There are four main different types of static light scattering (SLS) devices available: Right-Angle Light Scattering (RALS), Low-Angle Light Scattering (LALS), Hybrid RALS/LALS, and Multi-Angle Light Scattering (MALS). This article covers these instruments in detail.
RALS System
The simplest light scattering instrument is a RALS device, which measures the scattering light intensity at 90° to the incident beam, as illustrated in Figure 1A. From the measured intensity and the sample concentration, the sample’s molecular weight can directly be calculated. In the Zimm plot shown in Figure 1B, the scattering light intensity is measured far from the y-axis.
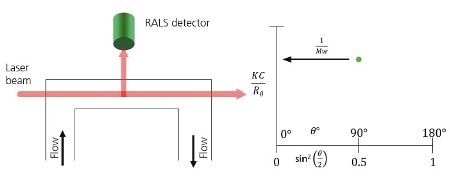
Figure 1. A. Schematic of a RALS detector showing the flow passing through the flow cell. Light from the laser enters at the end of the cell and is collected at 90°. B. When using RALS, the Debye plot is reduced to a single point which is assumed to be equal at every angle and therefore equal 1/Mw.
Advantages and Limitations of a RALS System
The following are some of the advantages of a RALS system:
- It is a simple instrument and does not involve any complicated optics. It features a flow cell with a window on the side, which enables the cell to have a low volume.
- As light is collected at 90°, any noise or flare generated by the change in refractive index is reduced.
- This low noise means the instrument has optimum sensitivity and a good signal-to-noise ratio
Some limitations of a RALS system include:
- The instrument makes measurements based on the assumption that the scattering light intensity is the same at 90° as it is as 0°. This is actually not true for any molecule that is of a size to exhibit any form of appreciable angular dependence in the light. Hence, a RALS detector cannot make accurate molecular weight measurements for molecules with an Rg > ≈15 nm.
- Since the measurement is being performed at one angle, it is not possible to make Rg calculations as the slope of the line on the Zimm plot is not measurable.
LALS Detector
A LALS device measures the scattering light intensity at an angle as close as possible to 0°, as illustrated in Figure 2A. This means that the estimated molecular weight value will be very near to the actual molecular weight of the molecule. From the Zimm plot shown in Figure 2B, a LALS measurement is very near to the axis, and eventually very near to the y-intercept, which means that the error is minimal by measuring at 7°.
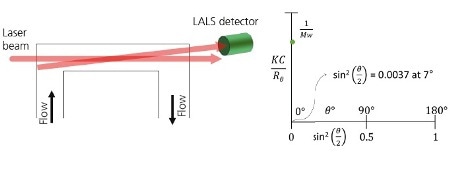
Figure 2. Schematic of a LALS detector showing the flow passing through the flow cell. Light from the laser enters at the end of the cell and is collected through the same exit window at a low angle e.g. 7°. B. When using LALS, the Debye plot is reduced to a single point, which is very close to the y-axis and therefore equal 1/Mw for all molecules.
Advantages and Limitations of a LALS Detector
The some advantages of a LALS detector include:
- Very accurate molecular weight measurement, as LALS measures the scattering light intensity very near to the axis of the Zimm plot.
- A dedicated LALS detector can have a tiny flow cell because it only needs to accommodate one measurement angle.
- The LALS principle applies to the molecular weight measurement of any molecule.
The following are the limitations of a LALS detector:
- As it measures the scattered light at a single angle, a LALS detector is not able measure Rg.
- The sensitivity of LALS detectors is inferior to that of RALS detectors, making them ineffective to measure weakly scattering samples
- It is a challenge to construct a LALS detector because the scattered light and the laser light are very near to each other so it is necessary to measure the scattered light without collecting the incident laser light as well.
- LALS detectors are susceptible to contamination, as contaminating particles are typically large, thus scattering predominantly in the forward direction.
However, modern dedicated LALS devices can isolate the scattered light from the incident light, thus reducing the noise and exhibiting superior sensitivity.
RALS/LALS Hybrid Detectors
RALS instruments can measure tiny isotropic scatterers and LALS detectors can provide accurate measurements of the molecular weight of large molecules. A RALS/LALS hybrid integrates these two measurements into a single cell, as illustrated in Figure 3A.
On the Zimm plot shown in Figure 3B, the RALS device is utilized to measure molecular sizes below the ∼15 nm threshold of isotropic scattering, while the LALS system is utilized to measure molecular sizes above the ∼15 nm threshold of isotropic scattering. Software automatically switches from LALS to RALS based on the detector signals.
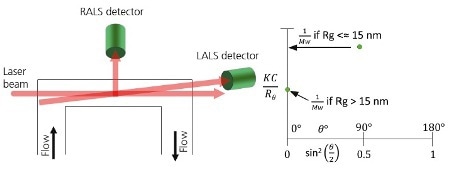
Figure 3. A. Schematic of a RALS/LALS hybrid detector showing the flow passing through the flow cell. Light from the laser enters at the end of the cell and is collected at 90° and through the same exit window at a low angle e.g. 7°. B. When using RALS/LALS, the Debye plot is reduced to two points where the RALS value is used to maximize sensitivity for isotropic scatterers and the LALS value is used for the most accurate molecular weight for anisotropic scatterers.
Advantages and Limitations of RALS/LALS Hybrid Detectors
The following are the advantages of RALS/LALS hybrid detectors:
- The RALS detector provides the superior sensitivity for smaller molecules.
- The LALS detector provides the superior sensitivity for larger molecules.
- Enables simultaneous comparison of two angles in software to obtain the best data at every point on the chromatogram.
- A RALS/LALS hybrid needs only a very small flow cell.
- The combination of the data from two angles makes the slope of the line on the Zimm plot measurable, thus providing a good estimate of Rg.
The following are the limitations of RALS/LALS hybrid detectors:
- The Rg calculation has a limited accuracy
- LALS data can be noisy when the GPC/SEC system is not clean
MALS Detector
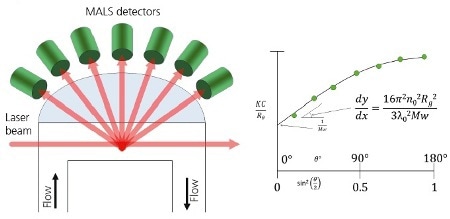
Figure 4. A. Schematic of a MALS detector showing the flow passing through the flow cell. Light from the laser enters at the end of the cell and scattered light exits at different angles. The scattered light is collected by multiple detectors. B. When using MALS, the Debye plot is completed and extrapolated back to 0°. The molecular weight is calculated from the intercept and Rg is calculated from the initial slop of the line.
A MALS detector measures the scattering light intensity at many angles, as depicted in Figure 4A. When these points are plotted on a Zimm plot, as shown in Figure 4B, it is possible to extrapolate a best fit line back to 0° from where the molecular weight can be estimated. From the initial slope of this line, the molecular size, Rg can be accurately calculated.
Advantages and Limitations of MALS Detectors
The following are the advantages of MALS Detectors:
- Possibility of comparing results from any given angle to its neighboring angles
- Possibility of measuring the molecular weight of all molecules from small to large
- Possibility of accurate Rg measurement thanks to measurements at many angles
- Analyzing the shape of the line in the Zimm plot provides insights into the angular dependence of the scattered light.
- Possibility of removing any angle that fails or includes too much noise without seriously affecting the result
The following are the limitations of MALS systems:
- It is a challenge to select the appropriate extrapolation fit and model that yields the correct answer as no data is available about the shape and structure of the molecule.
- Large volume is necessary for MALS flow cells to accommodate more angles, thereby causing increased peak broadening.
- Measurement accuracy is inferior because lower angles are often noisier when compared to a RALS or LALS measurement due to the complexity of the cell.
Conclusion
LALS is the ideal method to make accurate molecular weight measurements of large molecules of Rg ≥10-15 nm radius, as it reduces the effect of the scattered light’s angular dependence and eliminates the requirement for extrapolation.
MALS provides key information into the angular dependence of the scattered light for large molecules of Rg >10-15 nm radius, thus allowing for the more accurate Rg measurement than a RALS/LALS hybrid. Rg can be utilized for characterizing the structure for these large molecules.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information on this source, please visit Malvern Panalytical.