Inspecting foods for foreign materials is an essential component of food companies’ quality assurance systems. Metal detectors and X-ray scanners can identify microscopic particles embedded in food products; once found, it is crucial to accurately identify the material composition of these foreign bodies.
The root cause of any contamination must be identified and eradicated before it spreads further downstream throughout the processing lines, potentially leading to product recalls.
Examples of debris include clear plastics used to transport food items; other polymers and plastics found in the manufacturing plant (e.g., gaskets); glass and/or silicates; various metals used in process equipment; and materials originating outside the manufacturing facility.
Visually examining the found debris initiates the process of identifying unknown particles. Spectroscopic techniques using infrared (IR) and X-ray fluorescence (XRF) technologies can rapidly and accurately identify unknown particles.
As a result, these procedures aid in discovering the underlying source of contamination, reducing the need for massive recalls and reducing costs for food makers. Most importantly, identifying the root cause helps ensure safer food for the final consumer.
This article presents Fourier transform infrared (FTIR) spectrometry and handheld X-ray fluorescence (HHXRF) spectrometry as complementary techniques for rapid identification of various types of foreign materials in food, including organics/plastics, rubber, ceramics, glass, and metals.
The Thermo Scientific™ Nicolet™ Summit™ X FTIR Spectrometer and Thermo Scientific™ Niton™ XL5 Plus Handheld XRF Analyzer were used for the FTIR and handheld XRF analyses, respectively (Figure 1).

Figure 1. (left) Nicolet Summit X FTIR Spectrometer with a Thermo Scientific™ Everest™ diamond attenuated total reflectance (ATR) accessory; and (right) Niton XL5 Plus Handheld XRF Analyzer. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
The Nicolet Summit X spectrometer, alongside the Everest diamond ATR setup, is a powerful FTIR system that can be installed on the factory floor, expediting data acquisition. ATR is the most popular form of data acquisition for material identification using the Nicolet Summit X spectrometer: no sample preparation is required.
A sample is simply placed on top of an infrared inactive crystal, usually a diamond, and a pressure tower is used to engage the sample with the crystal. The pressure tower has a slip clutch that limits the amount of torque the crystal may receive, preventing damage to the ATR attachment.
Due to the hardness and chemical inertness of diamond, hard, sharp, or abrasive samples (e.g., glass), as well as acidic or basic samples (in the full pH range of 1-14), can be placed on the sampling region without causing damage to the diamond crystal.
Once a sample is placed on the crystal, data collection can commence, resulting in an infrared spectrum of the unknown component.
Data acquisition of a single spectrum typically takes about 20 seconds, and the spectrum can then be compared to a database of commercial libraries of known components for material identification.
Figure 2 shows an example of a clear unknown plastic sample studied on the Nicolet Summit X spectrometer, which appears to be polyethylene-based. Users can also create their own spectrum libraries using known material from their production or work settings, which can then be searched to identify compounds and pollutants.
Identifying the sample material indicates where foreign debris may be coming from (e.g., within the plant or from an external source), allowing the user to target the debris source and prevent future contamination.
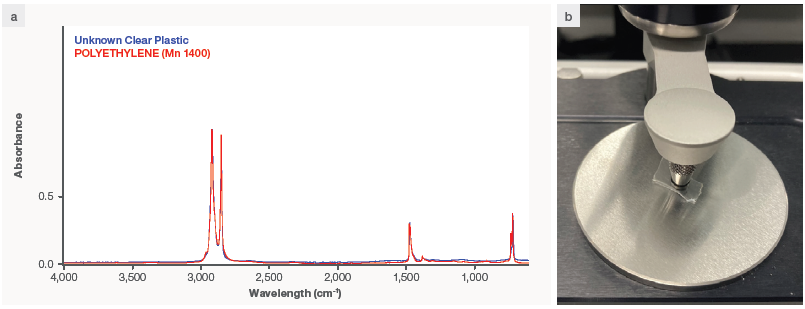
Figure 2. (a) Spectrum in Thermo Scientific™ OMNIC™ Paradigm software of unknown transparent plastic, overlayed with results of a library search, which generates a good match for polyethylene. (b) Example of a transparent plastic loaded onto the diamond ATR crystal for data acquisition. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
In another experiment, two materials, one rubber and one metal, were examined using both the Nicolet Summit X FTIR spectrometer and the Niton XL5 Plus HHXRF analyzer; the results demonstrate the complementary nature of the techniques (Figure 3).
The FTIR spectrometer produced a workable spectrum for the unknown rubber sample (Figure 3a, blue curve), which the program used to identify it as polydimethylsiloxane (PDMS), a silicone-based rubber (Figure 3a, red curve).
The FTIR spectrum of the stainless steel sample, on the other hand, showed no peaks (Figure 3b) and was flat, since metals are IR-passive.
The Niton XL5 Plus analyzer is well-suited for identifying inorganic samples, particularly metals and alloys like stainless steel 316 (Figure 3d). While the XRF analyzer cannot recognize the material as silicone, it may detect elements like silicon (Figure 3c) and additional metals in the sample.
FTIR and XRF are proven to be complementary techniques in the following example. If the unknown material was soda lime or borosilicate glass, the FTIR would produce a broad peak in the lower wavenumber region, making it difficult to identify; the XRF would be able to distinguish between those glasses based on silicon and calcium elemental compositions.
The FTIR spectrometer can confirm organic compounds, but cannot distinguish between different metal alloys. However, the handheld XRF analyzer can detect metal alloys, glasses, and minerals, but provides less information on polymer/rubber materials. Thus, HHXRF and FTIR measurements work together to identify both categories of unknown compounds.
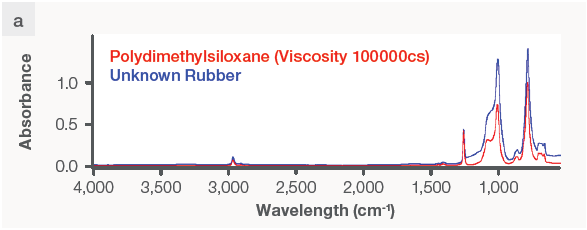
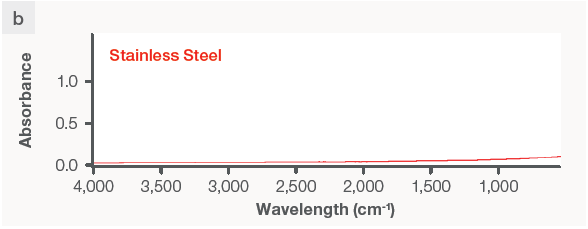
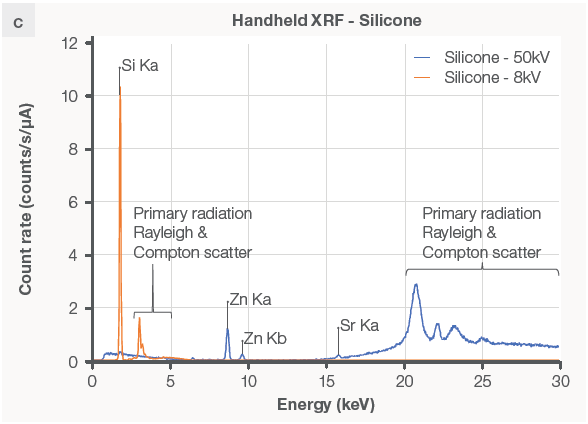
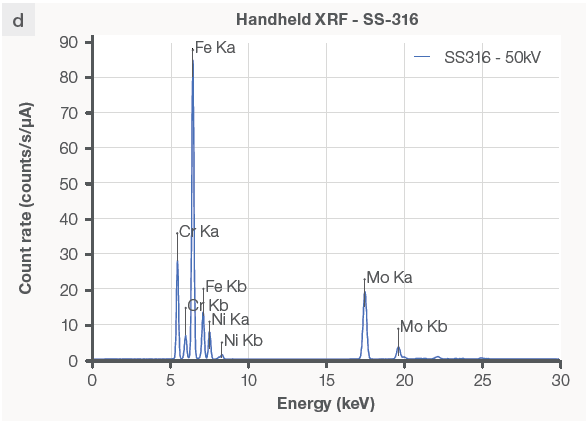
Figure 3. FTIR and XRF data on the same plastic and metal samples. The FTIR instrument can produce a reading on (a) silicone rubber but not on (b) stainless steel. The XRF analyzer detects (c) silicone and zinc in the rubber sample without being able to identify the type of rubber, but it does identify (d) the metal sample as stainless steel 316 grade. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
The Niton XL5 Plus is a compact, lightweight (approximately 3 pounds) handheld XRF analyzer that can be used directly on the manufacturing floor or elsewhere in the facility.
Modern HHXRF analyzers can identify and quantify elements with atomic numbers ranging from 12 (magnesium) to 92 (uranium) in a variety of materials, including metals, metal alloys, ceramics, glass, minerals, and polymers.
With the elements quantified, HHXRF can identify substances by comparing the composition of the measured sample to tabulated values of substances.
This method, known as identification via grade table, is used to identify metals by comparing the measured composition to tabulated values for over 500 standardized alloys, including steel or stainless steel, as well as alloys of aluminum, copper, nickel, cobalt, titanium, tin, lead, or tungsten.
Another method for identifying chemicals using HHXRF compares the spectra of an unknown substance with those of known substances. This kind of identification is known as spectral fingerprinting.
HHXRF studies are typically conducted in seconds for metal and alloy identification, and a few minutes for trace analysis. To examine large specimens, the user can either aim the analyzer and depress the trigger, or employ a test stand (Figure 4a).
The analysis is completely non-destructive, and the results in mass concentrations are displayed in real time on the analyzer’s LCD screen (Figure 4b).
While handheld XRF can evaluate a wide range of foreign bodies found in foods, including glass, ceramics, stones, bones, and plastics, it is particularly effective for measuring metal concentrations and identifying alloy grades.
The workflow for foreign body identification (FBI) with HHXRF normally includes mapping and obtaining data from all parts of the processing line that could contaminate goods, beginning with metal parts and progressing to ceramics and/ or glass parts. The user can compare the composition of foreign debris found in food products to that of potential or suspected sources of contamination.
This method is effective when different alloy grades are employed in a processing line, allowing the user to pinpoint contamination within the plant or rule out an internal source of contamination.
For example, material from a sieve mesh made of stainless steel 316 is easily distinguished from debris deriving from a frozen food block flaker blade made of stainless steel grade 430.

Figure 4. (a) The Niton XL5 Plus Handheld XRF Analyzer with a sample loaded in the test stand; (b) Live readout of stainless steel 316 and its elemental composition on the Niton XL5 Plus Analyzer. Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
When the same type of material, such as stainless steel 316, is used in several locations in the production line, it can be difficult to identify foreign entities produced from this material based on its composition or grade, as it may not be specific enough to pinpoint the source of contamination.
In such cases, a different strategy based on spectral fingerprinting is preferred. The user creates a library of spectra from measurements of the metallic and non-metallic materials obtained throughout the mapping process. Once the user has created the custom library, the spectral fingerprints of contaminating samples can be compared to the library entries or spectra of the suspected source to help locate and treat the contamination source.
If there is no match, it is possible that the pollutants were present in the raw materials, meaning that supplies should be rigorously inspected before entering the manufacturing process.
In one case, a single piece of glass was identified in a processed food product, and it was effective to use bespoke libraries, designed to help identify contamination sources. The discovery triggered an investigation to determine the source of pollution, and the production line was swiftly eliminated as a possible cause.
A subsequent careful analysis of the vegetable raw materials revealed the existence of further particles of glass debris. Figure 5 shows that one of these fragments (shown by the grey curve) had a spectral fingerprint that matched the foreign body identified in the processed food product (represented by the blue curve).
However, the spectra of the second glass fragment (orange curve) deviated significantly from that of the foreign body. This shows that the contamination in the vegetable raw materials likely came from several sources.
In addition to identifying contaminants, HHXRF allows food processing plant maintenance teams to quickly verify specifications for critical materials used in processing equipment such as auger conveyors, chopping blades, frozen food block flaker blades, mixing paddles, and so on, preventing non-compliant equipment failure.
A good example of this is the usage of grade 304 stainless steel in a sieve mesh, rather than grade 316. Grade 304 has a lesser corrosion resistance and, over time, increases the danger of failure and contamination of salt-containing foods such as sauces or soups.
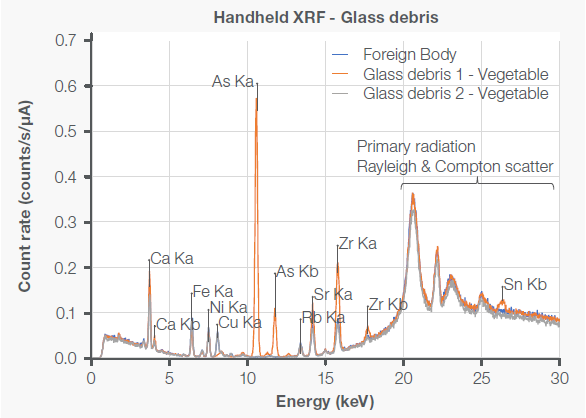
Figure 5. XRF spectra of a foreign body found in processed food and two pieces of glass debris subsequently detected in the vegetable used as raw materials. The foreign body (blue curve) has a similar spectral fingerprint to glass debris 2 (grey curve) but presents a noticeably different spectrum than glass debris 1 (orange curve) when looking at trace metals such as copper (Cu), arsenic (As), strontium (Sr), zirconium (Zr) or tin (Sn). Image Credit: Thermo Fisher Scientific – Handheld Elemental & Radiation Detection
Conclusion
FTIR spectrometry and handheld XRF spectrometry are quick and easy-to-use analytical tools that add significant value to food processing industries.
In enhanced quality assurance programs, metal detectors and X-ray inspection devices detect foreign materials, and these analytical approaches maximize the user’s capacity to identify the source of foreign body materials, allowing corrective steps to be applied quickly.
FTIR is well-suited to distinguishing between various types of organic and polymer-based materials, whereas handheld XRF is a useful analytical tool for analyzing inorganic materials such as metal, glass, and ceramics.
In addition to the FBI, both procedures can supply food manufacturers with evidence to back up claims of quality or refute false allegations of contamination.

This information has been sourced, reviewed, and adapted from materials provided by Thermo Fisher Scientific – Handheld Elemental & Radiation Detection.
For more information on this source, please visit Thermo Fisher Scientific – Handheld Elemental & Radiation Detection.