Non-steroidal antiinflammatory drugs (NSAIDs) are generally used for reducing fever and pain caused by inflammation. Ibuprofen drug belongs to this category. Figure 1 shows the 1H NMR spectrum of 200 mM ibuprofen in CDCl3. This spectrum was documented in just a single scan which took only 7 seconds to obtain. Both 1H-1H couplings and peaks are suitably resolved and hence could be assigned to the molecular structure.
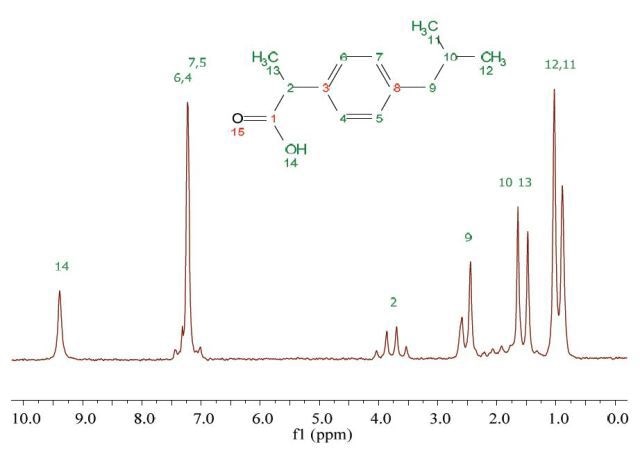
Figure 1. Proton NMR spectrum of 200 mM ibuprofen in CDCl3.
1H NMR Relaxation
Figures 2, 3 and 4 show the relaxation time measurements. Both T1 and T2 values of the exchanging hydroxyl proton can be seen in the spectrum. The first data point amplitude increases with the number of protons for the related peak.
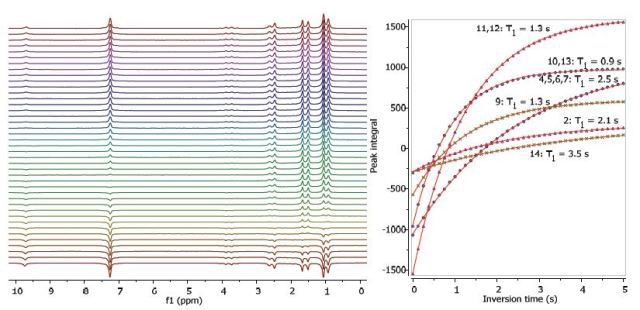
Figure 2. Proton T1 relaxation time measurement of 200 mM ibuprofen in CDCl3.
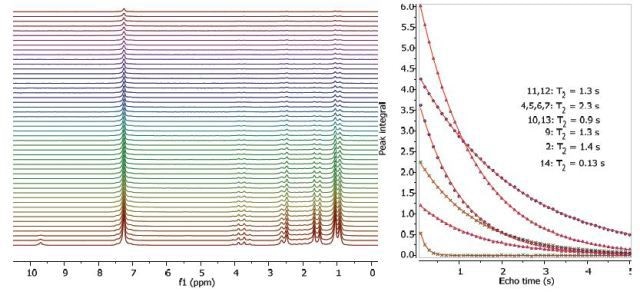
Figure 3. Proton T2 relaxation time measurement of 200 mM ibuprofen in CDCl3.
2D COSY
Figure 4 shows the 2D COSY spectrum where two spin systems (9,10,11,12) and (2,13) can be clearly seen. For instance, the methyl groups at 11 and 12 positions bond to CH2 and CH groups at 10 and 9 positions, while the methyl group at the 13 position only bonds to the CH group at the 2 position. No coupling occurs at positions (9,10,11,12) to either 13 or 2.
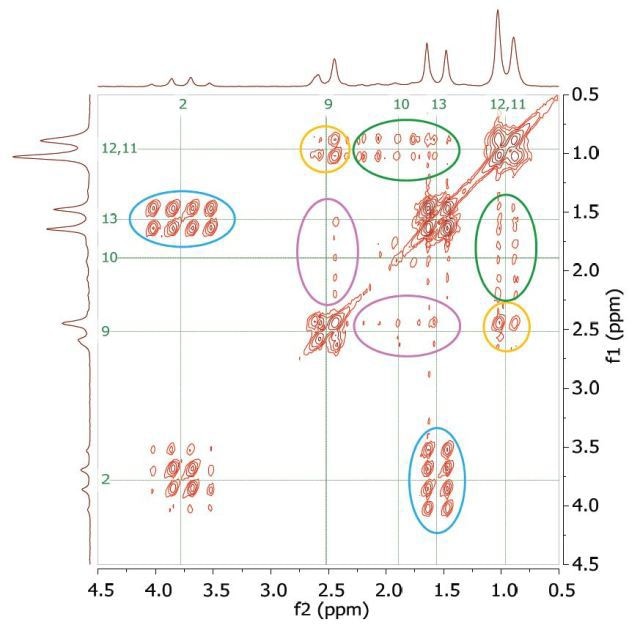
Figure 4. COSY spectrum of 200 mM ibuprofen in CDCl3. The cross-peaks and corresponding exchanging protons are labeled by colour-coded arrows and ellipses.
2D Homonuclear J-Resolved Spectroscopy
The chemical shift in the 2D homonuclear j-resolved spectrum appears along the f2 or direct dimension, and the effects of coupling between protons appear along the f1 or indirect dimension. This enables the entire assignment of chemical shifts of multiplets, and thus can help in measuring unresolved couplings. Also, a decoupled 1D proton spectrum is produced by the projection along the f1 dimension. The 2D homonuclear j-resolved spectrum of 200 mM ibuprofen in CDCl3, plus the 1D proton spectrum (blue line) are shown in Figure 5.
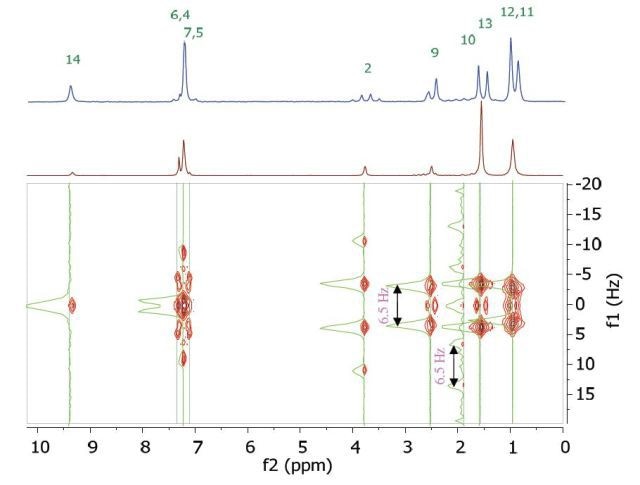
Figure 5. Homonuclear j-resolved spectrum of 200 mM ibuprofen in CDCl3. The multiplet splitting frequencies for different couplings are colour-coded as in Figure 4.
The projection which is vertical reveals how the overlapping multiplets disintegrate into a single peak and this makes the 1D spectrum more simplified. Peak multiplicities are produced by vertical traces from peaks in the 2D spectrum, as indicated by green lines in Figure 5, and help in determining the frequencies of proton-proton coupling. When coupling frequencies are compared between different peaks, information can be obtained regarding which peaks are bonded to each other. These couplings substantiate the results of the COSY experiment.
However, in this experiment, the effects of second order coupling appear as additional peaks in the f1 dimension that are equidistant from the coupling partners well detached from the zero frequency in the f1 direction. These peaks provide direct proof of second order coupling partners, but are generally considered as artifacts. Figure 6 shows these coupling partners and additional peaks are marked by arrows and ellipses that are colour-coded.
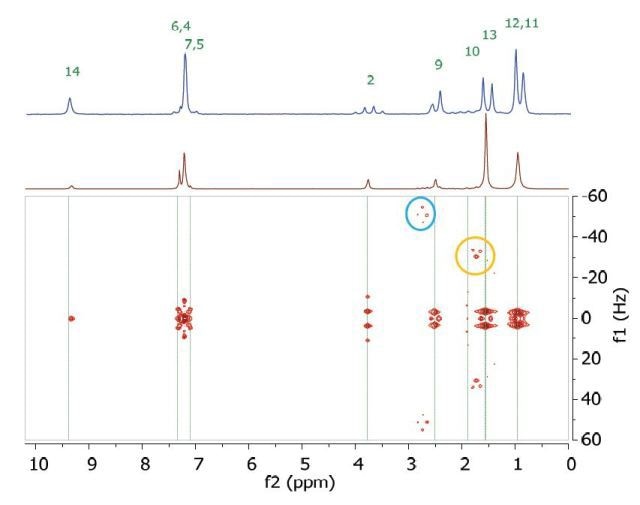
Figure 6. Homonuclear j-resolved spectrum of 200 mM ibuprofen in CDCl3 showing the extra peaks due to strong couplings.
1D 13C Spectra
Figure 7 shows the 13C NMR spectra of 2 M Ibuprofen in CDCl3. Since the 1D Carbon experiment is highly susceptible to the 13C nuclei in the specimen, 9 resonances are clearly resolved. The 13C DEPT method utilizes polarisation transfer between carbon and proton nuclei. In this experiment, only carbons coupled to protons are seen.
Given the fact that the DEPT spectra do not display the peaks at 137, 140 and 181ppm, they are assumed to be part of quaternary carbons. The DEPT-135 and the DEPT-45 experiments render signals of CH3, CH2 and CH groups, while the DEPT-90 experiment renders only the signal of CH groups. However, in the DEPT-135 experiment, the CH2 groups occur as negative peaks. Integration of the three DEPT spectra yields only the subspectra of the CH, CH2 and CH3 groups. In these subspectra, the peak at 45ppm includes CH2 and CH resonances with similar chemical shifts.
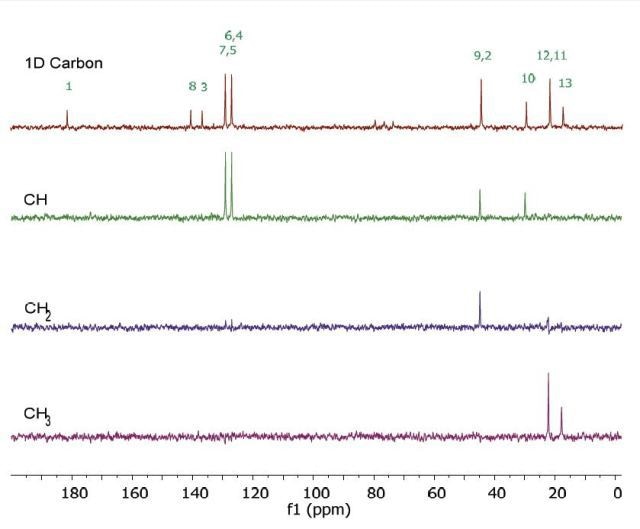
Figure 7. Carbon spectra of 2 M ibuprofen in CDCl3.
Heteronuclear Correlation
A range of Heteronuclear Correlation (HETCOR) NMR experiments were developed to identify coupling partners of varied nuclei. The HETCOR experiment helps in detecting the proton signal that appears along the f1 dimension and the carbon signal along the f2 dimension. Figure 8 shows the HETCOR spectrum of 2 M ibuprofen in CDCl3. in the 2D spectrum, the peaks reveal which proton is attached to which carbon. This experiment helps in resolving assignment uncertainty from the ID carbon spectra.
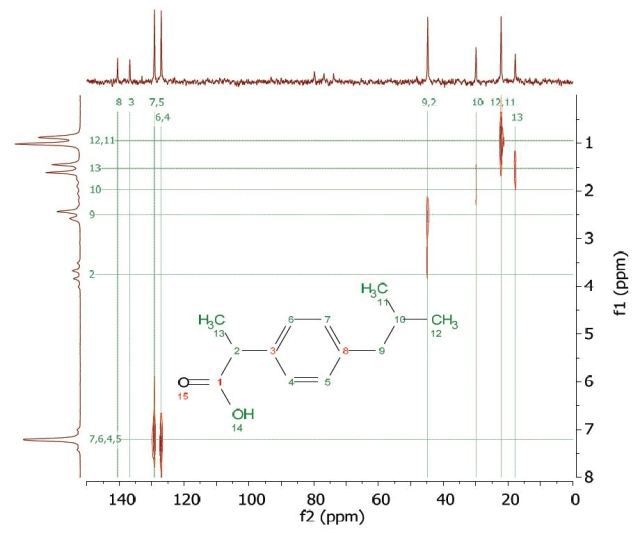
Figure 8. HETCOR spectrum of 2 M ibuprofen in CDCl3.
Heteronuclear Multiple Quantum Coherence
Heteronuclear Multiple Quantum Coherence (HMQC) is similar to the HETCOR experiment and is utilized to associate proton resonances to the carbons that are coupled directly to those protons. But, in the HMQC experiment, the proton signal appears along the f2 dimension and the carbon signal along the f1 dimension. Figure 9 shows the HMQC spectrum of 2 M ibuprofen in CDCl3. In the 2D spectrum, the peaks show which proton is attached to which carbon. For definite peak assignment, a similar experiment with the HETCOR spectrum can be carried out.
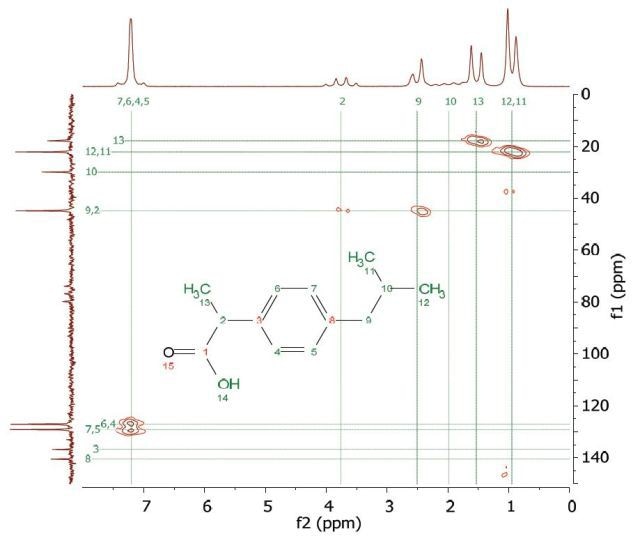
Figure 9. HMQC spectrum of 2 M ibuprofen in CDCl3.
Heteronuclear Multiple Bond Correlation
The Heteronuclear Multiple Bond Correlation (HMBC) experiment can be utilized to achieve long-range correlations of proton and carbon via two or three couplings. Similar to the HMQC experiment, the proton signal appears along the f2 dimension and the carbon signal along the f1 dimension. Figure 10 shows the HMBC spectrum of 2 M ibuprofen in CDCl3.
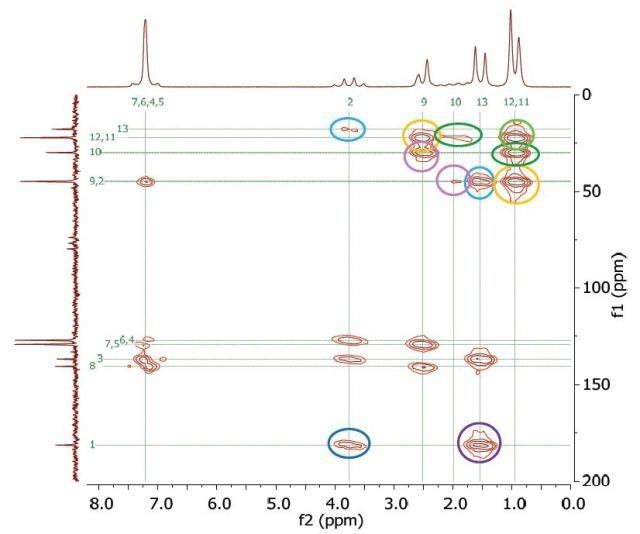
Figure 10. HMBC spectrum of 2M ibuprofen in CDCl3.
The couplings amid the molecular positions appear identical to the couplings seen in the COSY spectrum; however, the HMBC experiment also displays couplings to quaternary carbons. These couplings are not seen either in HMQC or COSY methods. In addition, a correlation between protons and carbons is observed, which is attributed to three-bond bonding from 11 and 12 and the other way round, as indicated in light green in Figure 1.
Spinsolve Carbon NMR Spectrometer
Magritek’s Spinsolve Carbon (Figure 11) is a high-performance and low-cost benchtop NMR spectrometer that now has 19F, 13C and 1H capability, in addition to 2D experiments such as 2D JRES, COSY, HMQC, HMBC and HETCOR. This advanced instrument is perfect for synthetic chemistry, reaction monitoring, chemistry education, and industrial QA and QC applications.

Figure 11. Spinsolve Carbon
Specifications of the Spinsolve Carbon are as follows:
- Resolution: 50% linewidth < 0.7 Hz (16ppb)
- Frequency: 10.8MHz carbon, 42.5MHz proton
- Lineshape: 0.55% linewidth < 20Hz
- Weight: 55kg
- Dimensions: 58 x 43 x 40cm
- Stray field: < 2 G all around system
- Magnet: Permanent and cryogen free
Conclusion
Magritek’s Spinsolve Carbon benchtop NMR spectrometer is a compact and low-cost instrument that is suitable for performing common 2D experiments such as 2D JRES, COSY, HMQC, HETCOR, etc. This revolutionary apparatus not only saves time and space , but also delivers unparalleled performance with excellent sensitivity.
About Magritek
Founded in 2004, Magritek is an advanced technology company exporting from Germany and New Zealand to customers all over the world. The initial technology and IP used in Magritek products was developed by research teams at RWTH Unviersity, Germany, and Massey University and Victoria University of Wellington in New Zealand.
Today, Magritek provides complete NMR and MRI system solutions for the oil and gas industry, as well as components and subsystems suitable for research laboratories and education.
This information has been sourced, reviewed and adapted from materials provided by Magritek.
For more information on this source, please visit Magritek.