Cellulose, a readily available and cheap biopolymer synthesized by plants, is primarily obtained by recycling used paper and processing deciduous and coniferous trees. However, supporting substances, such as hemicellulose and pectin, typically co-exist with plant-based cellulose.
Although such a chemical composition does not adversely impact the application potential of plant-based cellulose (PC), products with 100% cellulose content, such as BC, are often preferred over PC when used as a biomaterial.
Different bacteria, including Gram-positive bacteria such as Sarcina and Gram-negative bacteria such as Agrobacterium and Gluconacetobacter xylinus, can synthesize BC. Almost 59 bacterial strains from ripe vegetables and fruits can synthesize BC.
Cellulose with better structural and physicochemical properties can be synthesized in vitro using the cell-free enzyme system that employs enzymes derived from fungi or bacteria. The cell-free method is more efficient and uses less glucose compared to the bacteria-based methods, and the resulting fibers possess larger diameters.
The BC synthesis mechanism influences specific properties of cellulose, including the degree of polymerization and crystallinity, mechanical strength, porosity, water holding capacity, and biodegradability.
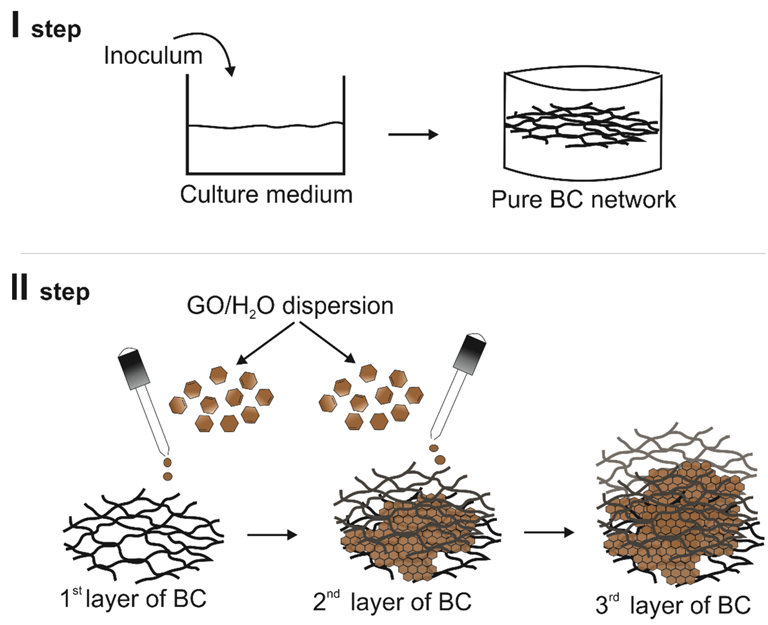
The process of obtaining modified BC. Image Credit: Fryczkowska, B et al., Polymers
BC can store, absorb, and desorb large amounts of water as a hydrogel owing to its nanofiber structure makes BC an exceptional biomedical material. BC can also form composite materials when combined with biopolymers such as collagen, chitosan, or alginates.
Composites such as BC/chitosan composite were already synthesized as a dressing material to treat different ulcers, wounds, and burns, while BC/alginate composites were used for tissue engineering.
BC can be modified using ex-situ and in situ methods to introduce undescribed and new properties. The in-situ method involves the addition of water-soluble nanoparticles or additives directly into the culture medium to modify BC.
Several studies demonstrated BC modification using electrically conductive polymers and carbon nanoadditives, such as GO or graphene, and electrically conductive polymers through the in-situ method.
The modification using carbon nanoadditives increases the porosity and specific surface area and improves the strength of the composite. GO has specifically gained attention among carbon nanoadditives for the synthesis of BC composite as the two-dimensional (2D) material strongly links to BC by forming hydrogen bonds.
Several BC/GO nanocomposites with multiple conformations were developed for different applications in the field of biomedicine. For instance, graphene oxide was used as a delivery carrier for medicines or genes and to develop biosensors.
The Study
In this study, researchers synthesized BC/GO composite membranes with different concentrations of GO using an in-situ method and investigated the influence of GO nanoparticles on the properties and structure of the synthesized membranes.
Researchers also investigated the feasibility of using the synthesized composite membranes as a potential active dressing with the sustained-release analgesic paracetamol. Paracetamol was selected for the in-vitro biologically active substance sorption study as it is a strong antipyretic and analgesic medicine.
Microorganisms obtained from the Golden Delicious apple vinegar were utilized to synthesize the BC. No additional nutrients were added for bacterial growth. Additions such as citric acid, yeast extract, and peptone were not used for cellulose biosynthesis.
A liquid sucrose medium with a 110 g/L concentration was prepared and sterilized at 121 oC in an autoclave for 15 min. Subsequently, 200 mL of apple cider vinegar, a 1 × 1 cm fragment of “mother of vinegar” containing live bacteria, yeast, and BC, and 800 mL of liquid sucrose medium were placed in a 2000 mL sterilized laboratory beaker and thoroughly mixed.
The resultant mixture was divided into four parts and placed in sterilized beakers. Every beaker was then covered with a watch glass and transferred to an incubator preheated to 25 oC. The BC/GO composite synthesis was performed for 12 days by adding different amounts of GO dispersion to each of the four samples.
The concentration of GO was maintained at 50 ppm to facilitate the synthesis. BC/GO composite membranes with 3.7%, 5.4%, and 7.1% of GO were designated as BC/GO/I, BC/GO/II, and BC/GO/III, respectively. The purification of the synthesized membranes was performed using a 2% w/w aqueous solution of potassium hydroxide (KOH) and distilled water.
An electronic micrometer screw and an electronic thickness gauge were employed to measure the thickness of the wet BC/GO membranes and dry BC/GO membranes, respectively. The water content in wet samples and water absorption in dry samples were measured using a laboratory electronic moisture balance.
The bioactive substance release kinetics from the membranes were studied in-vitro in phosphate-buffered saline (PBS) that mimics the pH, osmolarity, and ion concentration of human body fluids.
Four solutions containing PBS as a solvent and 20.0, 10.0, 5.0, and 2.5 ppm paracetamol were prepared and then the desorption and sorption of paracetamol from the synthesized membranes were investigated. Tests performed using dry membranes were unsuccessful. Thus, these experiments were performed using wet membranes as carriers of paracetamol.
Scanning electron microscopy (SEM), Fourier transform infrared (FTIR) spectroscopy, wide-angle X-ray scattering (WAXS) studies, and thermogravimetric analysis were performed for the characterization of membranes.

Samples of wet membranes placed on glass, prepared for thickness measurement; (a) top view; (b) side view. Image Credit: Fryczkowska, B et al., Polymers
Observations
BC/GO composite membranes with different GO concentrations and average water content of 400%, wet thickness of 1100 µm, and nanofiber diameter of 100 nm were synthesized successfully. The KOH treatment effectively eliminated organic matter such as bacterial cells from the membranes.
The BC nanofibers in the BC composites were arranged in a random manner, similar to classic non-woven fabric. The GO flakes were invisible in the SEM studies as they were covered by the BC nanofiber web. In wet membranes, the thicker BC fibers formed a loose structure containing a significant amount of water in the inter-fiber spaces.
GO inclusions were observed in the transparent dry BC structure in BC/GO/III, BC/GO/II, and BC/GO/I membranes. The GO additive was not dispersed in a uniform manner as GO was randomly woven into the BC nanofibers, and no GO agglomeration was observed.
No change in the BC structure was observed due to the addition of GO. However, the degree of crystallinity of BC/GO composites decreased with the increasing GO concentration as the GO nanoparticles disturbed the formation of BC nanofibrils.
Moreover, the addition of GO slightly increased the thermal decomposition temperatures of the composite membranes compared to pure BC membranes, which improved their thermal stability.
The increasing concentration of GO increased the thickness of the wet BC/GO composite membranes due to a rise in nanofiber thickness. The lowest and highest thickness of 470 µm and 1180 µm were observed in pure BC and BC/GO/III membranes, respectively.
However, the thickness declined substantially after drying the wet membranes due to the formation of intra- and intermolecular hydrogen bonds during cellulose drying. Moreover, the formation of additional hydrogen bonds in samples with high GO concentration increased the compactness of the structure, which reduced the thickness of these membranes.
The addition of GO increased the water content in wet membranes, with the highest water content observed in BC/GO/II membranes. However, the water content was reduced in BC/GO/III membranes due to additional hydrogen bond formation. The water absorption values of all dry membranes were almost the same, with a slight reduction in value observed in GO-incorporated samples due to hydrogen bonds.
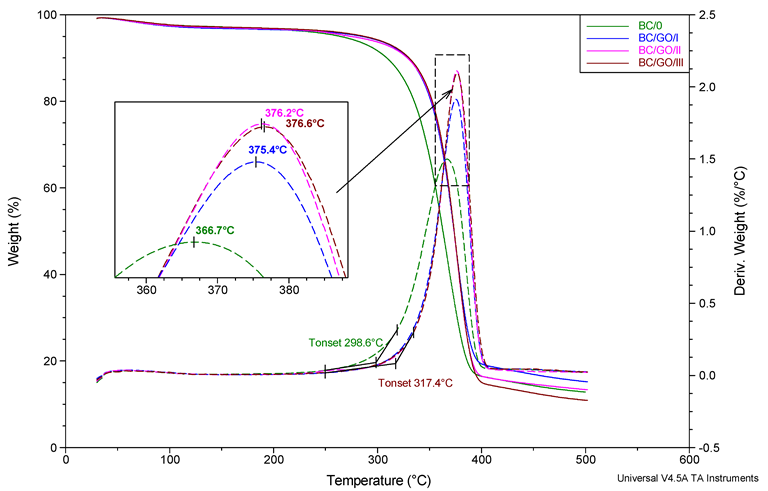
Summary of TG and dTG curves for membrane BC/0, BC/GO/I, BC/GO/II, BC/GO/III samples. Heating rate 20°/min. N2 atmosphere flow 60 mL/min. Image Credit: Fryczkowska, B et al., Polymers
The BC/GO composite membranes demonstrated 60 mg/g BC paracetamol sorption, a slow desorption time, a constant medicine concentration, and a high paracetamol release rate after 24 h.
The pure BC membranes adsorbed 63.3 mg/g BC of paracetamol, which decreased slightly to 62.4 mg/g BC in the BC/GO/III membrane due to the increase in GO concentrations. However, the desorption tests displayed that the composite membranes desorbed more medicine than pure BC membranes, with 42.0 mg/g BC paracetamol desorption observed in BC/GO/III membranes.
Pure BC membranes slowly and steadily desorbed paracetamol. However, the release rate was less than 55%. The BC/GO/I membrane showed intense paracetamol desorption during the initial release phase, which slowed down after 40 min and remained at 55%. Both BC/GO/II and BC/GO/III showed high release rates at 67% even after 24 h.
To summarize, the findings of this study demonstrated that the BC/GO composite membranes could be used effectively as potential active dressings with sustained-release analgesic medicine.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.