New research in the journal Science Advances demonstrates how a multicomponent perylene mixture can be used to reduce the fragility of glass. The research focuses on synthesizing an octonary perylene mixture.

Study: Vitrification of octonary perylene mixtures with ultralow fragility. Image Credit: Alex Ander
The classic process of vitrification involves rapid cooling of a liquid mixture to form a noncrystalline form of glass material. According to the study published in Science Advances by Christian Müller and his team, this process has a major disadvantage in that it yields a product with a very high fragility.
This has been a major concern for industrial manufacturers. However, this latest study involves the utilization of a slowly cooled multicomponent perylene mixture to generate glass formers that have a very low characteristic value of fragility.
This discovery is paramount as it could pave various paths for the successful development of glass-related formers and byproducts which have a very high strength value and could be used in various industries such as the solar-powered electronics industry, medical industry, as well as superconductor industry.
Methods, Materials, and Properties
The non-crystalline glass formation is highly dependent on thermal and kinetic factors, which are also the concerning factors for glass transitioning temperature T g and glass melting temperature T m. The research study states that the mixing of several distinct substances effectively reduces the crystallization process, and also a massive decrease in fragility is observed in isomers, antiplasticizers, etc.
The results showed a massive decrement in fragility up to 500% by implementing octonary perylene mixtures. Such mixtures constitute a single carbon-based molecule with no dissimilar atom or molecule. Pure perylene has a very high capacity to form crystalline structures with a melting temperature of 287°C.
However, the additives in octonary mixtures stop the crystallization process completely along with a rapid decrement in melting temperature. Increasing the number of chemically viable components is useful in decreasing the fragility of such mixtures up to a mere constant of 13, proving their efficacy.
The study showed that various essential techniques were implemented for validation. Differential Scanning Calorimetry (DSC) was performed for various samples. Mettler Toledo DSC2 comprising of a gas controller GC200 was utilized to perform DSC.
This was also used for the Fast Scanning Calorimetric (FSC) process. However, for the process of Emission Spectroscopy, a perylene mixture with Dichloromethane (DCM) was dried and pressed by another layer of the substrate. Linkam LTS420 hostage was effectively utilized for heating and efficient cooling for low fragility glass formation by utilizing a perylene mixture.

Image Credit: Roman Zaiets/Shutterstock.com
All of these processes were essential for the development of glass formers with an unconventionally low but desirable value of durability.
Conventional Industrial Utilization
These vitrification techniques using classical components have major disadvantages, but being the only option available at that time were used extraneously in many industries. It is essential for the development of important substances such as Silicon Dioxide, the organic semiconductor manufacturing sector, solar cell development, organic LED component manufacturing, nuclear waste disposal, amorphous pharmaceutical products, and essential products of the medical and protein drugs sector. Such industries have been utilizing this process for a very long time, assuring an improvement in human living standards.
To revolutionize these operational techniques, researchers were focused on manufacturing substances with non-crystalline high glass-forming ability with a suitable value of fragility. This modern study has efficiently surpassed all these baseline expectations, by octonary perylene mixtures with a very low fragility value indicating strength and superior thermal, electric, and mechanical properties as compared to its classical counterparts.
Latest Research on Octonary Perylene Mixtures
The latest research has effectively demonstrated the hypothesis that a very substantial fragility change would assist in designing high-strength glass-forming substances exceeding industrial requirements. The mixture consisted of eight unique substances which, although they independently possess a very high fragility value of greater than 70, form the final glass-forming octonary perylene mixture at a low value of almost 13.
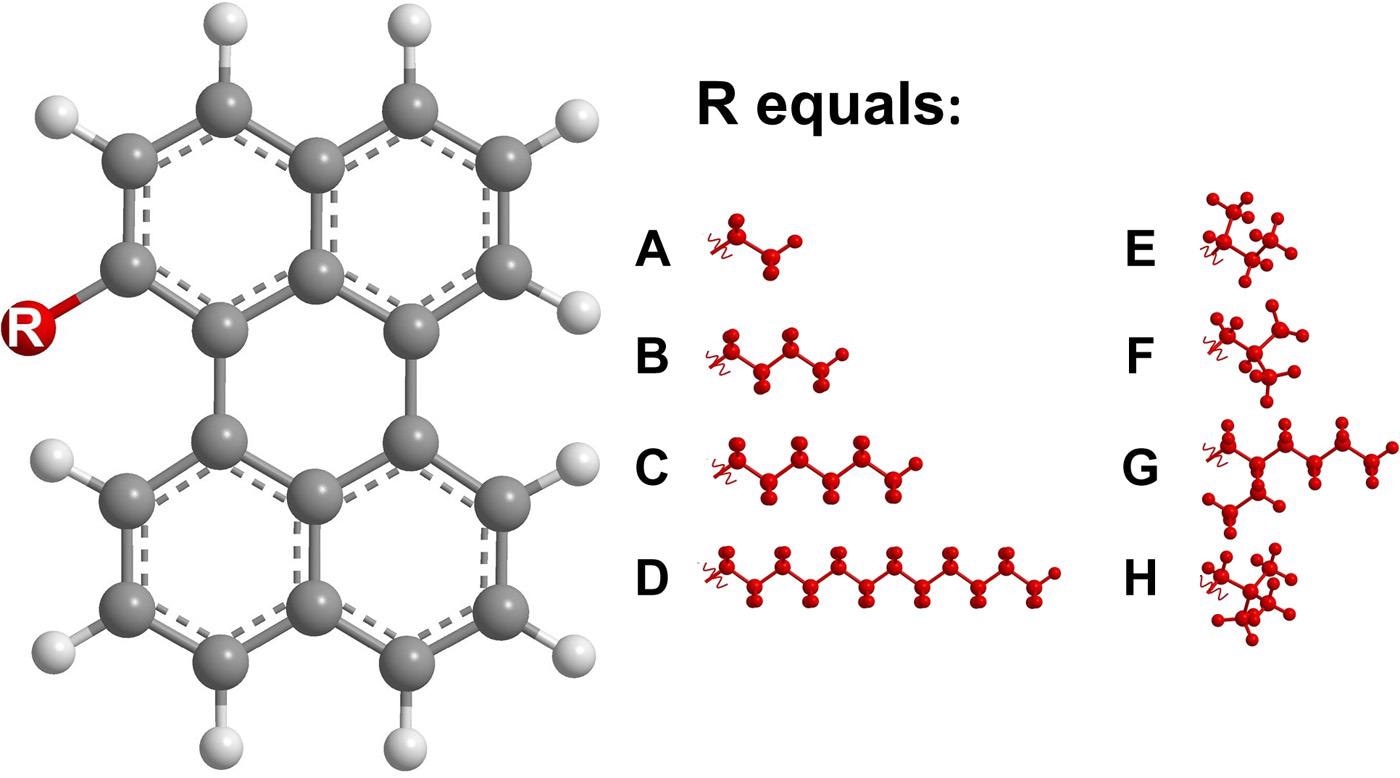
Molecular structures of the bay-substituted perylenes with different pendant alkyl groups. The molecules are labeled A to H. Image Credit: Hultmark, S et al., Science Advances
Such perylene mixtures contained a branched alkyl group held by the intermolecular forces at the substance bay position. The DSC thermogram technique was utilized which displayed a rate of -0.17K s-1. The crystallization inhibition property of this mixture was due to the twisting of the conjugated core fulfilling the basic purpose and requirement.
The next essential step in the study was the study of mixing impact which showed an effective reduction in glass transition temperature. The focus was on the ethyl, butyl, and hexyl attached branches. The DSC process validated a decrease in Tm due to the mixing of several components.
This is crucial for stopping crystal development. Since the basic requirements were completed, the study presents the impact of mixing variation on fragility, which is the essential novelty the mixture offers.
FSC efficiently assisted in the study of the effects of cooling rates on the fictive value of temperature Tf’. The technique of area matching method was utilized for this purpose. The results confirmed that the addition of components in a scientific method reduced fragility extensively.
The final fragility of the octonary mixture was measured by three different independent techniques which gave us a value of 13 (error of 2%), which is quite a lot better than the lowest value of organic or inorganic glass-forming substances, such as SiO2.
Future Industrial Insight and Limitations
As liquid chiral molecules for thermal energy storage and photon-up conversion, mixtures of bay-modified perylenes are of major interest. In a broader sense, glass-forming mixtures that avoid crystallization by undergoing a liquid-liquid transition may serve as a stepping stone for the development of active pharmaceutical products, organic semiconductors potentially setting the stage for more thermoplastic polymer synthesized organic solar cells, and prescription medications with extended shelf lives.
However, this technique is only synthesized using an octonary perylene mixture. If the constituent 8 distinct molecules are not available, this octonary perylene mixture of the glass former would not be synthesized. Hence, this fragility value is only obtained for this particular mixture; it cannot be standardized. A change in mixture composition would lead to a change in fragility value, and this is a major disadvantage.
In a nutshell, this study in Science Advances truly has revolutionized the glass-forming industry. The fragility value is the lowest ever documented, validating that the novel industrial technique has been successful in reducing glass former fragility.
References
Hultmark, S., Cravcenco, A., Kushwaha, K., Mallick, S., Erhart, P., Börjesson, K., & Müller, C. (2021). Vitrification of octonary perylene mixtures with ultralow fragility. SCIENCE ADVANCES. https://www.science.org/doi/10.1126/sciadv.abi4659
Monnier, X., Saiter, A., & Dargent, E. (2017). Vitrification of PLA by fast scanning calorimetry: Towards unique glass above critical cooling rate? Thermochimica Acta, 47-54. https://www.sciencedirect.com/science/article/pii/S0040603117302745
Phan, A. D., & Schweizer, K. S. (2020). Theory of Spatial Gradients of Relaxation, Vitrification Temperature and Fragility of Glass-Forming Polymer Liquids Near Solid Substrates. ACS Macro Letters, 448-453. https://pubs.acs.org/doi/abs/10.1021/acsmacrolett.0c00006
Schmelzer, J. W., & Tropin, T. V. (2019). Kinetic criteria of vitrification and pressure-induced glass transition: dependence on the rate of change of pressure. Thermochimica Acta, 42-53. https://www.researchgate.net/publication/345502720_Kinetic_criteria_of_vitrification_and_pressure-induced_glass_transition_dependence_on_the_rate_of_change_of_pressure
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.