Cinnamon is an important spice that is extensively used around the world. Although predominantly known for its culinary use, cinnamon is also valued for its major health benefits. For example, the spice reduces heart disease risk factors, lowers blood sugar levels, and positively impacts neurological disorders.
Cinnamon has high levels of antioxidants, which is the likely reason behind its health benefits. It is obtained from the dried inner bark of Cinnamomum verum (Ceylon cinnamon) and is called as “true” cinnamon. C. burmannii (Indonesian cassia), barks of C. cassia (Chinese cassia), and C. loureiroi (Saigon cassia), collectively called cassia cinnamon, are also marketed as cinnamon.
Ceylon cinnamon has been replaced by Cassia cinnamon, in particular Indonesian cassia, as they are cheaper, and are now the species most widely used in the United States, Europe, and Canada. It has been observed that cassia cinnamon includes high levels of coumarin, a naturally occurring compound that can be hepatotoxic to certain people sensitive to it. Conversley, Ceylon cinnamon has relatively lower levels of coumarin.
Addressing a Health Concern
Increased consumption of cassia cinnamon means an increased intake of coumarin. This has prompted the European Food Safety Authority (EFSA) to determine a tolerable daily intake (TDI) of 0.1 mg coumarin/kg body weight. Several clinical trials have revealed that cassia cinnamon possess limited antidiabetic activity. This has led to several dietary supplements that include both true and cassia cinnamon as ingredients. The labels on the supplement products proposed usage levels from 1 g up to 4 g of cinnamon per day. The quantities of coumarin and other marker compounds in cinnamon bark, foods, powder, and supplements have been evaluated in advance with a certified UPLC-UV/MS technique.
Bruker BioSpin have created a quick and fully automated high throughput technique to quantitate coumarin, the key flavor compound cinnamaldehyde, and other key parts in cinnamon-containing products using 1H-Nuclear Magnetic Resonance (NMR).
Materials and Methods
For the experiment, three brands of ground cinnamon powder (P1-P3) were bought from local stores. P1 and P2 were marked as cassia with no mention of species, while P3 was marked as Ceylon cinnamon. Four different brands of capsule cinnamon supplements were also purchased, (S1-S4), where S1 was marked as certified organic Ceylon cinnamon, and S2-S4 marked as Cinnamomum cassia or simply cassia.
NMR samples of cinnamon were made by measuring ~200 mg of cinnamon supplement sample and powdered cinnamon, then adding 1.5 mL CDCl3, sonicating, and finally filtering 600 uL of the CDCl3 extract within a 5 mm NMR tube. NMR standard samples of expected cinnamon parts such as coumarin 1, cinnamyl alcohol 2, cinnamaldehyde 3, cinnamic acid 4, eugenol 5, and cinnamyl acetate 6, were formulated by dissolving about 90 to 150 mg of each in 2 mL CDCl3 and then transferring 600 uL of each to 5 mm NMR tubes. A standard mixture (SM) of the predicted parts was prepared by integrating each of the predicted component solutions.
A Bruker AVANCE III spectrometer, using the 1H noesyigld1d pulse sequence, was employed to record NMR spectra at 600 MHz. Spectra were obtained with 64 scans and at a relaxation delay of 10 seconds, utilizing a sweep width of 64 K points and 20 ppm. Referencing was to the lock solvent. Using Bruker’s AssureNMR version 2.0 software package, NMR analysis was performed automatically. The software package uses the PULCON equation and an external quantification standard for complete quantification results.
In order to measure the daily dose of coumarin, the size of serving and the NMR data was used. The proposed serving sizes for the supplements as displayed on product labels (Table 1) were as follows: S1 = 2*500 mg capsules/day; S2 = 2*500 mg capsules daily, S3 = 1*1000 mg capsules twice a day; and S4 =2*1000 mg capsules twice a day. Daily coumarin doses were measured based on these recommendations. It should be noted that the coumarin content of supplement S4 surpasses the TDI for an average 60 kg human. For the sake of comparison, the overall coumarin content of 1.0 g of P1-P3 was also determined.
Generation of an NMR Spectra Database
The 1H NMR spectra of basic reference material for coumarin 1, cinnamyl alcohol 2, cinnamaldehyde 3, cinnamic acid 4, eugenol 5, and cinnamyl acetate 6 were then utilized to create a NMR spectral database (SBASE) that functioned as a chemical shift reference library for the automated identification of the chief metabolites of cinnamon. Using AssureNMR, SBASE entries were developed by eliminating solvent, noise, and impurity peaks, as well as wide exchangeable signals from the 1H NMR spectrum (Figure 1). The ensuing SBASE entry contained peak shape, peak position, coupling, and peak intensity data for individual compounds to facilitate precise identification match.
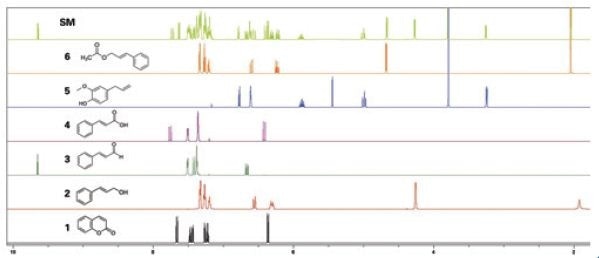
Figure 1. 1H-NMR spectra of coumarin (1), cinnamyl alcohol (2), cinnamaldehyde (3), cinnamic acid (4), eugenol (5), cinnamyl acetate (6), and Standard Mixture (SM). Image Credit: Bruker BioSpin Group
Detection and Quantification of Key Components
Bruker’s AssureNMR was used to develop an AssureNMR technique for automated detection and quantitation of coumarin and five other major components of cinnamon. The AssureNMR technique was created by importing the spectra from the NMR SBASE, detecting peaks for quantification, choosing lineshape fitting for the resolved compound resonances, and choosing reporting criteria. If quantitation was performed on more than one resonance of a compound, then the average was noted along with the standard deviation.
The AssureNMR technique can automatically utilize an external quantitation standard that employs the PULCON method and a known concentration of external sample to set up quantitative results. Once the automated AssureNMR technique has been implemented, quantitative 1H NMR spectra were acquired by utilizing a 1D-NOESY pulse sequence, as indicated in Figure 1; sample preparation data that is the weight of each dry component and volume of CDCl3, was then gathered by the software. The AssureNMR was used to perform all concentration measurements in automation. Following this, the quantification calibration file was automatically stored regarding each cinnamon sample, and this file was used by the software to measure the metabolite concentration in each sample using the PULCON method. Results for the cinnamon components were accounted by the software as mg compound/g cinnamon in a comprehensive ExpertReport file, which is automatically produced and includes quantitation detail and results. Excerpts of which are illustrated in Figure 2.
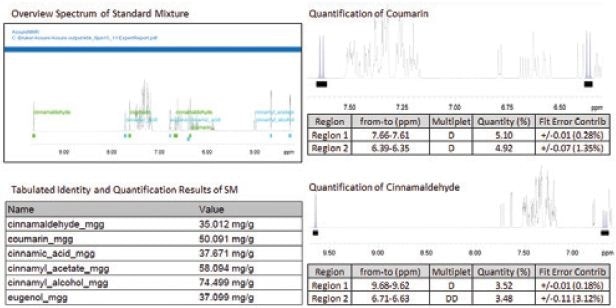
Figure 2. AssureNMR quantitation of the Standard Mixture (SM), results and quantitative details from the ExpertReport. Image Credit: Bruker BioSpin Group
Results
Automated analysis revealed the existence of certain metabolites that were possibly present in samples of cinnamon. Table 1 shows the quantification results obtained from the ExpertReports. Visual inspection of the spectra established that there was no detectable amounts of the components listed as “nd”.
Table 1. Concentration measured for each components (mg/g cinnamon). Source: Bruker BioSpin Group
| Sample |
Source |
Coumarin |
Recommended Serving (g/day) |
mg coumarin per day* |
| 1 |
2 |
3 |
4 |
5 |
6 |
| S1 |
C. verum |
0.009 |
nd |
0.840 |
nd |
nd |
nd |
1 |
0.009 |
| S2 |
Cassia |
0.498 |
nd |
2.071 |
nd |
nd |
nd |
1 |
0.498 |
| S3 |
Cassia |
0.972 |
nd |
6.091 |
nd |
nd |
nd |
2 |
1.944 |
| S4 |
Cassia |
1.519 |
0.257 |
13.575 |
nd |
nd |
nd |
4 |
6.076 |
| P1 |
Cassia |
0.885 |
0.177 |
6.343 |
nd |
nd |
nd |
1 |
0.885 |
| P2 |
Cassia |
3.082 |
0.413 |
23.882 |
nd |
nd |
nd |
1 |
3.082 |
| P3 |
C. verum |
nd |
0.265 |
3.041 |
nd |
0.195 |
nd |
1 |
~0.000 |
*mg of coumarin that would be consumed based on the daily recommended serving and the actual amount of coumarin detected. The TDI for a 60 kg human is 6.0 mg/day. “nd” denotes “not detected”
As predicted, results revealed that coumarin 1 is identified in Cassia samples at considerably higher amounts than samples taken from C. verum. Significant difference in the concentration of coumarin happens in the Cassia samples, ranging from 0.498 to 3.082 mg/g of cinnamon. Concentrations of the key flavor part, cinnamaldehyde 3, were also found to be highly variable in the samples ranging from 0.840 to 23.882 mg/g of cinnamon.
Conclusion
Nuclear Magnetic Resonance (NMR) serves as an important tool for detection and quantification of components in cinnamon, a natural health product, and eliminates the necessity for extra purification or separation processes. NMR’s high reproducibility allows complete automated NMR acquisition, examination and reporting, and utilizes an NMR spectral database that can be further extended to explore more cinnamon components. It is possible to transfer the NMR SBASE and AssureNMR technique to other instruments and sites for product appraisal without any need for recreating reference spectra.
NMR spectroscopy can also be adapted for other compound mixtures where component authentication is essential for the appraisal and quality control of products.

This information has been sourced, reviewed and adapted from materials provided by Bruker BioSpin Group.
For more information on this source, please visit Bruker BioSpin Group.