Sponsored by HORIBAOct 24 2013
Raman spectroscopy is a non-contact and non-destructive and does not require any sample preparation. Hence, the small traces of evidence can be preserved for further investigations of the same specimen to confirm results. Additionally, the confocal system’s high spatial resolution enables performing a complete analysis with small sample volume. These qualities make the technique as an efficient and robust analytical method in the field of Forensic Science.
The high sensitivity of the Raman effect to slight variations in crystallographic structure and chemical composition is very useful in the analysis of illegal drugs, providing key insights into the origin of the method of drug synthesis. This article discusses the application of Raman spectroscopy in the analysis of cocaine and its derivatives to demonstrate the key advantages of the method in drug analysis.
Instrumentation
A LabRAM integrated with a 785nm diode laser and a 633nm laser was used to perform the analysis. The 785nm diode laser was used to record all types of Raman spectra on the LabRam. This laser wavelength hinders interference caused by fluorescence that could be emitted by contaminants. Nevertheless, the use of the standard 633nm HeNe laser provided good results, if not better, when compared to those captured with the diode laser.
Since glass emits a fluorescence spectrum at 785nm, the spectra captured in glass vials may exhibit a large background at around 1400cm-1 because of the glass being close to the focal volume. Nevertheless, the standard feature of the LabRAM software can be used to generate a spectrum of the sample devoid of the glass contribution through effectively subtracting the glass background.
Raman bands of Cocaine
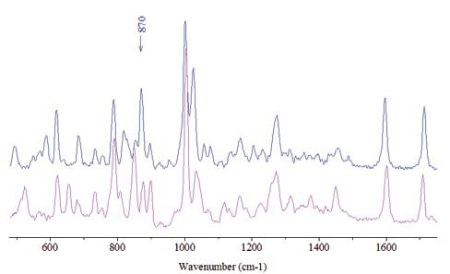
Figure 1. Spectra of free base cocaine (bottom) and of cocaine HCl.
Cocaine HCl, water and ammonia or sodium bicarbonate are used to prepare free base cocaine (Crack). The hydrochloride is removed from the mixture when heated. The Raman spectra of free base cocaine and cocaine hydrochloride are illustrated in Figure 1. Spectra were directly recorded through the glass vials consisting of the free particles, by means of a long working distance objective for particle analysis far behind the glass window. This approach eliminates the risk of sample degradation or contamination. The most prominent spectral features are listed in Table 1.
Table 1. Position of the main Raman bands of Cocaine HCl and free base cocaine in the range of 700-1800cm-1.
| Free Base |
HCI |
| 788 cm-1 |
786 cm-1 |
| 847 cm-1 (s) |
851 cm-1 (m) |
| 872 cm-1 (m) |
870 cm-1 (s) |
| 896 cm-1(m) |
894 cm-1 (m) |
| 1003 cm-1 |
999 cm-1 |
| 1601 cm-1 |
1596 cm-1 |
| 1713 cm-1 |
1709 cm-1 |
With an accuracy of about 1 cm-1, the measurement results were able to clearly distinguish the differences between the frequencies. Nonetheless, comparing the signatures in the spectral region of 845-900 cm-1 (grey cells in Table 1) is the simplest approach to differentiate the two cocaine species. In the cocaine HCl sample, the most intense among the triplet is the central band (870cm-1), while the lowest frequency band (847cm-1) is the most intense in the free base form.
Cocaine Related Compounds
The Raman spectra of cocaine related compound are depicted in Figure 2, each showing distinct Raman features and thus can be easily distinguished.

Figure 2. Spectra of three cocaine related compound.
Detecting Drug Particles Within a Mixture
In these experiments, the physical and chemical form of the cocaine powders is characterized.
Mixture with Cutting Agents
The analyst often needs to analyze mixtures of drug particles and cutting agents. Manitol, myoinositol and sorbitol have been analyzed as possible cutting agents and their Raman spectra are depicted in Figure 3. The results clearly show that the spectra of cutting agents differ from each other as well as from the reported spectra of drugs.

Figure 3. Spectra of three possible cutting agents.
Vials of calibrated mixtures of cocaine and different possible cutting compounds were studied to determine the possibility of identifying and recording the presence and form of cocaine. The example shown in Figure 4 is a (50:50)%w. mixture of cocaine HCl with Manitol. These Raman spectra show very low Raman signal of Manitol, with an intensity pattern between 845 and 900 cm-1, thereby indicating the presence of cocaine HCl instead of the free base.

Figure 4. Spectrum obtained after substraction of a (50:50)%w. mixture of cocaine- HCl:manitol.
Traces Analysis: Mixture of Three Drugs
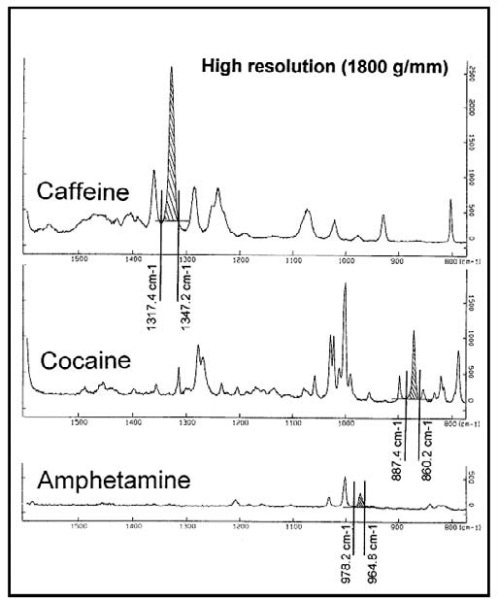
Figure 5. Reference Raman spectra of three drug compounds and the integration range under their characteristic band.
The Raman spectra maps of a surface containing trace quantities of regularly controlled drug substances such as Cocaine, Caffeine and Amphetamine are shown here. The reference spectra (Figure 5) provided the spectral features of the three different drugs. Confocal point mapping was used to analyze an area of 25µmx35µm on the contaminated surface. A 2D Raman image was then constructed by integrating the area of the characteristic band of each compound (Figure 6) to reveal localization of the three substances.

Figure 6. Confocal mapped images generated from Raman spectra by the integration under the characteristic band of each compound.
Conclusion
The results clearly demonstrate the ability of Raman spectroscopy to analyze suspected drug products for confirming the nature of the material. The Raman analysis enables identification of slight differences. The use of confocal Raman spectroscopy allows determination of traces on surfaces or in vials that are transparent to the visible excitation wavelength such as polymer and glass. Characteristic Raman features can be obtained from mixtures of different drug compounds or mixtures of drug particles and additives (cutting agents). These features facilitate identifying the nature of each component and generating Raman maps to determine the distribution of each component.

This information has been sourced, reviewed and adapted from materials provided by HORIBA.
For more information on this source, please visit HORIBA.