The aniline point in mineral base oils is a key indicator for lubricant formulation, particularly in sensitive or high-performance applications.
This metric is essentially the base oil’s solvency, indicating how well it dissolves additives and other components when used in a lubricant formulation.
A lower aniline point indicates greater solvency, making it suitable for dissolving polar additives such as dispersants, detergents, and antioxidants. This is typical of naphthenic or Group I base stocks and offers potential benefits for applications that require high additive loading or precise varnish and deposit control.
Group II and III base stocks typically feature higher aniline points, however, indicating reduced solvency but improved oxidation stability and higher viscosity index. These features make Group II and III base stocks ideally suited to use in contemporary, thermally stable formulations.
High solvency is essential for a range of industrial applications, including rubber plasticization and the production of metalworking fluids and greases. The global transition to more highly refined, less soluble products presents significant challenges for the industry, including technical challenges.
Traditional Determination of the Aniline Point
Product development and incoming product quality control are dependent on knowing the actual aniline point of base oils. This value is provided in °C and describes the lowest temperature at which aniline remains in solution under test conditions.
Measurement of the aniline point is generally performed in line with the ASTM D611 method standard. Numerous suppliers offer measuring devices suitable for determining the aniline point, with varying degrees of automation.
The fundamental issue is that every instance stems from aniline itself. Aniline is toxic, has the potential to cause physical damage, is extremely bad for the environment, and is suspected of being carcinogenic. Every operator tries to avoid using this substance for these reasons.
It is also important to note that, depending on the method, a single determination takes between 20 and 60 minutes, including sample preparation.
Using FTIR Infrared Spectroscopy to Estimate the Aniline Point
FTIR (Fourier Transform Infrared Spectroscopy) measurements are routinely used throughout the lubricant industry due to the method’s rapid, non-destructive capabilities and its ability to offer detailed chemical analysis of both fresh and used lubricants. It is also often necessary to monitor degradation and contamination, as well as to determine the presence and depletion of additives.
FTIR passes infrared light through a sample, measuring the absorption of different wavelengths and generating a spectrum that reveals the molecular composition of the sample.
In this context, chemometrics refers to the use of mathematical and statistical methods. These methods enable the extraction of meaningful chemical information from FTIR spectra, offering useful insight into lubricant properties and conditions. This principle has already proven applicable for predicting Total Base Number (TBN) and Total Acid Number (TAN).
A pilot project was undertaken, confirming that it was possible to estimate the aniline point of base oils using FTIR with the support of an appropriate chemometric model.
Proof of Concept with FTIR Infrared Spectroscopy
The proof of concept employed a total of 30 base oil samples with kinematic viscosities at 40 °C ranging from 8 mm2 per second to 115 mm2 per second. This corresponded to aniline points between 70 °C and 105 °C.
Measurements were performed with eralytics’ ERASPEC OIL FTIR spectral lubricant analyzer.
Figure 1 plots all measured infrared spectra for comparison. It is clear that no straightforward approach is applicable in this instance, but certain variations can be observed across sections, for example, in the range between wavenumbers 650 cm-1 and 950 cm-1.
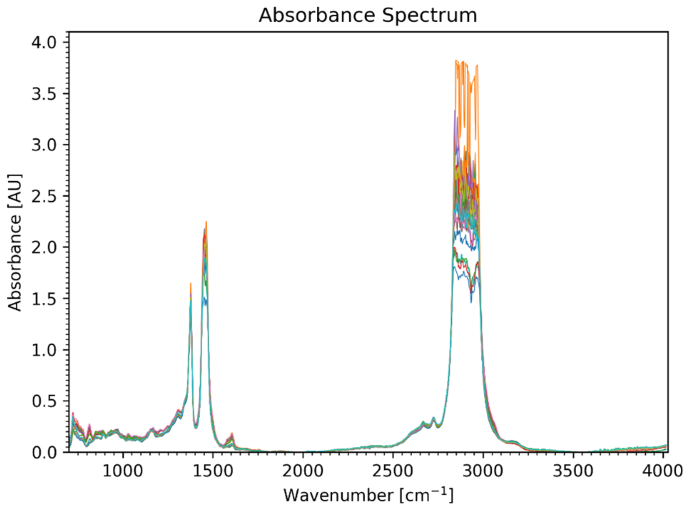
Figure 1. Transmission spectra of all 30 samples. Image Credit: eralytics GmbH
A chemometric multilinear regression (MLR) model was developed to integrate information from multiple areas. Figure 2 shows the cross-plot of calculated and reference values for the aniline point. The calculated Standard Error of Calibration (SEC) in this instance was 1.0 °C.
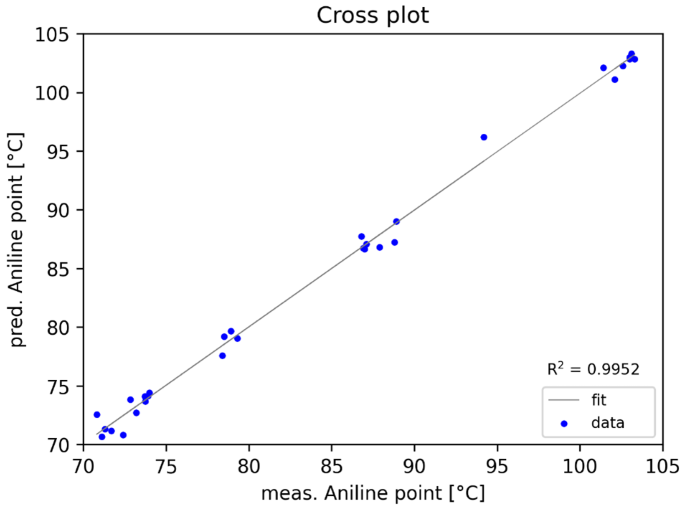
Figure 2. Cross plot showing aniline point versus the predicted value. Image Credit: eralytics GmbH
This number is compared against the reproducibility of 0.5 °C for clear and light-colored samples stipulated in ASTM D611, providing a very reasonable outcome. It is important to note that all samples were used across a wide viscosity range.
It can therefore be assumed that further studies will enhance the significance of this approach. It is also possible to consider the density of the respective sample in the model, because this is generally determined.
Conclusion
Knowledge of the aniline point of base oils poses challenges and offers technical opportunities, yet remains extremely important in the lubricating oil and process oil industries.
FTIR infrared spectroscopy offers an alternative approach to the traditional determination, which relies on the solubility of the extremely questionable substance aniline. This measurement provides in-depth insight into the composition and condition of the base oils and takes just a few minutes to perform.
Acknowledgments
Produced from materials originally authored by Thomas Feischl from eralytics GmbH and Daniel Abbenor from Nynas AB.

This information has been sourced, reviewed, and adapted from materials provided by eralytics GmbH.
For more information on this source, please visit eralytics GmbH.