The agri-food industry generates a large amount of waste, which not only results in a loss of market value but also represents a waste of valuable environmental resources. Fruit and vegetable waste could be transformed into a valuable resource with huge economic potential, according to the circular economy concept.
The recovery of bioactive natural compounds from the discarded agro-food materials, on the other hand, must rely on methods that do not produce more polluting waste than the raw biomass disposal itself, are safe, and ensure good revenues for the whole value chain to maintain the green connotation.
Enzyme-assisted extraction (EAE) of biomolecules is one of the safest and environmentally friendly procedures available, and it can be employed as a pre-treatment or as a stand-alone technique to improve the effectiveness of a coupled extraction system.
The use of pricey commercial enzymes, on the other hand, would put a stop to the entire process, jeopardizing the extraction's economic sustainability. Bacillus subtilis (B. Subtilis), a Gram-positive soil bacteria, is one of the most powerful enzyme manufacturers. B. subtilis based growing medium contains a cheap and diverse set of generally recognized as safe (GRAS) enzymatic activities, making it perfect for breaking down the plant matrix and improving bioactive release.
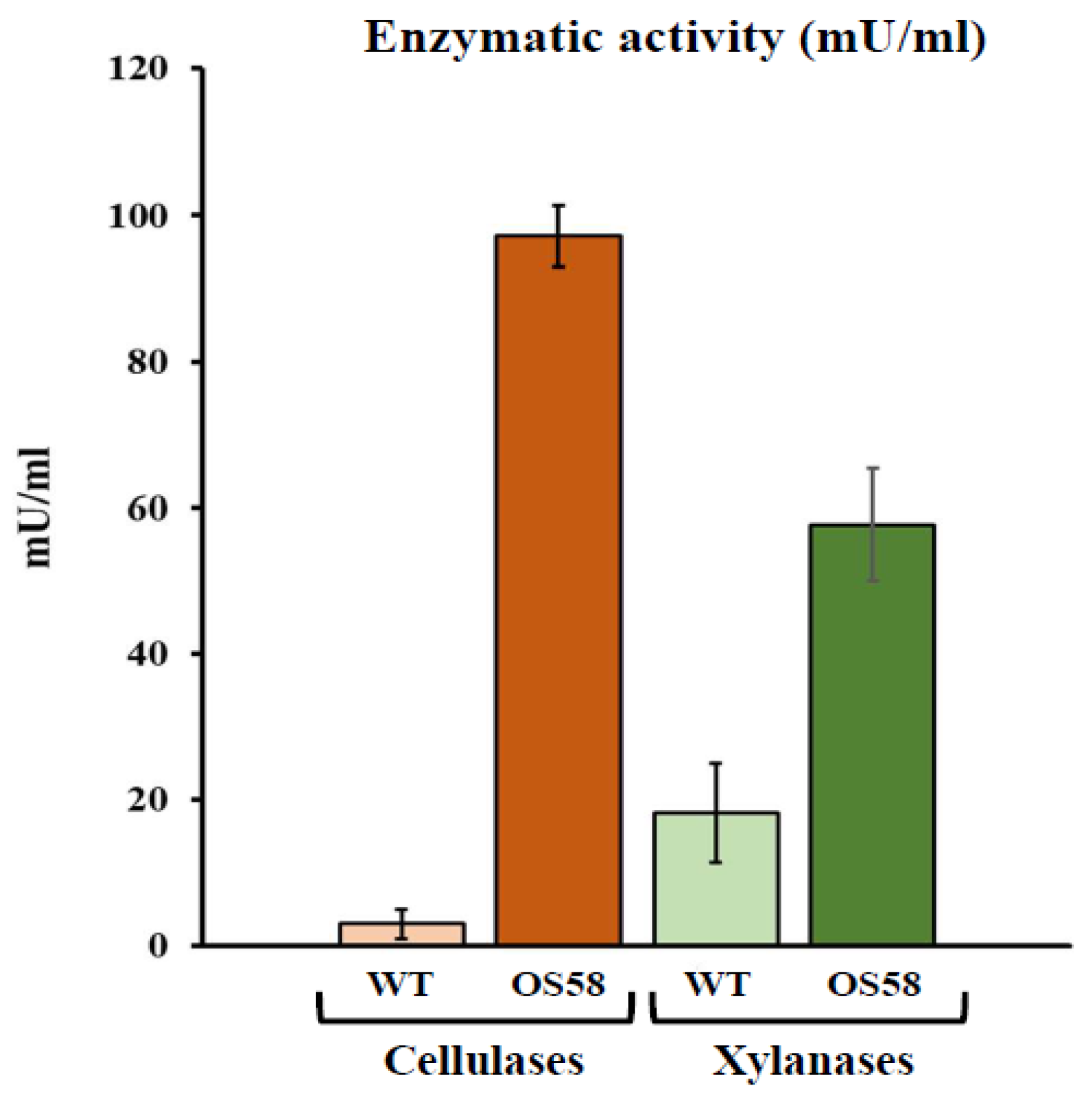
The enzymatic activity of cellulases (in orange) and xylanases (in green) in the WT and OS58 strains (as indicated on the x-axis). Cells were grown in a chemically defined medium for 24 h and assayed with a commercial kit. Enzymatic activity (in mU/mL of spent medium) is reported on the y-axis. Values represent the average of at least five independent experiments. Error bars represent the standard error of the mean (SEM). Image Credit: Doria, E et al., Plants
About the Study
In this study, the authors presented the extraction of bio-actives utilizing isothermal pressurization cycles which were aided by a raw bacterial culture supernatant. A novel B. subtilis strain with the higher secretion of cellulases and xylanases, key enzymes for the breakdown of the plant cell wall, was also employed in addition to a wild-type B. subtilis strain.
The researchers assessed the efficacy of a low-cost EAE pre-treatment approach for the long-term extraction of bioactive components from vegetable waste. The procedure's economic feasibility was based on the utilization of crude culture supernatants of B. subtilis acquired from wild-type (WT) and a novel B. subtilis strain that overproduced cellulases and xylanases (OS58). The extraction procedure was carried out in an aqueous environment, which eliminated the usage of any polluting solvent and ensured the process had long-term viability.
The team utilized cauliflower, i.e., Brassica oleracea L. conv. botrytis (L.) Alef. var. botrytis L., as a substrate, which was derived from a quota of raw products. Two B. subtilis strains were utilized to test the efficacy of the low-cost enzymatic pre-treatment.
The WT strain was identical to the well-used lab strain JH642. The second strain, OS58, was created by genetically engineering the WT strain to improve the intrinsic cellulolytic and hemicellulolytic tendency of B. subtilis. After a 24-hour incubation period, the activities of cellulases and xylanases secreted by the two strains in the growth medium were measured.
The authors prepared a novel B. subtilis strain, namely OS58, and used it as a pre-treatment before cyclic pressurization extraction for the recovery of important secondary metabolites from cauliflower, with the optimal synthesis of at least two enzymes relevant for EAE i.e., cellulases and xylanases. A WT B. subtilis strain and control containing sterile bacterial growth medium were used to compare OS58's performance to that of a WT B. subtilis strain.
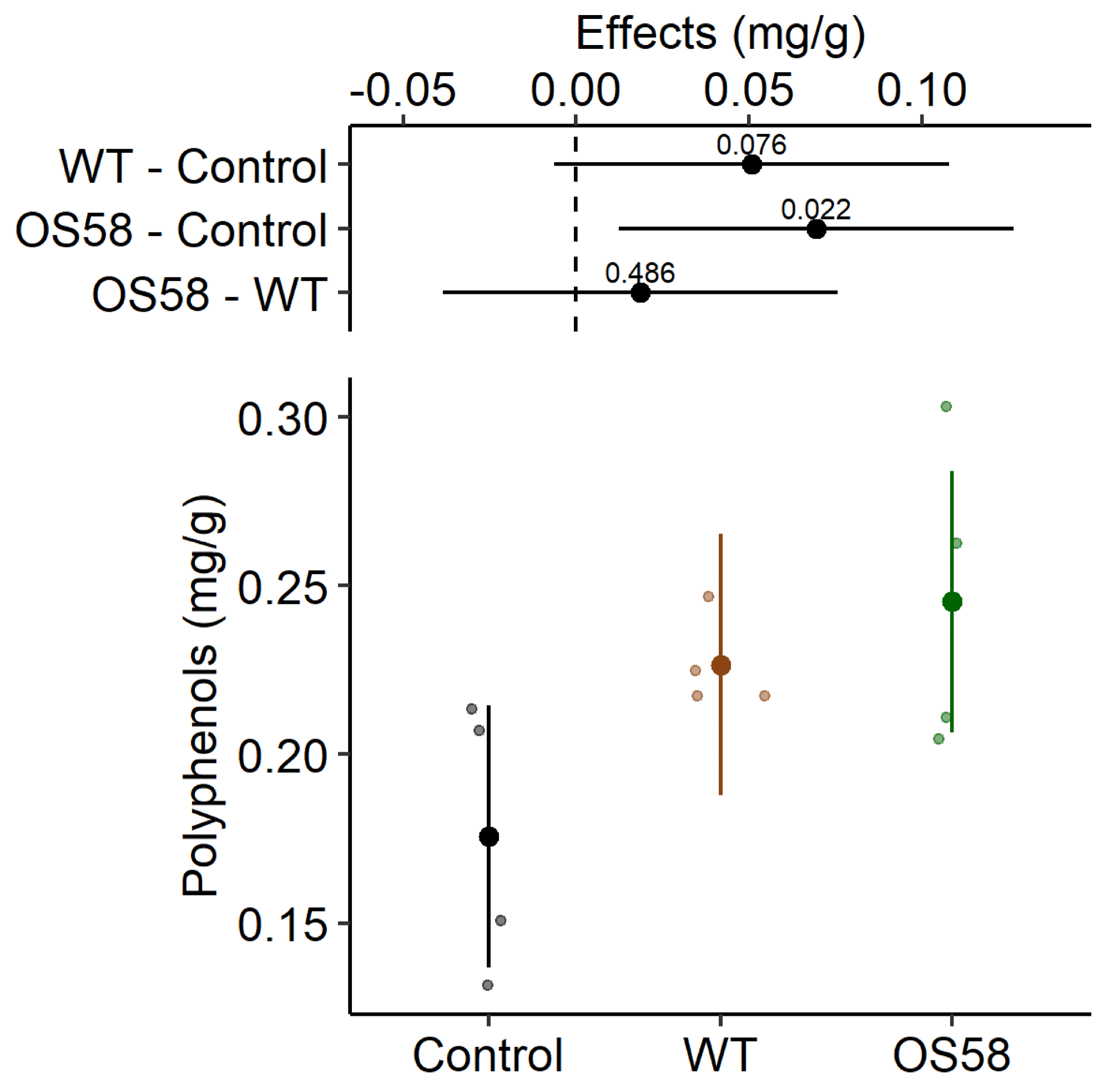
The total polyphenols content (calculated as mg per gram of fresh cauliflowers, before freezing) is reported for Control, WT, and OS58 groups as estimated means and 95% confidence intervals (larger dots and colored lines, respectively) by the linear mixed model (LMM). On the upper side of the plot, treatment effects are reported in mg/g as the estimated means and 95% confidence intervals for each comparison, as difference from the control or between enzymatic treatments, as indicated on the left (degrees of freedom-df = 10.7). The level of significance (p-value) is reported above each horizontal line; the dashed line corresponds to the “no effect” hypothesis. The values reported were obtained from the model using the emmeans package (n = 4, for each group). Image Credit: Doria, E et al., Plants
Observations
The OS58-treated group showed a 1.3-fold improvement in recovery. The enzymes produced by the WT strain were able to release two-fold more chlorogenic acid than the control treatment, while the recovery of chlorogenic acid after treatment with the OS58 enzymatic pool was 2.9-fold greater than the untreated control.
The WT enzymes allowed for two-fold more epigallocatechin gallate, 1.8-fold more epicatechin, and 1.3-fold more epicatechin gallate extraction than the control. The effect of the bacterial pre-treatment was stronger with the optimized OS58 strain, which increased yield by 2.8-fold for epigallocatechin gallate and 2.4-fold for epicatechin when compared to the control.
When compared to the WT strain, the enzyme units detected in OS58 culture broth incremented by over three-fold for xylanases and 30-fold for cellulases. The polyphenols recovered after treatment by the WT strain incremented by 1.3-fold compared to the control; however, when the treatment was performed with the optimized strain OS58, the recovery increased by 1.4-fold compared to the control.
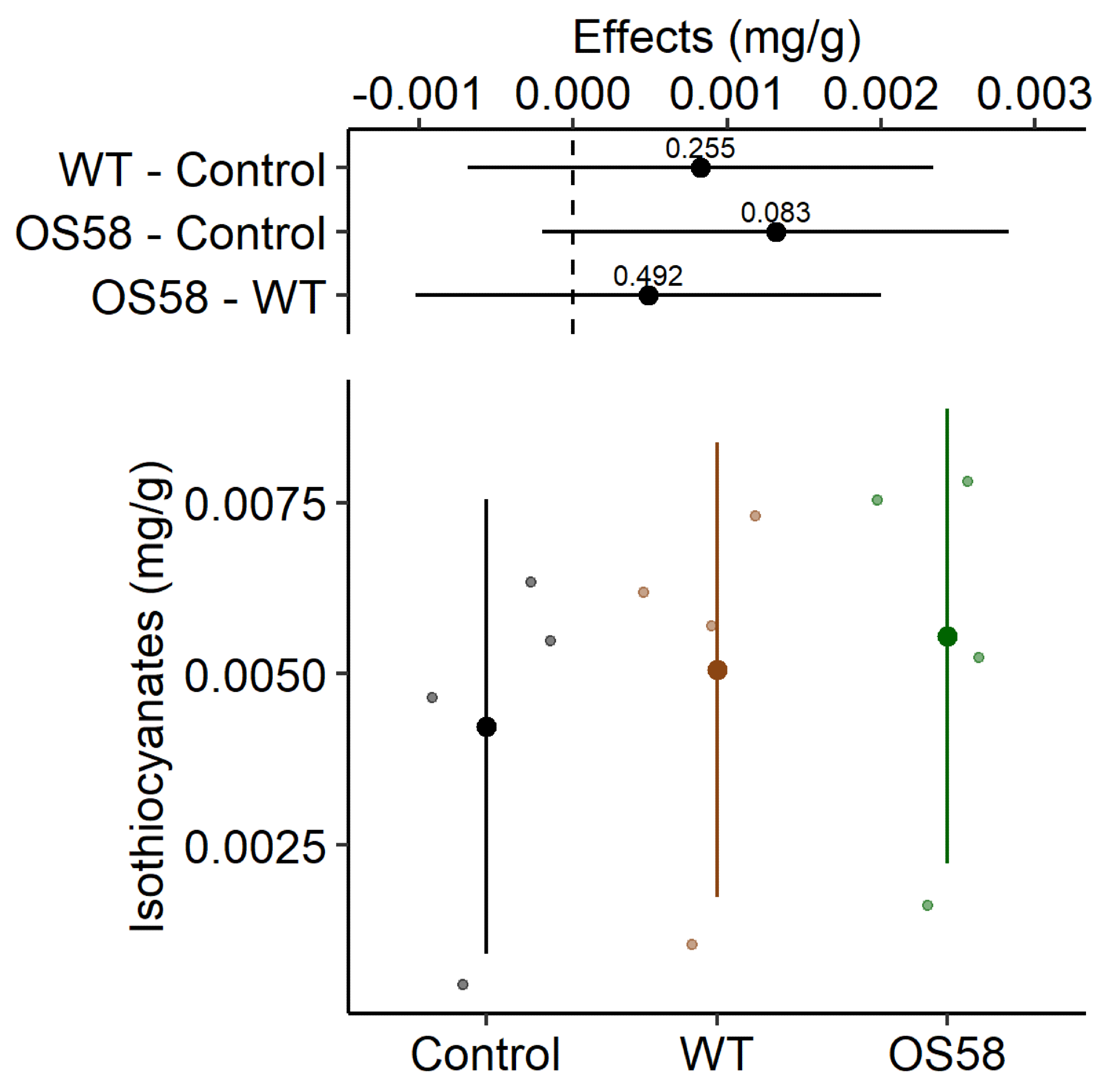
The isothiocyanates content (calculated as mg per gram of fresh cauliflowers) is reported for the Control, WT, and OS58 groups. Image Credit: Doria, E et al., Plants
Conclusions
In conclusion, this study elucidated that the number of cellulolytic enzymes used correlates with the recovery of chemicals, which suggested that pre-treatment with non-purified culture broth effectively enhances the release of bio-actives from the vegetable matrix. The OS58-based sustainable extraction procedure was found to be more effective than the WT strain-based extraction procedure in recovering bio-actives. Impurified bacterial enzymes significantly improved the retrieval of valuable phytochemicals from the matrix in a concentration-dependent manner.
The authors emphasized that the proposed method for recovering bioactive chemicals from food waste is both valid and sustainable.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.