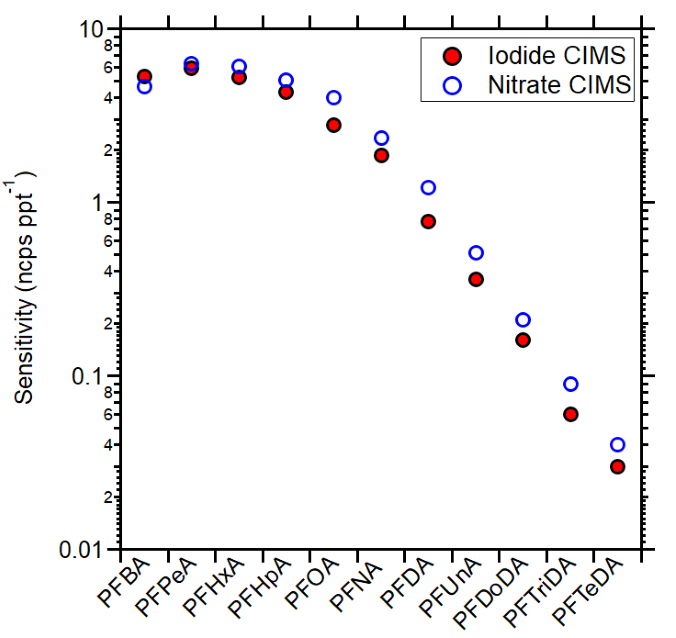
Figure 3. Measured PFCAs sensitivities using iodide CIMS (red full circles) and in nitrate CIMS (blue open circles). Image Credit: TOFWERK
The findings from the evaluation of the two calibration methods (single syringe injections of identical volume and the continuous syringe pump injection technique) are presented in Figure 4. Generally, these two methods agree within 30 % for the volatile fraction of PFAS.
However, for some compounds, considerable variability in signal response was noted with single injections of the same volume and concentration, as illustrated in Figure 4c. Although the direct injection approach is relatively simple and cost-effective, it introduces variability due to human involvement.
Conversely, the syringe pump method demonstrated enhanced stability in response to variations in injected flow rates and effectively mitigated operator-induced errors. Consequently, all subsequent results reported herein are based on calibrations using the syringe pump method.
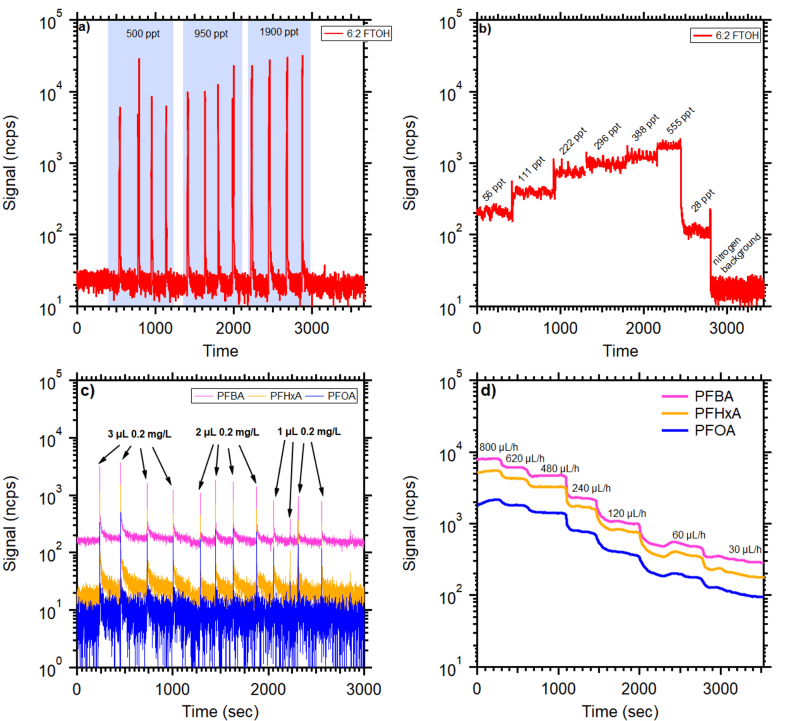
Figure 4. Comparison between direct injections (a & c) and syringe pump approach (b & d) for 6:2 FTOH and selected PFCAs. For manual injections, the same volumes of solution with increasing concentration were used. For syringe pump injection, one concentration solution with varying injection rates was used. Calculated mixing ratios of 6:2 FTOH in the air are highlighted by a blue shaded area. Image Credit: TOFWERK
To accurately measure in environments where relative humidity (RH) fluctuates, such as ambient air, it is essential to understand how sensitivity changes with varying humidity levels. Water can profoundly influence the sensitivity of species detected by CI, with RH-dependent sensitivities differing across various compound classes.
As a result, measurements in environments with changing humidity necessitate time-consuming calibrations for distinct RH conditions. To mitigate this water dependency within the Vocus Aim Reactor, the team employed a water vapor control system comprising a regulated flow of five sccm of acetonitrile as a so-called dopant molecule.
The dopant displaces water molecules that would typically attach to the reagent ions, ensuring that the modified reaction mechanism is no longer significantly dependent on fluctuating water vapor conditions. Figure 5a illustrates the relative sensitivity shift for selected PFAS as a function of increasing humidity while employing the dopant.
Our findings indicate a minor decreasing trend. Nonetheless, it is important to recognize that the interpretation of this trend is complicated by the inherent measurement error, particularly concerning reproducibility across replicates. Furthermore, the increased variation in sensitivity with rising humidity levels adds to the complexity of interpretation.
Figure 5b outlines the influence of matrix solvent selection on the sensitivity of PFAS detection. Significant divergence was observed with dichloromethane, which is likely attributable to its heightened volatility. Differences between methanol and ethyl acetate were minimal, with the latter emerging as the preferred option due to its lower toxicity.
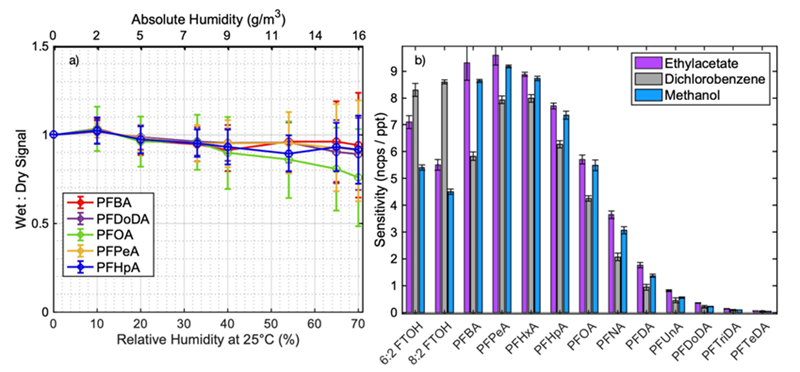
Figure 5. a) Sensitivity normalized to dry conditions as a function of increasing humidity in relative (25 °C) and absolute values. The error bars represent the standard deviation, calculated from nine measurements conducted on different days. b) Sensitivity differences for various solvents. Image Credit: TOFWERK
Conclusion
This study demonstrated the Vocus Aim Reactor’s capability to deliver real-time detection and quantification of PFAS, with temporal resolution superior to that of traditional indirect techniques.
With sensitivities ranging from 0.5–5 ncps/ ppt and detection limits extending to hundreds of ppq levels, this method offers high sensitivity and specificity. While these detection limits surpass typical requirements for background monitoring of ambient air, the speed and precision of the Vocus Aim Reactor make it well-suited for applications such as real-time identification of volatile PFAS sources or locating potential leaks where concentrations are expected to be considerably higher.
Furthermore, it can be seen that Aim is appropriate for measurements in ambient settings with fluctuating humidity. This technique also holds potential for improving understanding of PFAS environmental pathways through atmospheric chamber experiments, indoor air monitoring, consumer product assessments, and material emission testing. Additionally, it can aid in regulatory monitoring of flue gases to ensure compliance with emission standards.
References
- Glüge, J., et al. (2020). An overview of the uses of per- and polyfluoroalkyl substances (PFAS). Environmental Science: Processes & Impacts, 22(12). DOI: 10.1039/d0em00291g. https://pubs.rsc.org/en/content/articlelanding/2020/em/d0em00291g.
- Evich, M.G., et al. (2022). Per- and polyfluoroalkyl substances in the environment. Science, 375(6580). DOI: 10.1126/science.abg9065. https://www.science.org/doi/10.1126/science.abg9065.
- Davern, M.J., et al. (2024). External liquid calibration method for iodide chemical ionization mass spectrometry enables quantification of gas-phase per- and polyfluoroalkyl substances (PFAS) dynamics in indoor air. The Analyst, 149(12), pp.3405–3415. DOI: 10.1039/d4an00100a. https://pubs.rsc.org/en/content/articlelanding/2024/an/d4an00100a.
- Barber, J.L., et al. (2007). Analysis of per- and polyfluorinated alkyl substances in air samples from Northwest Europe. Journal of Environmental Monitoring, 9(6), p.530. DOI: 10.1039/b701417a. https://pubs.rsc.org/en/content/articlelanding/2007/em/b701417a.
- Riedel, T.P., et al. (2019). Gas-Phase Detection of Fluorotelomer Alcohols and Other Oxygenated Per- and Polyfluoroalkyl Substances by Chemical Ionization Mass Spectrometry. Environmental Science & Technology Letters, 6(5), pp.289–293. DOI: 10.1021/acs.estlett.9b00196. https://pubs.acs.org/doi/10.1021/acs.estlett.9b00196.
- Bowers, B.B., et al. (2023). Evaluation of iodide chemical ionization mass spectrometry for gas and aerosol-phase per- and polyfluoroalkyl substances (PFAS) analysis. Environmental science. Processes & impacts, 25(2), pp.277–287. DOI: 10.1039/d2em00275b. https://pubs.rsc.org/en/content/articlelanding/2023/em/d2em00275b.
- Mattila, J.M., Li, E.Y. and Offenberg, J.H. (2023). Tubing material considerably affects measurement delays of gas-phase oxygenated per- and polyfluoroalkyl substances. Journal of the Air & Waste Management Association, 73(5), pp.335–344. DOI: 10.1080/10962247.2023.2174612. https://www.tandfonline.com/doi/full/10.1080/10962247.2023.2174612.
- Riva, M., et al. (2024). Evaluation of a reduced pressure chemical ion reactor utilizing adduct ionization for the detection of gaseous organic and inorganic species. DOI: 10.5194/egusphere-2024-945. https://egusphere.copernicus.org/preprints/2024/egusphere-2024-945/.
Acknowledgments
Produced using materials originally authored by Spiro Jorga and Veronika Pospilova from TOFWERK.

This information has been sourced, reviewed, and adapted from materials provided by TOFWERK.
For more information on this source, please visit TOFWERK.