Nov 13 2018
Researchers have found a novel technique to synthesize furan-2,5-dicarboxylic acid (FDCA) in a high yield from a glucose derivative of non-food plant cellulose, making way for the substitution of petroleum-derived terephthalic acid with biomaterials in plastic bottle applications.
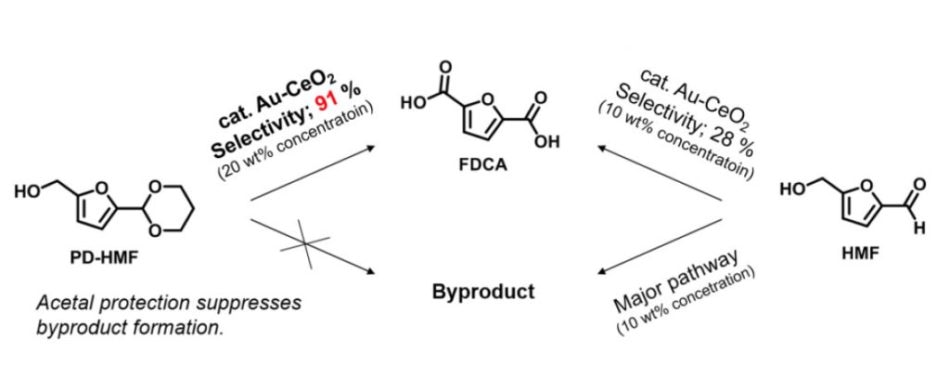 Conventional methods produce by-products making large-scale FDCA production difficult, while this new method yields FDCA efficiently without by-products formation. (Kim M., et al., Angewandte Chemie International Edition, May 14, 2018)
Conventional methods produce by-products making large-scale FDCA production difficult, while this new method yields FDCA efficiently without by-products formation. (Kim M., et al., Angewandte Chemie International Edition, May 14, 2018)
The chemical sector is hassled to create energy-efficient chemical procedures that do not produce by-products, and to use renewable resources wherever possible. Researchers feel that if resources from non-food plants can be used without placing a burden on the environment, it will help preserve prevailing social systems.
It has been stated that numerous useful polymers can be synthesized from 5-(hydroxymethyl)furfural (HMF), the biomaterial used in this research. A high yield of FDCA can be gained when HMF is oxidized in a diluted solution under 2 weight percentage (wt%) with several supported metal catalysts. But a huge stumbling block to industrial application lies with the use of a concentrated solution of 10-20 wt%, which is vital for the efficient and scalable manufacture of FDCA in the chemical sector. When HMF was basically oxidized in a concentrated solution (10 wt%), the FDCA yield was only about 30%, and a large number of solid by-products was formed at the same time. This is because of intricate side reactions induced from HMF itself.
In the research reported in Angewandte Chemie International Edition, a Japan-Netherland research team led by Associate Professor Kiyotaka Nakajima at Hokkaido University and Professor Emiel J.M. Hensen at Eindhoven University of Technology succeeded in subduing the side reactions and creating FDCA with high yields from concentrated HMF solutions (10~20 wt%) minus the by-products formation. Specifically, they first acetalized HMF with 1,3-propanediol to guard by-product-inducing formyl groups and then oxidized HMF-acetal with a supported Au catalyst.
Nearly 80% of 1,3-propanediol used to guard formyl groups can be reused for the following reactions. Furthermore, radical improvement in the substrate concentration decreases the number of solvents employed in the production process. Kiyotaka Nakajima says “It is significant that our method can reduce the total energy consumption required for complex work-up processes to isolate the reaction product.”
These results represent a significant advance over the current state of the art, overcoming an inherent limitation of the oxidation of HMF to an important monomer for biopolymer production. Controlling the reactivity of formyl group could open the door for the production of commodity chemicals from sugar-based biomaterials.
Kiyotaka Nakajima, Associate Professor, Hokkaido University
This research was conducted in collaboration with Mitsubishi Chemical Corporation.
This research received support from Advanced Low Carbon Technology Research and Development Program (JPMJAL1507), Japan Science and Technology Agency (JST), and a KAKENHI, Grant-in-Aid for Young Scientists (A) (15H05556) from the Japan Society for the Promotion of Science (JSPS).