Perovskites are calcium titanium oxide minerals which are made up of calcium titanate - CaTiO3. The mineral was first discovered in 1839 in Russia and it has since been discovered that the embedding of cations in the perovskite structure can allow engineering of different materials. Typically, they follow an ABO3 formula where usually ‘A’ is an alkali or rare-earth metal and ‘B’ is a 3d, 4d A or 5d transition metal.
It has been discovered that perovskites have excellent magnetic, electric, and superconducting properties. The ability to incorporate ions and the flexibility of the bond angles permits engineering of different perovskite materials. These properties make perovskites very attractive for a number of different applications, such as photovoltaics.
Using Raman Spectroscopy to Probe the Degradation of Perovskite Film
Since their introduction in 2009, the efficiency of lead halide perovskites has significantly increased. Their cost-effective manufacture and excellent performance make them perfect for photovoltaic applications.
Yet, lead halide perovskites are known for their instability when subjected to adjustments in temperature, light, and oxygen, as well as problems with homogeneity. To better understand the degradation mechanics, it is crucial to test their stability in a practical situation, such as in a device stack.
In order to examine the degradation of individual perovskite layers, in addition to the degradation kinetics, Raman spectroscopy is a powerful probing tool. Raman mapping methods can also be employed to examine the homogeneity of the perovskite film. When humidity and temperature were altered, a Linkam RH95 and THMS600-H were utilized to carry out in-situ Raman spectroscopy and understand the degradation kinetics in more depth.
Firstly, the device stack was checked for thermal degradation by heating to 150 °C. Whilst photo-degradation was established to be dependent on top and bottom layers, thermal degradation was revealed to be irreversible and impact the entire MAPI film. It is dictated by the homogeneity of the film instead of the structural layers.
Using in-situ humidity experiments, Raman signals establish that the dehydration of the perovskite is almost totally reversible once dry. Yet, further exploration of the raman peaks showed that dehydration stayed at the Au region, which suggests that some moisture remained trapped in this area. If trapped moisture can be removed, device performance may be recovered fully.
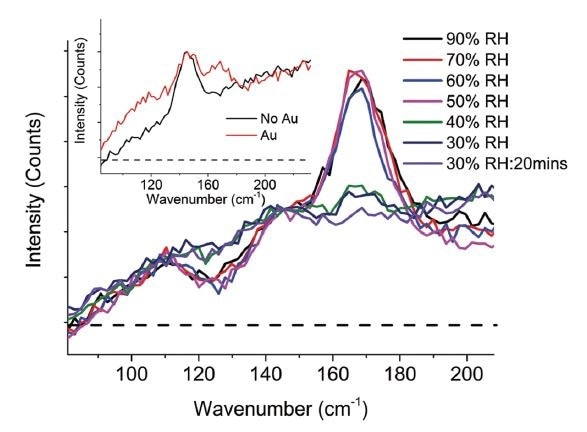
Figure 1. The in situ raman spectra highlights the reversible dehydration when relative humidity is decreased but also indicates the presence of a dehydrated species in the Au region (Hooper, 2017).
Improving Function Using Controlled Engineering
Commonly used as substrates for functional oxide thin films, LaAlO3 and SrTiO3 are wide band gap insulators which have perovskite-based structures. The addition of ions tunes their electronic band structure and so enhances their optical and magnetic properties. The interface between these oxides can interact to prompt conductive and magnetic properties from otherwise non-magnetic, insulating oxides.
In a number of studies, concern has been raised about leakage-current which is caused by structural defects. Understanding these defects experimentally and theoretically is crucial for better utilization of such materials in electronic and optical applications.
Lattice strain is also known to affect the dynamics and role of defects. Popular theories suggest that specific manipulation of these defects can give numerous different functionalities for transition metal oxides. A helpful technique to probe these defects is further provided by Raman spectroscopy.
The low temperature Raman spectroscopy was conducted by setting a temperature range between -196 °C to 600 °C, utilizing a WiTec Raman spectrometer and a Linkam HFS600-PB4 with a liquid nitrogen pump. The temperature stage was employed to carry out temperature dependence of the angular momentum states over 80 to 300 K (-193 °C to 26 °C), which permitted examination of the decay of the angular momentum states with increasing temperature.
Table 1. Raman peaks that were observed for LaAlO3 at low temperatures (Saha, 2016).
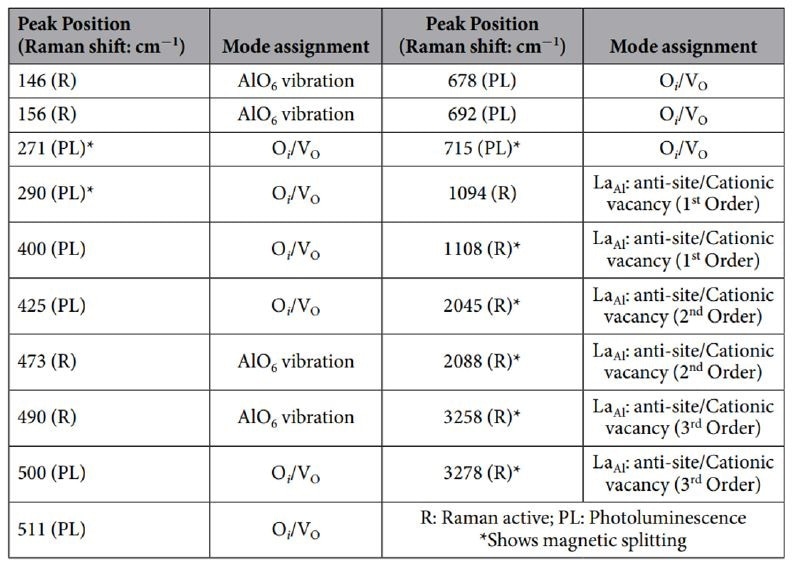
The tests at low temperature supplied evidence for novel transitions which disappear at room temperature. It was found that these transitions were magnetically sensitive, which suggests a magnetic degree of freedom produced by the defects.
Conclusion
Comprehending how to optimize perovskite function and properties is crucial to enhance efficiency. The existence of a heavy element within the transition metal oxide was found to be the key to magnetic sensitive field states.
These angular momentum states and the magnetic interactions can be customized for novel optical applications. The magnetic degrees of freedom could be altered and optimized in rare earth perovskites for optical applications.
Finally, knowing how humidity influences perovskite layers differentially within a stack has shown that targeted drying would improve and regain device performance. Optimizing such performances will have huge advantages for the future development of telecommunication and microelectronics.
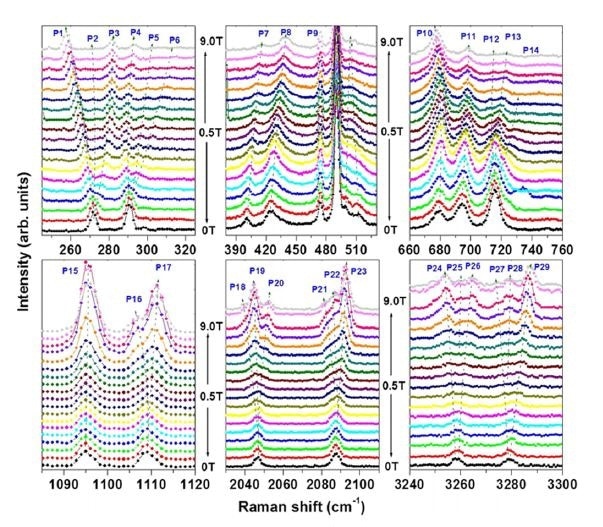
Figure 2. Magnetic-field dependence of new peaks seen at low temperatures. The peaks show Zeeman splitting which indicates the presence of magnetic degrees of freedom (Saha, 2016).
Sources and Further Reading
- Hooper, K.E.A. et al. (2017). Probing the degradation and homogeneity of embedded perovskite semiconducting layers in photovoltaic devices by Raman spectroscopy. Phys. Chem. Chem. Phys. 19, 5246; DOI: 10.1039/c6cp05123e
- Lufaso, Michael W.; Woodward, Patrick M. (2004). “Jahn–Teller distortions, cation ordering and octahedral tilting in perovskites”. Acta Crystallographica Section B. 60: 10. doi:10.1107/S0108768103026661.
- Peña, M. A.; Fierro, J. L. (2001). “Chemical structures and performance of perovskite oxides” (PDF). Chemical Reviews. 101 (7): 1981–2017. doi:10.1021/cr980129f. PMID 11710238.
- Saha, S. et al. (2016). Magnetic Modes in Rare Earth Perovskites: A Magnetic-Field-Dependent Inelastic Light Scattering Study. Sci. Rep. 6, 36859; doi: 10.1038/srep36859
- Wenk, Hans-Rudolf; Bulakh, Andrei (2004). Minerals: Their Constitution and Origin. New York, NY: Cambridge University Press. p. 413. ISBN 978-0-521-52958-7

This information has been sourced, reviewed and adapted from materials provided by Linkam Scientific.
For more information on this source, please visit Linkam Scientific.