Liquid scintillator detectors are often used in neutrino and astroparticle detection experiments. Linear alkylbenzene (LAB) is a liquid scintillator that is increasingly used in large-scale detection investigations.
LAB is a particularly appealing scintillator because of its low cost, high flash point, and low toxicity, which make it easier to handle than other volatile, toxic organic solvents. LAB is typically used in combination with the fluor 2,5-diphenyloxazole (PPO), which improves luminescence yield and pushes it to longer wavelengths.
The Daya-Bay1 and RENO2 neutrino detection experiments already use the LAB/PPO scintillator system, and the forthcoming SNO+3 and JUNO4 detectors will also use it.
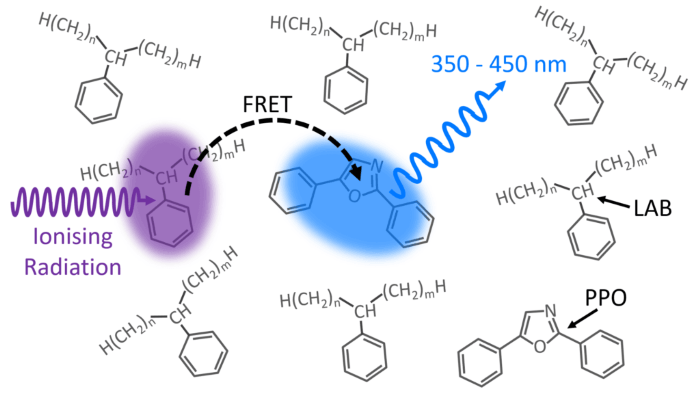
Figure 1. LAB/PPO Scintillator System. Image Credit: Edinburgh Instruments
Figure 1 shows a schematic of the scintillation process in the LAB/PPO system. Ionizing radiation, such as γ-rays, travels through the LAB solvent and excites a LAB molecule to a higher energy state.
The extra energy in the LAB is non-radiatively transmitted (FRET) to a neighboring PPO molecule, propelling it to an excited state. Radiative de-excitation of the PPO to its ground state yields detectable violet/blue fluorescence.
Because energy is exchanged between the LAB and PPO via FRET, the luminescence behavior is determined by the number of PPO molecules and their distance from excited LAB molecules.
Higher concentrations of PPO reduce the average spacing between excited LAB and PPO molecules, boosting luminescence production while lowering average luminescence lifespan.
Many detection investigations use pulse shape discrimination techniques to discriminate between radioactive particles by analyzing the luminescence decay profile after a scintillation event.
This type of analysis requires a thorough understanding of the scintillator system's luminescence decay behavior.
This article explores the effect of PPO concentration on the luminescence decay curve of the LAB/PPO system using time-resolved X-ray excited luminescence spectroscopy using an Edinburgh Instruments FLS1000 Photoluminescence Spectrometer.
Experimental
The experiment involved dissolving PPO (Sigma Aldrich) in LAB (Cepsa Canada) at concentrations ranging from 1 to 20 g/L and pipetting it into quartz cuvettes.
The X-ray stimulated luminescence spectra and decays of the LAB/PPO solutions were measured with an Edinburgh Instruments FLS1000 Photoluminescence Spectrometer connected to the XS1 X-ray Sample Chamber (Figure 2).
The XS1 X-ray Sample Chamber is an X-ray shielded container that can accommodate several X-ray sources for the study of novel scintillator materials.
To obtain X-ray stimulated luminescence spectra of LAB/PPO, the XS1 was outfitted with a small 60 kV continuous-wave X-ray source. A 40 kV pulsed X-ray source, optically pumped by an Edinburgh Instruments HPL-450 pulsed diode laser, was used to ignite the LAB/PPO for luminescence decay measurements.
The X-ray stimulated luminescence from the scintillator is collected by a liquid light guide and sent to the FLS1000 for wavelength selection and detection. The FLS1000 was outfitted with a PMT-900 detector sensitive to 200-870 nm and time-correlated single-photon counting (TCSPC) circuits.
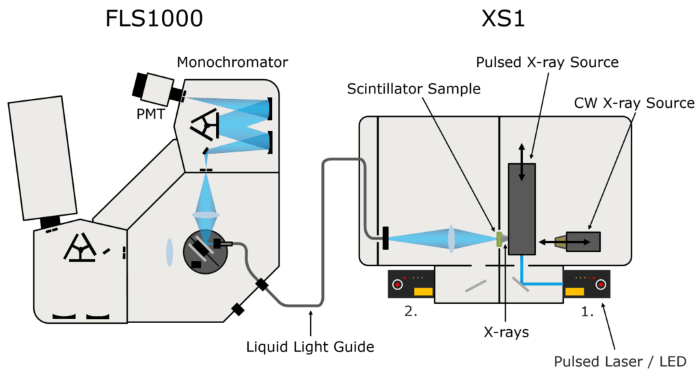
Figure 2. FLS1000 & XS1 X-ray excited luminescence spectroscopy setup. Image Credit: Edinburgh Instruments
Results and Discussion
Figure 3 shows the X-ray excited luminescence spectra of LAB/PPO obtained using a continuous wave X-ray source. The luminescence exhibits a peak at 365 nm and is entirely due to the PPO component, with no luminescent emission from the LAB.
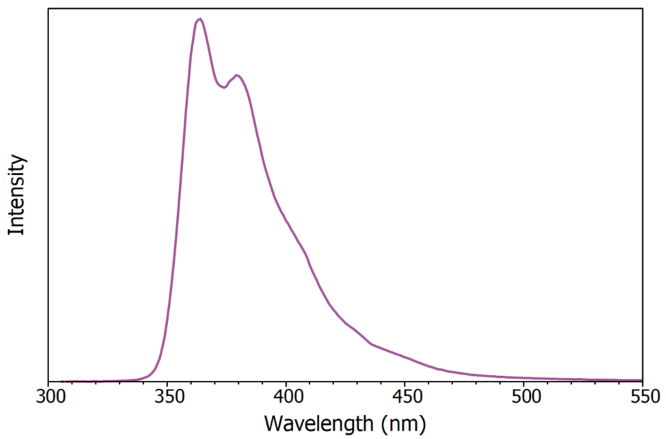
Figure 3. X-ray excited luminescence spectrum of LAB/PPO (8 g / L). Image Credit: Edinburgh Instruments
Next, the influence of PPO concentration on the luminescence decay curve of LAB/PPO was studied. Eight LAB/PPO solutions with various PPO concentrations were stimulated by a 40 kV pulsed X-ray source, and their decay was monitored using TCSPC (Figure 4).
The decay profiles are complicated, consisting of an early prompt component followed by a long tail. The prompt component can be attributed to the radiative de-excitation of PPO from its singlet excited state to the singlet ground state, which is a permitted transition and so occurs quickly.
In contrast, molecules excited to their triplet states cannot de-excite directly to the ground state and must undergo other de-excitation pathways, such as triplet-triplet annihilation, which leads to the long tail in luminescence decays.
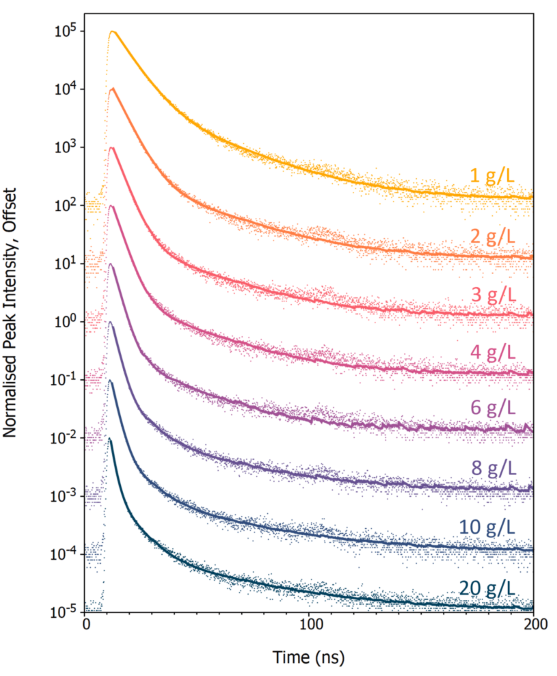
Figure 4. X-ray excited luminescence decays of LAB/PPO at varying PPO concentrations acquired using TCSPC. The scatter plots are the experimental TCSPC decays, and the solid lines are a two or three-exponential component best fit to each decay. Image Credit: Edinburgh Instruments
To identify the decay components, the luminescence decays were fitted with a two (1 - 6 g/L) or three (8 - 20 g/L) exponential decay model by reconvolution using Edinburgh Instruments FAST lifespan analysis software (solid lines in Figure 4).
Table 1 shows the lifetime and intensity fractions for each decay component. The quickest decay component (τ1), which corresponds to the radiative singlet de-excitation of PPO, is the dominating contribution to the overall luminescence for all PPO concentrations, with a luminescence percent ranging from 72-96 %.
Table 1. Decay times (τi) and decay component fractions (fi) for varying PPO concentrations in LAB after pulsed X-ray excitation. Source: Edinburgh Instruments
| PPO Conc.(g / L) |
τ1(ns) |
f1(%) |
τ2(ns) |
f2(%) |
τ3(ns) |
f3(%) |
| 1 |
7.1 |
85.8 |
26.6 |
14.2 |
|
|
| 2 |
5.4 |
90.9 |
28.3 |
9.1 |
|
|
| 3 |
4.3 |
94.1 |
27.2 |
4.3 |
|
|
| 4 |
3.7 |
94.2 |
26.5 |
5.8 |
|
|
| 6 |
3.0 |
96.2 |
22.9 |
3.8 |
|
|
| 8 |
2.5 |
89.5 |
9.3 |
7.1 |
36.9 |
3.4 |
| 10 |
2.3 |
79.9 |
8.8 |
13.0 |
39.7 |
7.1 |
| 20 |
1.7 |
71.8 |
7.5 |
19.9 |
38.5 |
8.3 |
Figure 5 presents the variation of τ1 with PPO concentration. The rate of non-radiative energy transfer from an excited LAB molecule determines the excitation of a PPO molecule (τ1).
As the concentration of PPO grows, the average separation between an excited LAB molecule and a nearby PPO molecule reduces, increasing the energy transfer rate and resulting in a drop in τ1 with concentration (Figure 5).
At high PPO concentrations, energy transfer is no longer the rate-limiting step, and τ1 approaches the intrinsic radiative lifetime of PPO in LAB, which is less than 2 ns. 6
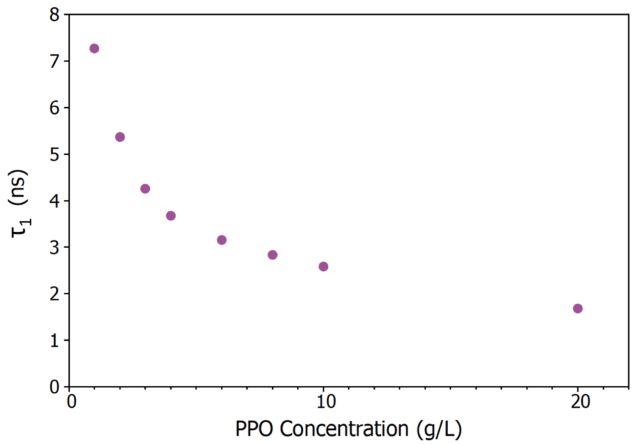
Figure 5. Variation of τ1 in LAB/PPO with PPO concentration. Image Credit: Edinburgh Instruments
Conclusion
The X-ray excited luminescence decay properties of LAB/PPO at different PPO concentrations were examined using an Edinburgh Instruments FLS1000 and the XS1 X-ray Sample Chamber Accessory.
The major decay component's lifespan decreased from 7.1 ns to 1.7 ns when the PPO concentration grew from 1 to 20 g/L.
The XS1 X-ray Sample Chamber Accessory expands the capabilities of the FLS1000 PL spectrometer into the X-ray range, resulting in a strong characterization tool for the development of new scintillator materials.
Acknowledgements
The authors are grateful to Dr. Sasmit Gokhale of Brookhaven National Laboratory for supplying the LAB and PPO in this article.
References
- An, F.P., et al. (2012). Observation of Electron-Antineutrino Disappearance at Daya Bay. Physical Review Letters, 108(17). DOI: 10.1103/physrevlett.108.171803. https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.108.171803.
- Ahn, J.K., et al. (2012). Observation of Reactor Electron Antineutrinos Disappearance in the RENO Experiment. Physical Review Letters, 108(19). DOI: 10.1103/physrevlett.108.191802. https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.108.191802.
- Andringa, S., et al. (2016). Current Status and Future Prospects of the SNO+ Experiment. escholarship. (online) DOI: 10.1155/2016/6194250. https://onlinelibrary.wiley.com/doi/10.1155/2016/6194250.
- An, F., et al. (2016). Neutrino physics with JUNO. 43(3), pp.030401–030401. DOI: 10.1088/0954-3899/43/3/030401. https://iopscience.iop.org/article/10.1088/0954-3899/43/3/030401.
- H.M. O’Keeffe, E. O’Sullivan and Chen, M.C. (2011). Scintillation decay time and pulse shape discrimination in oxygenated and deoxygenated solutions of linear alkylbenzene for the SNO+ experiment. Nuclear Instruments and Methods in Physics Research Section A Accelerators Spectrometers Detectors and Associated Equipment, 640(1), pp.119–122. DOI: 10.1016/j.nima.2011.03.027. https://www.sciencedirect.com/science/article/abs/pii/S0168900211005882.
- T. Marrodán Undagoitia, F. et al. (2009). Fluorescence decay-time constants in organic liquid scintillators. Review of Scientific Instruments, 80(4). DOI: 10.1063/1.3112609. https://pubs.aip.org/aip/rsi/article-abstract/80/4/043301/282226/Fluorescence-decay-time-constants-in-organic?redirectedFrom=fulltext.

This information has been sourced, reviewed, and adapted from materials provided by Edinburgh Instruments.
For more information on this source, please visit Edinburgh Instruments.