Driven by an increased demand for innovation, compliance, and high-performance solutions, the manufacturing sector continues to make significant advances. The 2023 Market Analysis Report valued the global industrial services market at $32.65 billion in 2023, with a projected compound annual growth rate (CAGR) of 6.5 % from 2024 to 2030.
These projections, in line with current growth, mean manufacturers are under mounting pressure to implement state-of-the-art containment technologies that meet ever-changing regulatory expectations while mitigating cross-contamination risks, improving operational efficiency, and protecting worker safety.
Hosokawa Micron Ltd is committed to delivering the next-generation of containment systems. This means going above and beyond standard exposure control strategies to ensure complete operator protection and protect environmental safety.
Hosokawa Micron Ltd.’s engineered solutions have been developed to ensure the following:
- Control hazardous substances and contain airborne particles
- Protect operators from toxic materials and potent compounds
- Safeguard product integrity throughout the lifecycle of the manufacturing process
This guide offers an examination of the entire containment journey: from laboratory-scale development to all-out production and fulfilment. This highlights how Hosokawa’s technologies facilitate compliance, worker and product safety, as well as performance across a range of industrial applications.

Section 1: Containment – Definition and Importance in Powder Handling
1.1 What is Material Containment?
Material containment involves the controlled isolation of hazardous, potent, or sensitive substances during processing. This is achieved by using specifically engineered systems such as isolators, gloveboxes, and containment downflow booths.
These systems have been developed to ensure the following:
- Operator protection from exposure to active pharmaceutical ingredients (APIs), toxic chemicals, or other biological agents.
- Preventing cross-contamination and maintaining aseptic conditions to ensure product integrity.
- Implementing environmental safeguards to ensure there is no release of airborne particulates or volatile compounds.
Containment is a central manufacturing component across the pharmaceutical and chemical industries, where tight monitoring of the processing environment is crucial for meeting the standards set out by Good Manufacturing Practice (GMP) and Occupational Exposure Limits (OELs).
Tailor-made containment solutions are crucial in industries such as pharmaceuticals, chemicals, petrochemicals, nuclear processing, and FMCG. This is due to the high-risk nature of the materials being handled.
In other areas, such as biotechnology, agriculture, and research laboratories, there is a reliance on containment systems to maintain cleanroom integrity and protect operator safety. These systems act as sealed, verified enclosures, designed to eliminate all opportunities for leaks, exposure, or contamination across a product’s lifespan: from the laboratory to full-scale production.
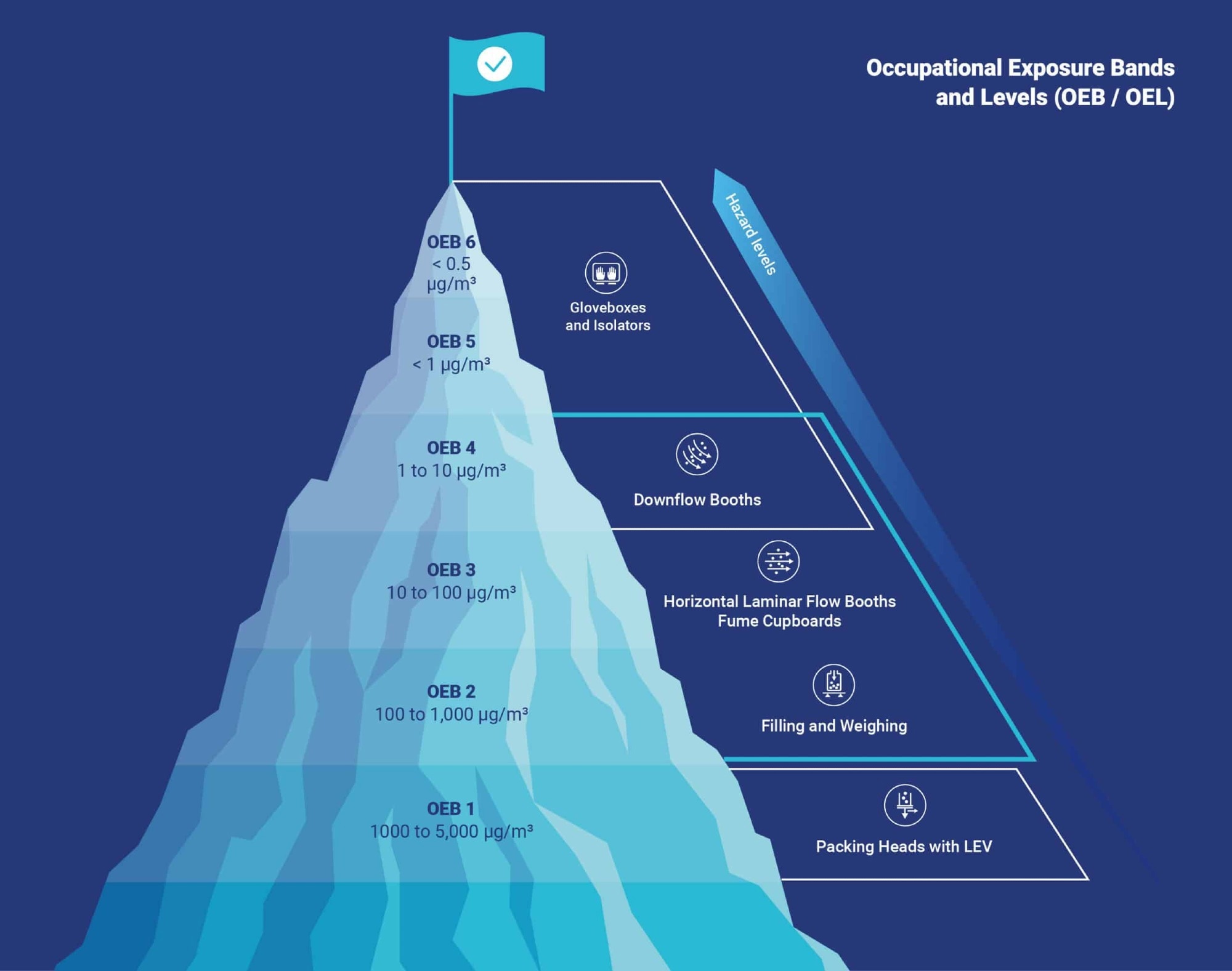
Image Credit: Hosokawa Micron Ltd
1.2 Why Does Containment Matter?
Containment is vital as it helps ensure the following:
- By preventing operator exposure to hazardous substances, it ensures safety and reduces health risks to meet compliance standards in line with occupational safety regulations.
- Maintains good product quality as sterility prevents contamination, safeguarding the efficacy and safety of pharmaceutical products.
- Supports adherence to global standards, guidelines, and regulations such as EU GMP Annex 1, FDA 21 CFR, and ISO 14644, which meet compliance and help manufacturers avoid costly penalties and recalls.
- Minimizes downtime due to contamination events, optimizes workflow, and supports lean manufacturing principles to boost operational efficiency.
As the manufacturing industry responds to an increasing demand for innovative and reliable solutions in line with an evolving industry, containment systems must also be advanced.
With values of $32.65 billion in 2023 and a projected CAGR of 6.5 % from 2024 to 2030, there is a pressing need for adaptive containment technologies that adhere to more stringent regulatory guidelines while supporting high-performance manufacturing environments.
Hosokawa is delivering the next-generation of containment solutions as it moves from exposure control to ensuring total operator protection and environmental safety.
Hosokawa’s systems have been developed to control particles, ensure operator safety, and preserve product integrity across all stages of the manufacturing process.

Image Credit: Hosokawa Micron Ltd
Section 2: Historical and Technological Evolution of Containment in Powder Handling
2.1 Key Historical Milestones in Containment Development
The progress made in containment technologies across the pharmaceutical manufacturing sector is closely related to advancements made in GMP, aseptic processing, and occupational health standards.
1950s–1970s: Origins of Passive Containment Systems
As developments in sterile drug production spurred expansion, the industry introduced open Restricted Access Barrier Systems (RABS) to limit microbial ingress from operator intervention. These passive systems were the first mark of controlled environments in cleanroom operations.
1970s: Biosafety and Risk-Based Containment
Advancements in molecular biology and recombinant DNA technologies demanded a structured approach to biosafety. The Asilomar Conference set the stage for risk-based containment strategies, coordinating containment levels with the hazard classification of biological agents.
1990s: Closed RABS and Isolator Technologies
The pharmaceutical sector adopted closed RABS with independent HVAC systems, enabling precise control of key manufacturing conditions, including temperature, humidity, and differential pressure. Simultaneously, isolator systems became a prominent feature, ensuring Grade A environments through physical separation and HEPA filtration. These developments reduced operator-product interaction and mitigated contamination risk.
2.2 Integration of Real-Time Monitoring
Modern containment systems typically include real-time monitoring systems to guarantee continuous compliance with GMP standards and occupational exposure limits (OELS).
Hosokawa offers carefully engineered solutions that proactively detect filter degradation in Downflow Containment Booths and Isolators, sounding alerts before exceeding performance thresholds.
These systems help facilitate preventative maintenance while ensuring operator safety, complying with EMA and FDA standards for containment control strategies (CCS).
2.3 Industrial Drivers of Containment Innovation
Starting off as a response to concerns surrounding industrial hygiene, containment technologies were introduced during the early phases of manufacturing in the pharmaceutical and chemical industries.
Initially, these solutions were comprised of basic enclosures which then became specialized glove boxes, fume hoods, and high-containment isolators designed to control potent active pharmaceutical ingredients (APIs), and other toxic materials.
Today, containment is a fundamental component of high-potency manufacturing, with systems developed to meet the rigor of OEB (Occupational Exposure Branding) and OEL standards.
Today’s innovations are crucial when it comes to ensuring operator safety, product integrity, and meeting regulatory compliance in both sterile and non-sterile manufacturing environments.
However, this only covers the basics of high-containment engineering, from OEB classification and airflow control to regulatory compliance and operator protection.
In the full Complete Containment. Engineered Protection guide, you’ll discover how these principles are put into practice across a range of real-world industrial solutions in high-risk sectors.
The guide includes the following:
- Application examples across FMCG, pharmaceuticals, chemicals, nuclear, and battery materials
- Comprehensive detailing of containment equipment, including isolators, downflow booths, gloveboxes, and nuclear systems
- Insights into how to safely handle radioactive, toxic, and ultra-fine materials
- A careful insight into what to consider when selecting a containment partner
- The engineering, compliance, and validation standards behind high-performance containment systems
- A step-by-step breakdown of Hosokawa’s implementation process, from OEB definition to global installation and aftercare
The guide also covers how advanced containment solutions merge with upstream and downstream processes, while mitigating contamination risk and supporting long-term operational efficiency.
Whether you're considering new containment systems or upgrading existing infrastructure, this guide provides a clear roadmap from risk assessment to validated performance.
Download your copy to see how engineered containment solutions are setting new benchmarks in safety, compliance, and industrial performance.
Download the full guide here


This information has been sourced, reviewed and adapted from materials provided by Hosokawa Micron Ltd.
For more information on this source, please visit Hosokawa Micron Ltd.