Obtained from chitin, chitosan is a naturally abundant polysaccharide found in insect exoskeletons, as well as in crab and shrimp shells. Chitosan is produced by chemical deacetylation of chitin, removing acetyl groups.
Its versatility sees chitosan used across a wide range of industries, including:
- Food, where chitosan is used to form protective films designed to slow microbial growth and extend product shelf life. Chitosan is also used as a thickener or emulsifier to improve stability and flow.
- Pharmaceuticals, where chitosan’s biodegradability and biocompatibility make it ideal for use in drug carriers, sustained-release systems, and wound dressings that feature antibacterial and hemostatic properties.
- Water treatment using chitosan as a flocculant for contaminant removal.
- Agriculture, where chitosan acts as a seed coating, improving disease resistance and germination.
Molecular weight is key to chitosan’s performance.
Low-molecular-weight chitosan dissolves easily, penetrates membranes more readily, produces low-viscosity solutions, and is often associated with stronger antitumor or antimicrobial activity.
High molecular weight chitosan, in contrast, forms stable gels and scaffolds for tissue engineering, creates viscous solutions, and offers a higher adsorption capacity for metals and organics, though at slower kinetics.
The accurate measurement of molecular weight is, therefore, essential for selecting the most appropriate chitosan grade for specific applications.
Experimental
The study presented here used an SEC system equipped with light-scattering (LS) and refractive index (RI) detectors.
The light-scattering detector fitted to Bettersize Instruments’ BeSEC LS2 includes 90 ° and 7 ° angles. The BeSEC workstation integrates light scattering with RI or UV signals, enabling the accurate calculation of molecular weight averages (Mn, Mw, and Mz) and distributions.
System Configuration
The system configuration employed was as follows:
- Detector: Light Scattering (LS) + Refractive Index (RI)
- Column: Shodex Ohpak LB-806M
- Mobile phase: Water with 0.2 M NaNO3
- Flow rate: 0.7 mL per minute
- Injection volume: 100 μL
- Column temperature: 40 °C
- dn/dc: 0.129 mL/g
Sample Preparation
A water-soluble chitosan sample was weighed before being dispersed in 0.2 M NaNO3. The solution was then stirred to clarity (2–5 mg/mL) and filtered through a 0.22 μm PES syringe filter before being transferred to vials. It is important to note that the high ionic strength mobile phase is key to minimizing electrostatic interactions between the column and the chitosan sample.
Results and Discussion
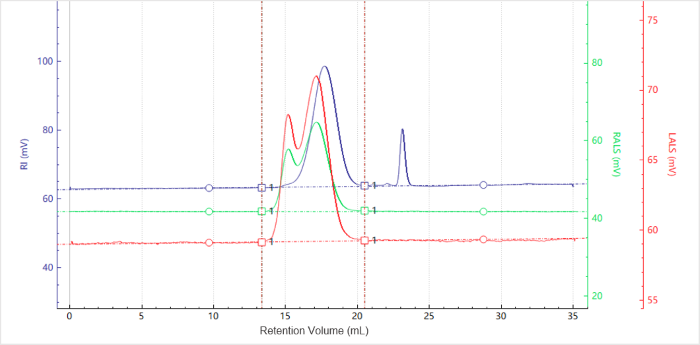
Figure 1. Elution profiles of the multi-detector signals for Chitosan. Image Credit: Bettersize Instruments.
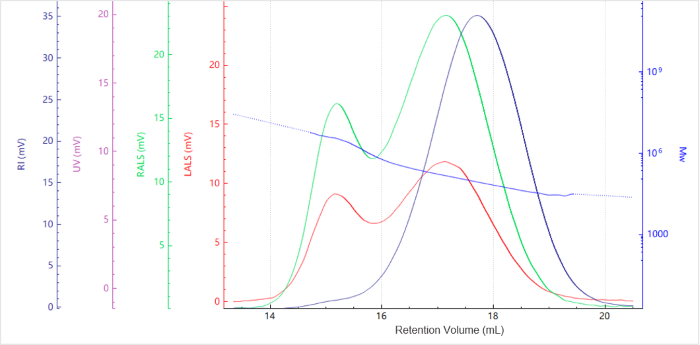
Figure 2. Elution profile of the molecular weight for Chitosan. Image Credit: Bettersize Instruments.
Figure 1 features chromatograms acquired via multiple detectors. These included:
- Refractive index (RI, blue)
- Right-angle light scattering (RALS, green)
- Row-angle light scattering (LALS, red)
Figures 1 and 2 confirm the collection of clean signals with flat baselines, strong signal-to-noise ratios, and minimal noise. The molecular weight profile decreased gradually with increasing elution volume. These findings are consistent with SEC principles, which state that larger species elute before smaller ones.
The light-scattering peaks show no tailing, while the molecular-weight curve remains stable, without an upturn at the end. This indicates that size-based separation was effective.
Table 1. Molecular weight results. Source: Bettersize Instruments.
| Sample |
Mn (Da) |
Mw (Da) |
Mz (Da) |
| Chitosan |
71,254 |
181,663 |
3,266,638 |
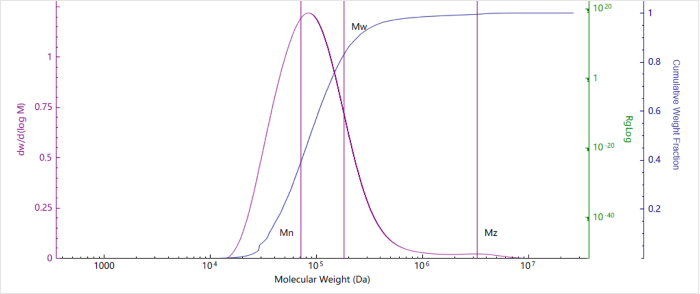
Figure 3. Molecular weight distribution of Chitosan. Image Credit: Bettersize Instruments.
Conclusion
This experiment confirms that the water-soluble chitosan sample analyzed exhibits a broad distribution and a relatively high molecular weight.
The presence of a small fraction of larger chains is confirmed by the tailing peak in the molecular weight profile. These findings provide a clear indication of sample heterogeneity, offering useful guidance on selecting an appropriate chitosan grade for processing or formulation.
Acknowledgments
Produced from materials originally authored by Zhibin Guo from Bettersize Instruments.

This information has been sourced, reviewed and adapted from materials provided by Bettersize Instruments.
For more information on this source, please visit Bettersize Instruments .