Jun 1 2017
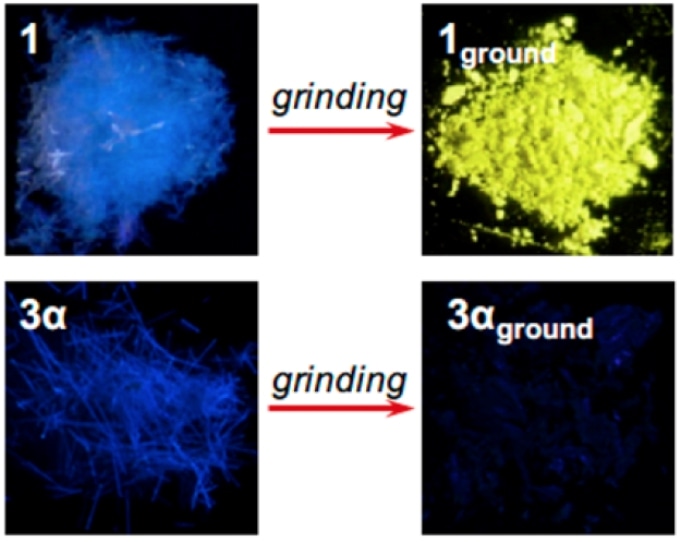 9-anthryl gold (I) isocyanide (3α) turns invisible and emits infrared after it’s ground whereas phenyl gold (I) isocyanide (1) turns yellow. (Seki T. et al., Journal of the American Chemical Society, May 2, 2017)
9-anthryl gold (I) isocyanide (3α) turns invisible and emits infrared after it’s ground whereas phenyl gold (I) isocyanide (1) turns yellow. (Seki T. et al., Journal of the American Chemical Society, May 2, 2017)
Certain materials luminesce, changing their color and intensity when exposed to mechanical forces such as rubbing or grinding. These luminescent “mechanochromic” materials can create a range of emission colors in the visible light spectrum, from blue to red. Their color-changes under force are properly documented, and are caused by variations to the molecules’ crystal structures.
Recently, a big change from the visible spectrum to the infrared has been discovered and described in the Journal of the American Chemical Society. Such a large variation is unparalleled and is exciting because of its possible applications in bio-imaging and invisible inks.
In an effort to create new mechanochromic compounds, a research team at Hokkaido University in Japan discovered that a gold compound called 9-anthryl gold (I) isocyanide complex has a unique feature. In its original form, the material created a visible blue fluorescence having a wavelength of 448 nm. After being ground up into a fine powder, the material created infrared emissions (phospholescence) having a wavelength of 900 nm. The infrared emissions cannot be seen by the naked eye.
This is the first time a material has been reported to make such a dramatic shift – a change of 452 nm – that also reaches into the infrared part of the light spectrum.
Tomohiro Seki, Hokkaido University
The research team’s X-ray crystallographic evaluation exposed that the large shift is based on a crystalline-to-amorphous phase transition which should generate robust intermolecular interactions between the gold ions.
The development of infrared emissive materials is generally difficult, and appropriate design strategies remain limited. However, in this case, simple grinding can afford an infrared emissive material. Infrared is invisible to the naked eye but detectable using a spectrometer. So, our material has a great potential for bio-imaging and security inks.
Hajime Ito, Hokkaido University