 Interview conducted by Alex SmithAug 5 2021
Interview conducted by Alex SmithAug 5 2021In this interview, AZoM talks to Ed Sprake, Principal Product Manager at Waters, about the work Waters do with ambient ionization in mass spectrometry and the potential that this innovation has.
Could you start by giving our readers a brief introduction to Waters Corporation and its work?
Waters Corporation is the world's leading specialty measurement company. They have pioneered chromatography, mass spectrometry and thermal analysis technology, serving the life, materials and food sciences for more than 60 years.
Waters employs more than 7,000 employees worldwide and operates directly in 35 countries with 15 manufacturing facilities. Waters is committed to customer success and enabling solutions that promote human health and wellbeing.
How widespread is the use of ambient ionization currently and what are some of the challenges of implementing this method?
Ambient ionization is an area with a great deal of interest as it promises faster, more convenient sampling compared to other techniques, such as LCMS or GCMS. There are several ambient direct ionization techniques currently in use, and this is a rich and widespread research area.
Scientists are always looking to improve upon issues that other spectroscopic techniques have failed to address. This is the case across many fields of interest, including food security, forensics, chemical manufacturing and pharmaceuticals.
This continuous improvement means that ambient ionization techniques are often bespoke, built by individual research teams around the world and tailored to fit their existing MS instrumentation.
While this level of customization makes bespoke solutions ideal for their respective applications, their limited applicability has meant that many of these techniques have stayed tied to research-based environments and have not emerged into more mainstream use.
Several techniques have seen commercialization by either MS instrument manufacturers or independent companies. These techniques have seen more widespread use than the more experimental techniques mentioned.
However, even these techniques can be limited in application and exposure since they are designed to fit a range of existing products. This might introduce barriers to adoption due to the operational complexity and relatively high pricing of the base MS instrumentation.
One of the key motivators behind the development of the RADIAN ASAP instrument was to reduce the barriers to adoption of ambient ionization technologies and enable more mainstream use of this type of technique.

Image Credit: Waters Corporation
What are some of the benefits and challenges of ambient ionization techniques?
ASAP (Atmospheric Pressure Solids Analysis Probe) has several benefits as an ionization technique. It is a quick and versatile method that lends itself to rapid analysis with minimal requirements for sample preparation.
The ionization process is essentially analogous to APCI, and it is compatible with many different types of samples and analytes. Since there is no chromatography involved, there is also a minimal need for the use and disposal of expensive solvents.
Conventional ASAP is not a perfect analysis technique, however. It often requires a vertical or near-vertical probe loaded at the top of the instrument. This presents challenges in environments where it is not possible to physically reach the probe on the bench.
Cleaning the ASAP consumable usually involves baking it at a high temperature – a potentially complex process where users must manually alter gas flows and temperatures to optimize consumable cleaning and then reset them to the optimum conditions for analysis afterward.
There are several different implementations of ASAP, but most of them do not physically guide the consumable into the source. Thus, there is a risk of contaminating the source surfaces with sample, therefore introducing the need for background subsequent analyses.
As ASAP technologies are typically add-on peripherals used with existing mixed-use technology, workflows require significant knowledge of that underlying technology to get the best out of the system. This steep learning curve means that only some of the desired functionality is achievable; for example, there could be a lack of flexibility with temperature ramping methods for ASAP.
How has the RADIAN ASAP instrument been developed to meet these challenges?
We looked at a range of implementations and the benefits and challenges that are inherent with ASAP ionization. Then we looked at how we could implement changes to the design of the system hardware and software to improve the functionality.
The RADIAN name is based on a contraction of rapid direct analysis, which sums up what the instrument is designed to do. ASAP (otherwise known as the Atmospheric pressure Solids Analysis Probe) is the technology that we use to generate ions and enable mass analysis. It can also analyze liquid samples, solutions, slurries and even heterogeneous mixtures.
The system is based upon proven and robust technologies. ASAP ionization itself was developed by Chuck McEwen and his group in around 2005. Since then, it has enjoyed widespread use via multiple vendors and types of mass spectrometry technology, including our own Universal Ion Source.
For the RADIAN instrument, we have redesigned the ASAP source to incorporate a horizontal sample loader, which not only makes it easier to use but also fits better with typical sampling and analysis workflows.
The mass analyzer is based on the compact format, single quadrupole technology which we premiered in the Waters QDa in 2013, and has been used in thousands of customer laboratories ever since.

Image Credit: Waters Corporation
We have brought these two technologies together and modified the designs so that RADIAN ASAP can provide a standalone, compact, robust and easy-to-use instrument, which enables rapid, low cost-per-sample analysis of solids, liquids and solutions.
The hardware design is supported by cutting edge informatics tools such as LiveID™, which are designed to provide results quickly and easily with minimal training and knowledge of mass spectrometry.
Could you give our readers an overview of the RADIAN ASAP's standard workflow?
The overall workflow for sampling and sample introduction on RADIAN ASAP can be broken down into four simple steps.

Image Credit: Waters Corporation
The first step is to clean the capillary to remove any background material which may have been picked up from the packaging, or from how the capillary has been handled. To do this, simply load the capillary into the system and use the dedicated bake-out button in the software.
This automatically heats the capillary to 600 °C for one minute, increasing the gas flow and raising the corona current to remove any contaminants which may result in background ions in the analysis.
The second step is to load the sample onto the clean capillary. There are several ways to do this; if the sample is a liquid or in solution, you can pipette a known amount onto the end of the glass capillary with it still sat in the loader, or you can simply dip the end of the capillary into the sample, handling it using the capillary holder.
If the sample is a solid, you can either touch the capillary on the surface of the solid (being careful not to overload it) or you can dissolve a small amount of the solid sample in a solvent, then use one of the two methods for liquid samples.
Once the sample is on the capillary, the holder and capillary can be placed into the loader on the front of the instrument. This third step will automatically start the acquisition.
The instrument will acquire a background spectrum before an instruction from the software (written or via the LEDs on the front of the instrument) will prompt the user to push the sample loader (holding the capillary and sample) into the instrument. If a temperature ramp or step temperature profile is set up, this will automatically start when you push the sample into the instrument.
The fourth and final step is acquiring the results. The LiveID software allows users to match a sample against a library or statistical model in real-time, providing library matches or sample classifications as the analysis is running, depending on the user's workflow.
Some users opt to use automatic data processing, using packages like OpenLynx to process a mass confirmation workflow or IonLynx to generate automatic reports based on ratiometric analysis or for semi-quantitative methodologies using an internal standard.
Can you provide some more detail on how the ASAP ionization technique works?
The source geometry for RADIAN ASAP will be familiar to those with prior knowledge of mass spectrometry techniques. The sample is introduced into the source using a disposable glass capillary from the front of the instrument before a stream of heated nitrogen gas is introduced.
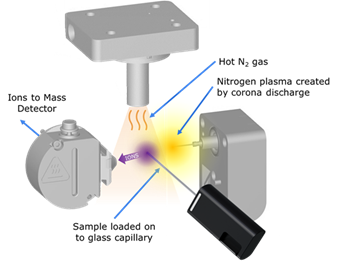
Image Credit: Waters Corporation
This serves two purposes; firstly, it provides a heat source to the glass capillary to volatilize the sample. Secondly, it creates nitrogen plasma in the region around the corona discharge pin, which is the basis of one of the predominant mechanisms of ionization.
The nitrogen plasma indirectly creates ions from the volatilized molecules, which are then guided into the instrument and sorted based on differing mass-to-charge ratios before being detected and converted into a mass spectrum.
What data analysis options are available for use with the RADIAN ASAP?
Once the mass spectrum is produced, there are several options for processing and viewing the data. The choice is largely dependent on what users are looking for from their analysis.
MassLynx is the standard data package that provides instrument control and basic data acquisition. It has the standard tools available to develop methods, and view and analyze data. This can be useful for applications such as reaction monitoring, where users may be looking for a quick method to confirm the reaction progress.
MassLynx includes OpenLynx processing capabilities, allowing users to automatically generate mass confirmation reports and view data in a simple browser format.
Users interested in semi-quantitative analysis or ratiometric analysis should look at IonLynx, an excellent tool for processing non-chromatographic data and presenting results in an easy-to-use and informative format. This can be useful for applications such as chemical formulation monitoring.
The LiveID software enables real-time library matching and real-time chemometric modeling workflows.
What are the key differences between library matching and model building workflows in LiveID?
Library matching and model building workflows seem to have similar uses, but they do ask subtly different questions.
The multivariate statistical modeling approach is typically used to answer the question, "Are these samples similar to my gold standard, or are they different?"
This is ideal for applications such as monitoring food fraud, looking at adulteration or authenticity studies. This approach requires users to build a statistical model using known authentic samples so that LiveID can then compare this data against new samples to identify significant differences.
Library matching, on the other hand, asks whether any of the compounds in a predefined library are contained in an unknown sample. The difference in the two workflows is very subtle, but library matching is a very useful approach for quickly triaging seized drugs for forensic analysis, for example.
A standard library that includes 50 of the most common drugs of abuse is available for the RADIAN ASAP. This is available to download free of charge.
What are some typical uses of the RADIAN ASAP, and how does its design benefit these applications?
RADIAN ASAP includes a redesigned sample introduction system to load the consumables into the source at bench height, meaning it is accessible to everyone. There is also a dedicated single button in the software to bake out and clean new capillaries, flush the source volume with gas and keep the corona pin clean.
The instrument features mechanically guided sample entry into the source and an automated sample door, which ensures that the sample never comes into direct contact with surfaces in the source. This keeps background to a minimum and reduces cleaning requirements.
RADIAN ASAP also gives users the ability to define temperature ramps that are appropriate to the samples being analyzed and the type of analysis being performed.
An intuitive and directed sample insertion workflow guides users through the whole analysis process.
Real-time library matching using RADIAN ASAP and LiveID is ideal for streamlining the workflow for the rapid triage of seized materials, including drugs of abuse. The software's default library contains 50 of the most common drugs of abuse, and LiveID enables users to match an unknown sample against the components in the library.
The analysis is run with a method that simultaneously acquires data at four different cone voltages. Spectra acquired at the lowest voltage typically show ions from the intact species of interest. As the voltage increases, higher levels of fragmentation become evident, essentially creating four spectral fingerprints which are indicative of the species being analyzed. This provides greater specificity than a single mass spectrum.
LiveID matches the data acquired from the RADIAN ASAP against the library in real-time, looking for matches against the spectral fingerprint at each cone voltage. This returns a match score out of 1000, with scores close to 1000 indicating that a compound is likely to be present and lower scores indicating that the compound of interest is unlikely to be present.
This result is displayed just seconds after the sample is inserted into the instrument. This high-speed analysis allows for rapid multiple acquisitions of the same sample to satisfy accreditation. Results can be stored as a PDF file where required.
We are also looking at using RADIAN ASAP in food authenticity and adulteration, using the multivariate statistical modeling workflow that is available in LiveID.
To provide a more specific example, we recently looked at the adulteration of dried oregano with other contaminants such as olive leaves.
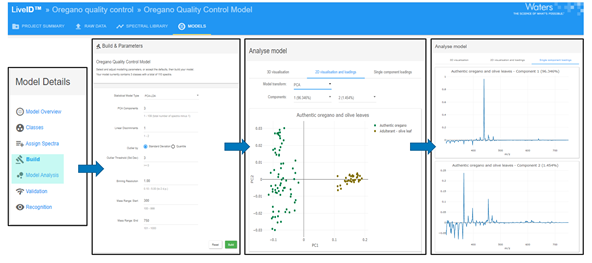
Image Credit: Waters Corporation
To ensure like-for-like analyses, simple solvent extraction was carried out using methanol, and the resultant extract was dipped with the capillary to load the sample prior to analysis. Real-time sample classification was completed against a statistical model using LiveID.
To generate this model, we ran two technical replicates of 35 authentic oregano samples and 18 authentic olive leaf samples in a randomized order over two days and generated a PCA/LDA model using this data.
This provided good separation between the two groups, which was based on three principal components and accounted for more than 96% of the variance in the data. The model was then tested with various adulterants to simulate anticipated fraud scenarios.
A random selection of seven authentic oregano and two olive leaf samples were tested on one of four different instruments by one of three different analysts, using either the dipping or pipetting sample introduction technique. In all cases, the LiveID software returned the correct classification with a 100% confidence score.
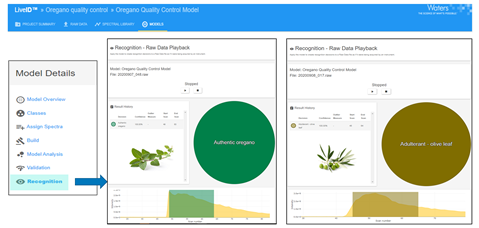
Image Credit: Waters Corporation
What are the benefits of using temperature ramping with the RADIAN ASAP instrument?
Several types of temperature ramping can be applied with RADIAN ASAP. The simplest of these is an isothermal method which can be optimized depending on the analyte. There are also options to perform temperature step methods and ballistic temperature gradients, both of which can be used for different types of samples.
Stepped temperature ramping can be used to help deconvolute complex sample types such as oils or polymer formulations. These typically tend to generate several peaks or steps in the thermal desorption profile. It is also a very useful tool for method development and can help determine the optimal temperature for isothermal work.
Step temperature methods will have slightly longer run times than their isothermal equivalent due to the different temperature steps involved. The other alternative is a ballistic temperature ramp, whereby the temperature is increased very rapidly from a low to a high setting to quickly volatilize the components in the sample.
This method creates less separation across the thermal profile, but it does benefit from a faster acquisition time than the full step methodology.
The use of temperature ramping coupled with real-time data acquisition allows users to monitor reactions in a range of scenarios; for example, looking at degradation markers of samples in applications such as accelerated stability testing or degradation monitoring.
Temperature ramping is also useful for reaction optimization, where multiple analytes are required to assess the progress of a chemical reaction under a variety of different conditions – it is possible to use the temperature ramp feature to optimize the analysis in this kind of scenario.
Once optimized, this method is very quick and very easy to run, enabling rapid decision-making based on the progress of the reaction.
There are also applications for this type of analysis within the pharmaceutical industry, with reaction monitoring being a critical step in the synthesis of new drug candidates. Other common temperature ramping applications include quality testing of raw materials, analyzing time or moisture sensitive components or the analysis of purification fractions.
Simple extraction of the spectra from the total ion chromatogram shows the status of the reaction, with subsequent analyses demonstrating the reaction progress over time.
Despite offering a simple temperature ramping workflow, RADIAN ASAP provides information-rich nominal mass data as the reaction progresses. This information also includes the relative intensities of products and reactants, as well as nominal mass data for any impurities or degradants produced.
What is next for the RADIAN ASAP in terms of product development, new applications and markets?
The current RADIAN ASAP product is a standalone, small footprint, easy-to-use direct analysis system. We based the development of this system on robust technologies and worked to redesign the hardware and the software, both to streamline the workflow and provide extra functionality dedicated to the ambient ionization technique.
The LiveID product library's matching capabilities are brand new for this instrument, and we still have access to a multivariate statistical modeling workflow.
These systems combine to provide excellent real-time results from data, and alternative data processing options are available for users wanting to integrate data on their own or for users who want to use more automated options to generate reports for mass confirmation workflows ratiometric analysis or semi-quantitative analysis.
We continue to ask questions about where we can take this instrumentation in the future and what other applications may benefit from direct analysis. RADIAN ASAP has come a long way in making ambient ionization technologies more accessible, but there are still more things we can do.
Much of where we take the instrumentation next will depend on where people want to use this technology.
For example, do users want to take this instrument outside of the laboratory, and for what purpose? Do we need to automate it further so that there is less manual intervention? Are there different or more appropriate ionization techniques or technologies that we can utilize to provide better results in different circumstances? Are there ways we can start to incorporate other mass spectrometry or analytical technologies into existing workflows to provide better and more accessible results?
As a manufacturer, we can start to answer some of these questions, but I think these answers must come from users, their thoughts and feedback on the uses and the future of this technology. This user input will drive us forward and ensure that we keep producing instrumentation that is useful to so many different industries.
Click here to learn more about Radian ASAP
About the interviewee

Ed has worked for Waters since 2001 in a variety of roles including LC/MS Applications Specialist, Pharmaceutical Marketing, and Product Management. He is currently responsible for the quadrupole-based mass detection technologies including RADIAN ASAP, ACQUITY QDa Detector, and SQ Detector 2 as the Principal Product Manager for Mass Spectrometry.
Prior to Ed’s career at Waters, he worked as an Analytical Chemist at Zeneca Specialties and Avecia in the UK and received an MChem in Analytical Chemistry from U.M.I.S.T. in Manchester, UK.

This information has been sourced, reviewed and adapted from materials provided by Waters Corporation.
For more information on this source, please visit Waters Corporation.
Disclaimer: The views expressed here are those of the interviewee and do not necessarily represent the views of AZoM.com Limited (T/A) AZoNetwork, the owner and operator of this website. This disclaimer forms part of the Terms and Conditions of use of this website.