An in-service lubricant encounters heat, air, corrosive agents, pressure, and other factors that cause chemical changes in the oil. Changes in oil chemistry can also impact the lubricant’s ability to perform its job (for instance, if key additives have been depleted).
These chemical reactions can eventually lead to a build-up of harmful degradation by-products such as weak organic acids. Oil chemistry analysis always includes oxidation, and based on the application, it may also include sulfation and nitration. In some cases, oils may be tested to check for the depletion of specific additives, such as a ZDDP antiwear package.

Oxidation
Oxidation of oil occurs in the presence of heat and air (oxygen). The atmospheric oxygen reacts with the hydrocarbons in the lubricant to form carboxylic acids. These are weak acids, but given sufficient time, the concentration can become high enough to promote severe corrosion of machinery parts.
This process is inevitable and must be monitored. Antioxidant additives are included in almost all formulations in order to protect the lubricant. The additives will readily oxidize before the oxidization of more important oil components. After the depletion of these additives, the properties of the lubricant will be negatively affected.
The oxidation rate varies significantly with temperature, and the presence of contaminants (especially metals) in the lubricant can also affect the oxidation rate. So, keeping the oil dry, clean, and as cool as possible is the best way to manage oxidation.
Nitration
Nitration is a concern in engine oils, especially natural gas engine oils. In the presence of heat, atmospheric oxygen (O2) and nitrogen (N2) can react to form nitrous oxides (NOx). These nitrous oxides interact with the lubricant by forming organic nitrates or being picked up as either insoluble or soluble nitrous compounds.
Premature thickening of the engine oil can be caused by nitration. Typical causes of nitration include improper air-to-fuel ratio, inefficient exhaust of the combustion products, leaking piston seals, and low operating temperatures.
Sulfation
Sulfurous compounds, including sulfur-based acids, can be created by the reaction between heat, oxygen, water and sulfur from base oil or diesel fuel. Usually, these sulfurous compounds are released through exhaust, but some may remain and enter into the engine cavity.
When these acids react with the base stock of the lubricant or the additives in the oil, sulfation will occur. The acids can condense and more readily come into contact with the oil at a lower operating temperature, such as during start up. Sulfation can lead to increased viscosity and the formation of sedimentation, sludge, and varnish.
Methods of Measuring Oil Chemistry
Viscosity
Spectroscopy is the only direct method used to determine nitration, oxidation, and sulfation. Indirect methods are viscosity and impedance measurements. Viscosity is the measure of a fluid’s resistance to flow.
As sulfation, nitration, or oxidation occur in an oil, condensation products are produced that will lead to increased viscosity. Changes in viscosity, irrespective of the cause, is an important parameter to test for, so viscosity testing is a part of all lubricant condition monitoring programs.
There are many methods (ASTM 7279, ASTM D445, etc.), with instrumentation available for both field and lab testing, including the Q3050 portable kinematic viscometer from Spectro Scientific.
Impedance
Impedance testing is another indirect measurement for these properties, which measures a fluid’s dielectric or conductivity properties. These properties are strongly influenced by changes in polarity of the hydrocarbons, which is particularly sensitive to oxidation due to the formation of carboxylic acids.
A change in impedance can also be caused by the presence of other degradation byproducts and contaminants like water. So this test is commonly used for trending the oil’s overall condition.
Infrared Spectroscopy
Infrared spectroscopy uses a detector, a radiative source, and a computer to study the interaction between matter and light.
Sulfation products emerge as peaks in the IR spectrum between 1120 and 1180 cm-1, and nitration and oxidation products emerge as peaks in the IR spectrum between 1600 and 1800 cm-1. As no absolute reference standards are available for sulfation, nitration, and oxidation, the results are always compared to those of new oil.
For instance, if the nitration peak around 1650 cm-1 becomes significantly more intense while sampling the engine oil over a specified time, it means nitration has occurred, potentially due to improper fuel/air ratio.
Test methods are available for both portable field testing and laboratory grade FTIR measurement. ASTM E2412 describes the standard practice for FTIR measurement of these properties. Additionally, specific test methods have been defined for sulfation (D7415), nitration (D7624), and oxidation (D7414).
To monitor oil chemistry in the field, a grating infrared spectrometer like the FluidScan® is used by the ASTM D7889 method. The FluidScan® can be easily operated without any need for experienced technicians.
Advantages:
- Low cost per sample after initial equipment purchase
- Test is quick
- Great for direct trending of oil chemistry properties
Disadvantages:
- Requires expensive equipment

ASTM E2412 – Oxidation and nitration measurement in Crankcase Oils.
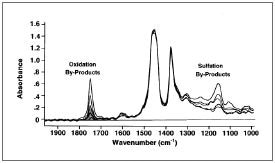
ASTM E2412 – Oxidation measurement in EP Fluids

Thermo Fisher Nicolet FT-IR Spectrometer

FluidScan Q1100
Conclusion
Testing for sulfation, nitration, and oxidation of a lubricant not only enables trending of useful oil life, but also signals failure of engine parts (piston seals), improper operating conditions, or the wrong lubricant for the application. Indirect methods are available to test the ability of a lubricant to perform its function, such as viscosity.
However, infrared spectroscopy is the only direct method that can be used to determine whether sulfation, nitration, or oxidation is happening. FTIR methods and instruments are available for intermediate to expert users in a laboratory setting and the Spectro Scientific FluidScan enables testing for oil chemistry both in the lab and in the field with a portable IR spectrometer.
References
1. http://www.machinerylubrication.com/Read/873/oil-tests
2. http://www.machinerylubrication.com/Read/30020/engine-oil-nitration
3. http://www.machinerylubrication.com/Read/524/natural-gas-engine-oil-analysis
4. ASTM E2412. Standard Practice for Condition Monitoring of In-Service Lubricants by Trend Analysis Using Fourier Transform Infrared (FT-IR) Spectrometry.
5. ASTM D7889. Standard Test Method for Field Determination of In-Service Fluid Properties Using IR Spectroscopy.

This information has been sourced, reviewed and adapted from materials provided by AMETEK Spectro Scientific.
For more information on this source, please visit AMETEK Spectro Scientific.