Titanium Dioxide is the world’s most widely utilized white pigment. It’s high refractive index, ease of dispensability and photochemical stability are unique. For this reason it is used to impart opacity and whiteness to an enormous variety of products including paints, plastics, inks, paper, cosmetics, foodstuffs and pharmaceuticals.
The Importance of Pigment Particle Size
One of the most important factors in determining the effectiveness of titanium dioxide pigments is the pigment particle size. The particle size controls several important properties including:
- Color
- Opacity (scattering strength)
- Gloss
- Tinting strength
- Viscosity
- Sedimentation rate
The Effect of Over-Sized Particles
The presence of over-sized particles can significantly impair the performance of the final pigment dispersion. It is therefore necessary to monitor the dispersion characteristics during production and use of the titanium dioxide in order to obtain optimal pigment performance.
Pigment Particle Size and Light Scattering Strength
The particle size of titanium dioxide pigments must be carefully controlled in order to maximize the light scattering from the pigment suspension. The particle size (D) at which the maximum scattering power is seen for a high refractive index pigment is given by Weber’s formula:
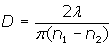 1
1
where λ is the wavelength of the incident light, n1 is the refractive index of the pigment and n2 the refractive index of the continuous phase. Table 1 shows the optimum size required for rutile titanium dioxide in water in order to see maximum scattering for blue, green and red light.
Table 1. Optimal particle size for rutile in water in order to see maximum scattering of blue, green and red light.
| Wavelength |
Size (µm) |
| Blue (430 nm) |
0.20 |
| Green (550 nm) |
0.26 |
| Red (660 nm) |
0.31 |
A particle size of around 0.25 microns yields the highest scattering efficiency across the entire visible spectrum for rutile titanium dioxide. For anatase titanium dioxide, the ideal particle size is around 0.3 microns. Milling to below this size significantly reduces the pigment scattering power and increases the Manufacturer’s costs, both in terms of milling time and the concentration of pigment required to give a certain tinting strength. Conversely, the presence of large particles or aggregates yields a yellow undertone due to poor scattering efficiency at the blue end of the visible spectrum. The presence of aggregates also reduces the tinting strength, gloss and opacity of the pigment suspension, as well as increasing the viscosity due to particle interactions. It is therefore important to carefully control the pigment particle size and eliminate any aggregates during milling.
Dispersion Monitoring Using The Mastersizer 3000
Primary Measurements
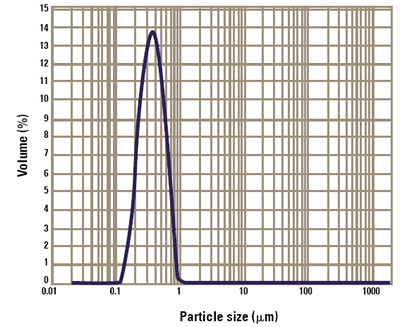
The particle size of a titanium dioxide suspension was determined using the Malvern Panalytical Mastersizer 3000. Measurements were first carried out in demonized water. Figure 1 shows the size distribution reported by the instrument. As can be seen, the "primary" particle size for this titanium dioxide sample is close to that required in order to see the maximum amount of scattering from the particles. Five individual results are over-plotted in figure 1.This shows the repeatability of the results obtained using the Malvern Panalytical Mastersizer 3000.
|
|
|
Figure 1. Primary particle size distribution for the titanium dioxide slurry.
|
Aggregate Detection
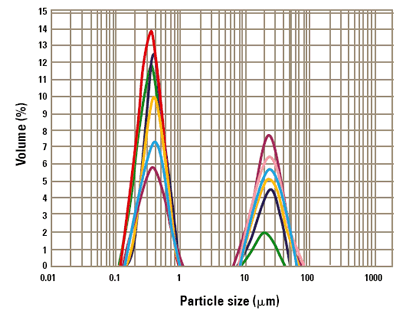
The ability to detect aggregates as well as "primary" particles is of paramount importance to the paint manufacturer. The titanium dioxide slurry was therefore destabilized by adding an excess of a highly charged electrolyte to the Malvern Panalytical Mastersizer 3000 sample dispersion unit. This reduces the charge on the particles, leading to particle aggregation. The particle size reported by the instrument for the unstable slurry is shown in figure 2.
As can be seen, the instrument is able to detect both the primary particles and the large aggregates formed during destabilization of the slurry. Figure 3 shows the change in the volume mean diameter (D[4,3]) reported by the Malvern Panalytical Mastersizer as the aggregation process is continued. The instrument successfully tracks the growth of the aggregates until an equilibrium particle size is attained.
|
|
|
Figure 2. Particle size distributions reported during the destabilization of the titanium dioxide slurry.
|
|
![Change observed in the volume average particle size (D[4,3]) during aggregation of the titanium dioxide slurry.](/images/Article_Images/ImageForArticle_2809_45076396745486111905.gif)
|
|
Figure 3. Change observed in the volume average particle size (D[4,3]) during aggregation of the titanium dioxide slurry.
|
Conclusions
The ability of the Malvern Panalytical Mastersizer 3000 to measure the particle size of titanium dioxide slurries has been demonstrated. The instrument provides an ideal method for monitoring the state of dispersion of titanium dioxide paints and pigments, an important factor in controlling the performance of the final product.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information on this source, please visit Malvern Panalytical.