To control and to optimize processing methods, granular materials and fine powders have to be precisely characterized for use in industrial applications. Chemical composition, morphology, granulation, and other characterization methods are related to the properties of grains and the density, flowability, blend stability and electrostatic properties of bulk powder.
Over the last decade, updates have been made to quality control laboratories as most of the techniques used in research and development (R&D) or quality control laboratories for the behavior of bulk powder are based on archaic measurement techniques. Now, meeting the present requirements of R&D laboratories and production departments, the measurement processes have become automatized and rigorous initialization methods were developed to obtain reproducible and interpretable results.
The use of image analysis techniques improves measurement precision. Therefore, we will focus on GranuCharge instruments even though a range of measurement methods has been developed to cover the requirements of industry processing powders and granular materials.
GranuCharge
During the flow of powder inside a mixer, silo, conveyor or another device, the triboelectric effect, which takes place at the contact between grains or between grains and the device, causes the apparition of electrostatic charges that are created inside a powder during a flow. This is an exchange of charge at the contact between the two solids. Therefore, the characteristics of the powder and the nature of the material used to assemble the device are vital parameters.
During a flow in contact with a certain material, the GranuCharge instrument automatically and concisely measures the electrostatic charge quantity created inside a powder. The sample flows inside a vibrating V-tube, falling then into a Faraday cup attached to an electrometer that measures the charge acquired by the powder during the flow. A rotating or a vibrating device is used to regularly feed the V-tube to gain reproducible results.
Powder Description
At low humidity, anhydride rings are closed and show low conductivity (Figure 1). Glass powder coated with an ethylene copolymer grafted with succinic anhydride (ESA) has been chosen for analysis. The succinic anhydride rings shown in figure 1a hydrolyze into succinic acid when exposed to moisture (Figure 1b).
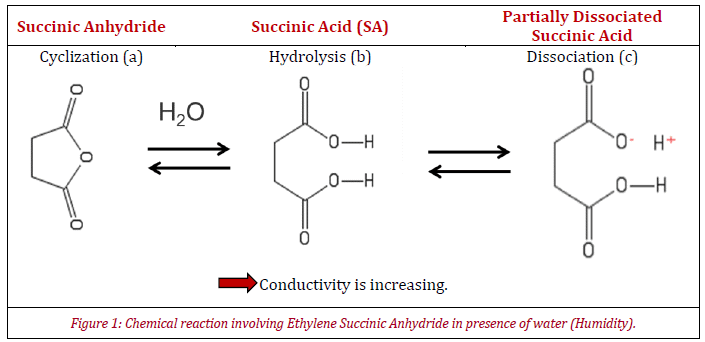
While a sample with low conductivity has difficulty in dissipating electrical charges, due to higher conductivity, the acid partially dissociates in the presence of water as it is weak (see Figure 1c). Therefore, this high conductivity displays a good ability to dissipate electrical charges.
GranuCharge Analysis
Experimental Protocol
For each experiment investigating triboelectric effect using the GranuCharge, stainless-steel 316L pipes and a rotating feeder were utilized (Figure 2). The powder was not recycled after measurement and the product amount used for each measure was 50 g. To show accuracy, testing has been repeated.

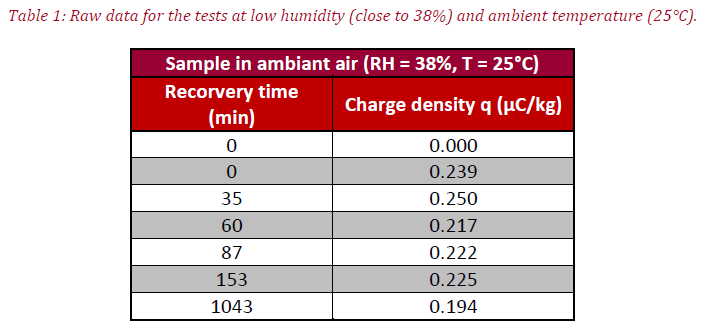
Two tests were carried out; the former analyzes the influence of low moisture content (at RH = 38% and T = 25 °C) and the latter displays the influence of high moisture content on the charge density (at RH = 85% and T = 80 °C). Prior to the experiment, powder mass (mp, in g) is recorded. At the start of testing, the initial powder charge (Qi, in μC) is measured by introducing powder inside the Faraday cup. Afterward, the powder is poured inside the rotating feeder and experimentation follows whereby the final charge is measured at the close (Qf, μC).
After putting the samples in contact with humidity, the last points are recorded. For both tests, measurement protocol is the same, the first point (at t = 0 min) corresponding to ambient air conditions after opening the sample box. The second point involves sample electrical charge after one hour in an oven at 170 °C.
Results
The tables summarize the raw results obtained for the first and second tests, meanwhile figure 3 represents charge density variation (Mean q, in μC/kg) versus the time of the experiment. Interestingly, at low moisture content (red curve) a slight decrease of electrical charge is seen after 1140 min in contact with humidity, caused by the modifications of the coating properties (see Figure 1) due to the transition between structures (a) and (c).
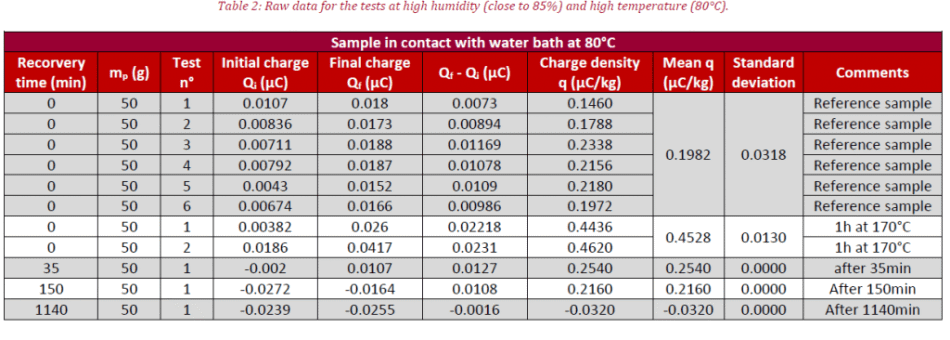
The same trend for charge density variation is seen here at high humidity (blue curve), where the transition between those two organic compounds is faster. This is due to compound (c) being anti-static because of high conductivity and the difference between the initial points in charge density for the two tests are explained by the fact that the chemical reaction is a chemical equilibrium (see Figure 1).
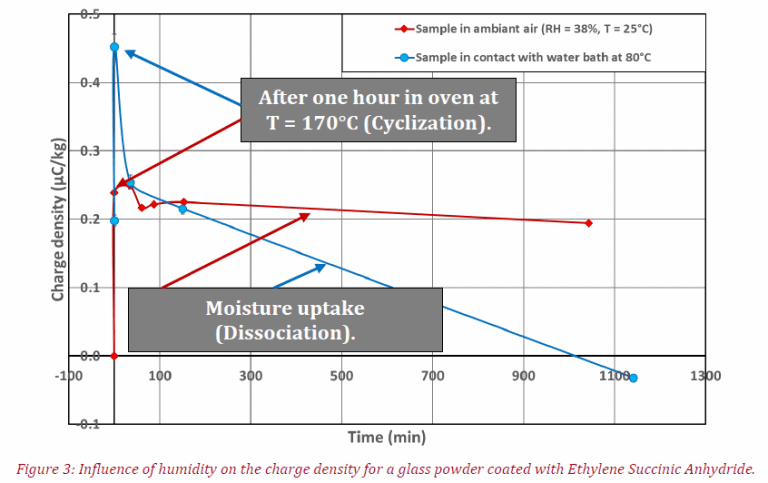
The coating composition of the glass powder could correspond to a blend of forms (a), (b) and (c) after removing the sample from its cardboard box. The sample has a negative charge density at the latter part of the experiment for testing with a high moisture value (blue curve). This could be due to the negatively charged ESA coating.
Conclusions
It is concluded that the GranuCharge can measure the influence of humidity on the coating of glass as well as some changes and developments in powder surface properties.
Bibliography
- Cascade of granular flows for characterizing segregation, G. Lumay, F. Boschin, R. Cloots, N. Vandewalle, Powder Technology 234, 32-36 (2013).
- Combined effect of moisture and electrostatic charges on powder flow, A. Rescaglio, J. Schockmel, N. Vandewalle and G. Lumay, EPJ Web of Conferences 140, 13009 (2017).
- Compaction dynamics of a magnetized powder, G. Lumay, S. Dorbolo and N. Vandewalle, Physical Review E 80, 041302 (2009).
- Compaction of anisotropic granular materials: Experiments and simulations, G. Lumay and N. Vandewalle, Physical Review E 70, 051314 (2004).
- Compaction Dynamics ofWet Granular Assemblies, J. E. Fiscina, G. Lumay, F. Ludewig and N. Vandewalle, Physical Review Letters 105, 048001 (2010).
- Effect of an electric field on an intermittent granular flow, E. Mersch, G. Lumay, F. Boschini, and N. Vandewalle, Physical Review E 81, 041309 (2010).
- Effect of relative air humidity on the flowability of lactose powders, G. Lumay, K. Traina, F. Boschini, V. Delaval, A. Rescaglio, R. Cloots and N. Vandewalle, Journal of Drug Delivery Science and Technology 35, 207-212 (2016).
- Experimental Study of Granular Compaction Dynamics at Different Scales: Grain Mobility, Hexagonal Domains, and Packing Fraction, G. Lumay and N. Vandewalle, Physical Review Letters 95, 028002 (2005).
- Flow abilities of powders and granular materials evidenced from dynamical tap density measurement, K. Traina, R. Cloots, S. Bontempi, G. Lumay, N. Vandewalle and F. Boschini, Powder Technology, 235, 842-852 (2013).
- Flow of magnetized grains in a rotating drum, G. Lumay and N. Vandewalle, Physical Review E 82, 040301(R) (2010).
- How tribo-electric charges modify powder flowability, A. Rescaglio, J. Schockmel, F. Francqui, N. Vandewalle, and G. Lumay, Annual Transactions of The Nordic Rheology Society 25, 17-21 (2016).
- Influence of cohesives forces on the macroscopic properties of granular assemblies, G. Lumay, J. Fiscina, F. Ludewig and N. Vandewalle, AIP Conference Proceedings 1542, 995 (2013).
- Linking compaction dynamics to the flow properties of powders, G. Lumay, N. Vandewalle, C. Bodson, L. Delattre and O. Gerasimov, Applied Physics Letters 89, 093505 (2006).
- Linking flowability and granulometry of lactose powders, F. Boschini, V. Delaval, K. Traina, N. Vandewalle, and G. Lumay, International Journal of Pharmaceutics 494, 312–320 (2015).
- Measuring the flowing properties of powders and grains, G. Lumay, F. Boschini, K. Traina, S. Bontempi, J.-C. Remy, R. Cloots, and N. Vandewall, Powder Technology 224, 19-27 (2012).
- Motion of carbon nanotubes in a rotating drum: The dynamic angle of repose and a bed behavior diagram, S. L. Pirard, G. Lumay, N. Vandewalle, J-P. Pirard, Chemical Engineering Journal 146, 143-147 (2009).
- Mullite coatings on ceramic substrates: Stabilisation of Al2O3–SiO2 suspensions for spray drying of composite granules suitable for reactive plasma spraying, A. Schrijnemakers, S. André, G. Lumay, N. Vandewalle, F. Boschini, R. Cloots and B. Vertruyen, Journal of the European Ceramic Society 29, 2169–2175 (2009).
- Rheological behavior of β-Ti and NiTi powders produced by atomization for SLM production of open porous orthopedic implants, G. Yablokova, M. Speirs, J. Van Humbeeck, J.-P. Kruth, J. Schrooten, R. Cloots, F. Boschini, G. Lumay, J. Luyten, Powder Technology 283, 199–209 (2015).
- The influence of grain shape, friction and cohesion on granular compaction dynamics, N. Vandewalle, G. Lumay, O. Gerasimov and F. Ludewig, The European Physical Journal E (2007).

This information has been sourced, reviewed and adapted from materials provided by Granutools.
For more information on this source, please visit Granutools.