Key Issues
- Optimizing a sensitive semi-batch reaction
- Producing real-time data analysis for reliable process control
- Ensuring minimal disturbance to the reaction system with non-contact optics
Introduction
There are several advantages of semi-batch reactions over more conventional batch reactions. These advantages include the greater selectivity of products, the capacity for gradual addition of reactants for better process control, as well as the ability to isolate a product as it is formed.
Semi-batch mode offers the ability to control the composition of the product, which is why polymerizations are done in semi-batch mode, facilitating the creation of high-performance materials with fine-tuned custom properties.
However, semi-batch reactions offer significant challenges. Due to the nature of these reactions, the concentrations of the reactants are in constant flux in absolute and relative senses. If this is left uncontrolled, unwanted properties in the product can result from this variation in the reactant concentrations. Naturally, this reaction mode requires real-time monitoring of the reactant concentrations, as just adding the correct ratios of the reactants is insufficient.
In addition to this, generating high-performance materials with finely tailored properties requires batch-to-batch consistency. In many manufacturing plants, the same process equipment has to be able to produce a wide variety of high-performance materials. Naturally, the process-control method must then be both precise and flexible.
There are several advantages offered by Raman spectroscopy for real-time process monitoring of semi-batch polymerizations. Raman spectroscopy can identify bands specific to particular functional groups such as the vinyl and alkyl moieties common to polymerization monomers, in addition to offering both easy sample interfacing and fiber-optic coupling of the base unit to the probe head, for flexible placement of the base unit relative to the process line.
Experimental
A semi-batch polymerization reaction was carried out in this experiment using three proprietary monomers, designated monomers A, B, and C. Figure 1 shows a schematic of the system.
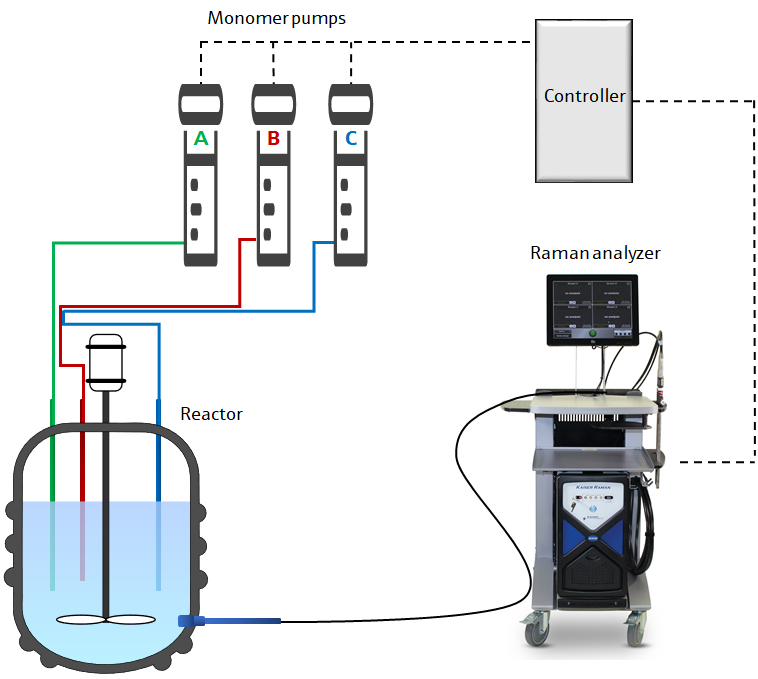
Figure 1. Schematic diagram of process control system for semi-batch polymerization.
To deliver and collect 785-nm near-infrared radiation from an Invictus™ laser, a Kaiser Raman analyzer from Kaiser Optical Systems, Inc, was used. To perform the analysis through a pressure- and temperature-rated sapphire viewport in the wall of the reactor, a non-contact optic was used.
Each sample acquisition consisted of 60 seconds of reactor contents illumination using 125 mW of laser radiation followed by a 20-second pause. Each monomer created five to seven data points, which were used to generate quantitative Raman data for real-time process control.
During the polymerization in this reaction, a vinyl functional group in each of the three monomers changes to an alkyl group. Traditional infrared spectroscopy is not sensitive to this, while Raman is. In this case, chemometric modeling was based on the Raman signal for the vinyl region in all monomers, the C–H wag in monomers A and B, in addition to the lactone region in monomer C.
Results and Discussion
Both open-loop and closed-loop conditions were used in this polymerization reaction. Open-loop refers to circumstances in which spectroscopy is used only for process monitoring, not for feedback control) and closed-loop refers to the process in which spectroscopy is used for process monitoring and real-time feedback control) conditions. The concentrations of these monomers over time in the open-loop experiment are displayed in Figure 2. The concentrations of the monomers vary widely over the course of the reaction, in accordance with the predictions.
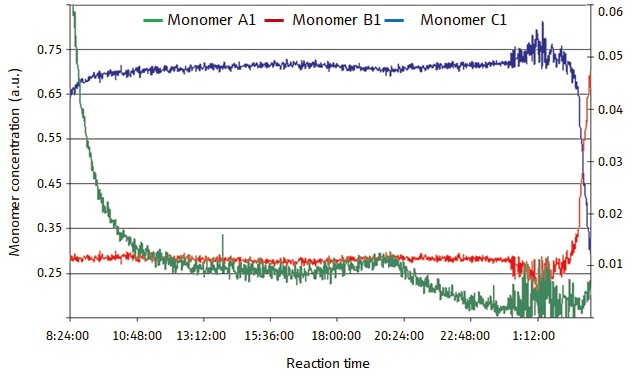
Figure 2. Figure 2 displays monomer concentrations in an open-loop (process monitoring only) semi-batch polymerization reaction. The concentrations of the monomers vary hugely in the course of the reaction, especially monomer A. Image Credit: Reprinted with permission from Ref. 1. © 2006 Compare Networks Inc.
In Figure 3, the concentrations of the three monomers in the closed-loop experiment are displayed, in which the Raman spectral data has been used in real-time feedback control of the process. The concentrations of the monomers in this case are much more constant.
This consistency is thanks to the fact that the process-control system makes sure that each monomer is added to the system as and when it is needed. The middle of the figure is marked by black and gray plotlines which indicate the feed flowrates determined by the process-control system, using the Raman spectral data.
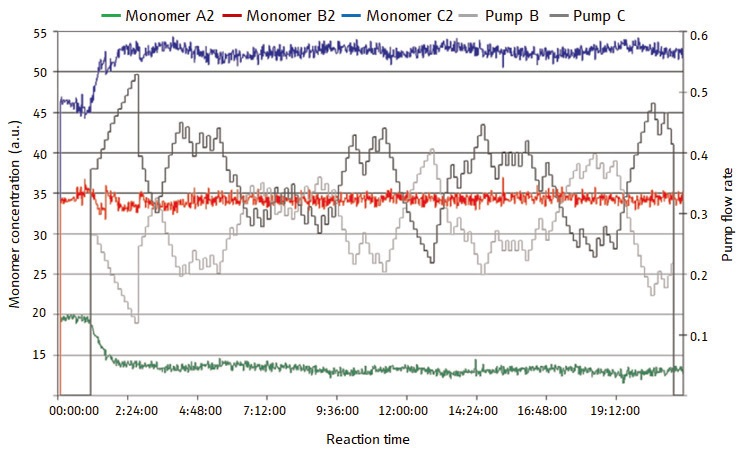
Figure 3. Figure 3 displays the monomer concentrations, in a closed-loop (process monitoring and control) semi-batch polymerization reaction. Over the course of the reaction, the concentrations of the monomers remain very constant. The feed flowrates determined dynamically using Raman data are represented by the plotlines in back and gray at the center of the plot. Image Credit: Reprinted with permission from Ref. 1. © 2006 Compare Networks Inc.
With average molecular weights of the products varying between 28700 and 30200 Daltons and polydispersity varying between 1.79 and 1.84, consistency between the batches was high.
Conclusion
In conclusion, it was demonstrated that Raman spectroscopy is an accurate, simple, and effective method of process analysis when it comes to the real-time monitoring and control of a semi-batch polymerization.
To maintain ideal process conditions in a closed-loop reaction system and for real-time feedback control of the process, Raman data was used. This Raman data ensured consistent quality of sensitive high-performance materials and produced a highly consistent product between batches.
References
- Müller, J. et al. “Semi-Batch Polymerization Analysis by Multi-Point Raman Spectroscopy.” Process Analytical Technology, September/October 2006, 1–6.

This information has been sourced, reviewed and adapted from materials provided by Kaiser Optical Systems, Inc.
For more information on this source, please visit Kaiser Optical Systems, Inc.