This article discusses testing of the base gel for a nail polish system cured by near-ultraviolet light. A typical gel nail polish comprises the application of a base gel, followed by color gel and top gel onto the fingernail. As per the recommendation of the manufacturer, each coat is cured before applying the next layer by exposing it to blue-violet LED illumination for a period of 45s.
Lambient Technologies performed the testing of the base gel by applying it onto Varicon sensors and exposing it to three lamps, namely “Normal,” “Wave” and “Gradual.” The objective of the test is to verify the impact of these different illumination schedules on cure times.
Experimental Procedure
A nail polish brush was used to apply a thin base gel layer over the Varicon sensor, followed by placing the sensor below the selected lamp. An LTF-631 High Speed Dielectric Cure Monitor performed the data acquisition by measuring curing with a 1kHz excitation at 0.1 sec/data point rate.
The maximum measurement speed of the LTF-631 is around 50ms/data point and is capable of handling rapidly curing materials such as thermosets and UV cured adhesives with rapid gelation times.
The lamp was switched on roughly 30s after the onset of data collection in order to determine whether curing occurred prior to light exposure. The exposure time recommended by the manufacturer was 45s, but data acquired by continuously exposing the base gel to near-UV light for several minutes. The CureView software from Lambient Technology was used for data acquisition and storage, as well as for post-analysis and presentation of the results.
Experimental Results
Figure 1a, 1b, and 1c are graphs of temperature, log(ion viscosity) and slope for cure of the base coat under different illumination schedules. The log(ion viscosity) line is flat and remains unchanged before turning on the lamps after 0.5 minutes, revealing the absence of cure. The onset of cure is observed immediately after light exposure as represented by the increased value of log(ion viscosity), which becomes flat after some time, indicating the end of cure.
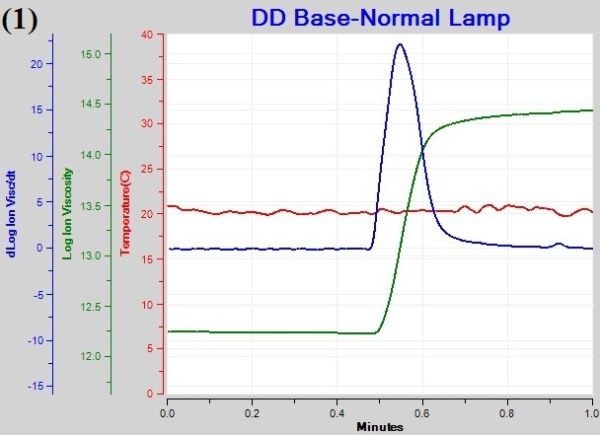
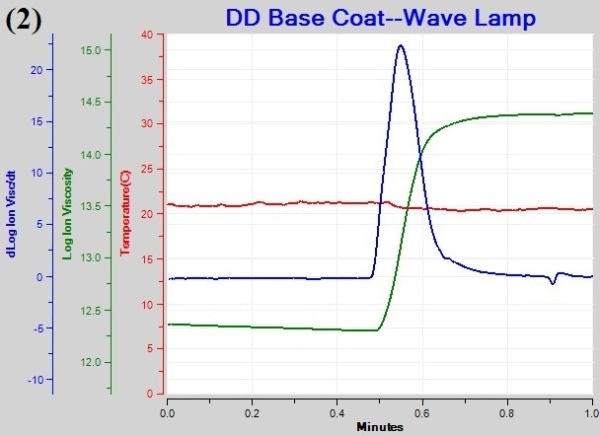
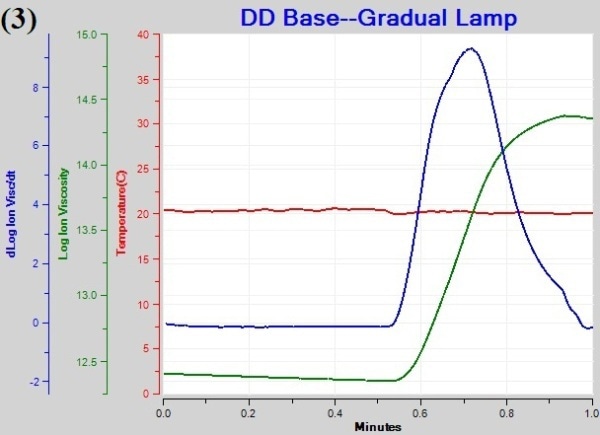
Figure 1. Plots of temperature (1), log(ion viscosity) (2) and slope for cure of the base coat (3).
Four Critical Points (CP) characterize the cure curve:
- CP(1) represents the material flow due to melting of the material. Since, softening and flow of the gel base do not occur following the onset of cure, CP(1) is irrelevant.
- CP(2) is the point of viscosity minimum. However, the viscosity of the base gel is at its minimum at the onset of cure, CP(2) does not provide any valuable information.
- CP(3) is the point of maximum slope, indicating the point of maximum curing rate. The value for the maximum slope in the case of “Normal” and “Wave” lamps is 25 when compared to the value of 9.5 for the “Gradual” lamp, indicating slower reaction rate under this lamp.
- CP(4) is a user-selected level that indicates the end of cure and varies with the application requirements.
The total cure time for the “Normal”, “Wave”, and “Gradual” lamp is 24.6. 19.8. and 38.4s, respectively. The CPs for cure of the base gel under exposure to different lamps are summarized in Table 1.
Table 1. Critical Points from base gel cure monitoring
| Cure Temp. (°C) |
CP(1) Crit. Visc. |
CP(2) Min. Visc. |
CP(3) Max Slope |
CP(4) Crit. Slope |
| Value |
Start of cure |
Value |
Time after start |
Value |
Time after start |
Value |
Time after start |
| Normal |
--- |
28.8 s |
12.25 |
28.8 s |
25.0 |
8.0 s |
0.1 |
24.6 s |
| Wave |
--- |
29.4 s |
12.25 |
29.4 s |
25.0 |
8.0 s |
0.1 |
19.8 s |
| Gradual |
--- |
31.2 s |
12.35 |
31.2 s |
9.5 |
10.8 s |
0.1 |
38.4 s |
Curing Behavior of a Thermoset
Ion viscosity is described as the frequency independent resistivity (ρDC). In most of the cases, ion viscosity varies in proportion to mechanical viscosity in the initial stage of curing and represents cure state in the latter stage of curing.
Ion viscosity obtained from data at a single frequency yields a characterization curve of the state of cure. Figures 2 and 3 depict the curing behavior of a typical thermoset with one temperature ramp step and one temperature hold step.
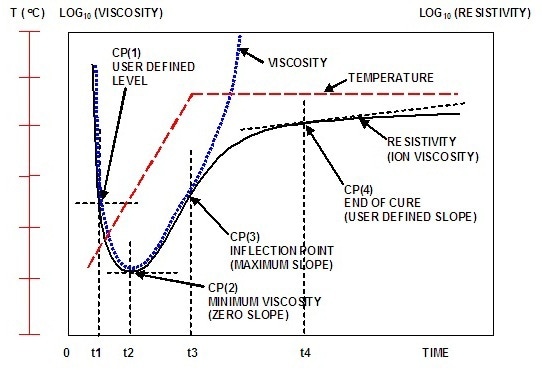
Figure 2. Typical ion viscosity behavior of a curing thermoset
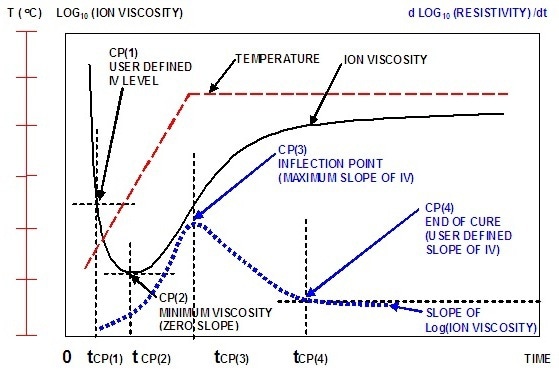
Figure 3. Ion viscosity curve and slope of ion viscosity of a curing thermoset
Ion viscosity decreases initially with an increase in temperature due to melting of the thermoset. The increasing material temperature accelerates the reaction rate. After some time, the onset of crosslinking increases the ion viscosity despite temperature increase. This point is called the ion viscosity minimum, which also takes place at the time of minimum mechanical viscosity.
Subsequent to the minimum point, there is a continuous increment in ion viscosity until the concentration of unreacted monomers decreases with decelerating reaction rate. As a result, there is a decrease in the slope of ion viscosity until the completion of the curing process, at which point slope of the ion viscosity will be zero.
The dielectric cure curve is characterized by four critical points:
- CP(1) is a user-defined level of ion viscosity to determine the start of material flow at the onset of cure.
- CP(2) is ion viscosity minimum indicating the onset of crosslinking and corresponding increase in viscosity.
- CP(3) is inflection point at which the crosslinking reaction begins to decelerate, and is generally used as a signpost that can be related to gelation.
- CP(4) is a user-defined slope capable of defining the end of cure. The slope decreases with decelerating reaction rate.
Conclusion
The curing characteristics of a gel polish cured by near-UV can be effectively observed with dielectric cure monitoring. The normal process requires an exposure time of 45s for curing by blue-violet LED illumination.
However, full cure can be achieved within 25s in the case of “Normal” and “Wave” lamps. The exposure time can be shortened to 30s for these lamps with some margin to ensure complete curing, while reducing the operator exposure to near-UV radiation.
Conversely, an exposure time of 39s is required for the “Gradual” lamp to achieve full cure due to its slower reaction rate. For this lamp, the 45-second exposure time makes sense to ensure complete curing.
The capability of real-time monitoring of gel polish cure not only quantifies the impact of various illumination intensities and formulations, but also determines optimal lamp exposure time.

This information has been sourced, reviewed and adapted from materials provided by Lambient Technologies.
For more information on this source, please visit Lambient Technologies.