Jun 12 2017
A form of ultrastrong, lightweight carbon, both electrically conductive and elastic, has been developed by a team including several Carnegie Scientists. A material with such an unusual combination of properties could have a wide range of applications starting from military armor to aerospace engineering.
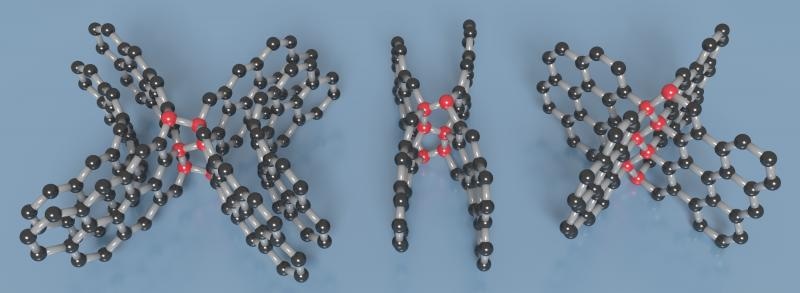 A visualization of the different types of diamond-like linkages (red spheres) formed at curved surfaces or between the layers of graphene (black spheres) in this new type of compressed glassy carbon. Images are provided courtesy of Timothy Strobel.
A visualization of the different types of diamond-like linkages (red spheres) formed at curved surfaces or between the layers of graphene (black spheres) in this new type of compressed glassy carbon. Images are provided courtesy of Timothy Strobel.
Carbon is considered to be an element of seemingly unlimited possibilities. This is due to the configuration of its electrons that allow for numerous self-bonding combinations that result in a variety of materials with different properties. For instance, superhard, transparent diamonds and opaque graphite, which is employed for both industrial lubricant and pencils, are exclusively made up of carbon.
In this global collaboration between Yanshan University and Carnegie—which included Carnegie’s Zhisheng Zhao, Timothy Strobel, Yoshio Kono, Jinfu Shu, Ho-kwang “Dave” Mao, Yingwei Fei, and Guoyin Shen— a structurally disordered form of carbon know as glassy carbon was pressurized and heated by Scientists. The glassy carbon starting material was brought to almost 250,000 times normal atmospheric pressure and heated to approximately 1,800 °F in order to develop the new elastic and strong carbon. Their findings are featured in Science Advances.
Earlier, Scientists tried to subject glassy carbon to elevated pressures at both room temperature, referred to as cold compression and very high temperatures. However, the so-called cold-synthesized material failed to maintain its structure when brought back to ambient pressure and nanocrystalline diamonds were developed under the very hot conditions.
The newly produced carbon is made up of both diamond-like and graphite-like bonding motifs, which leads to the exceptional combination of properties. Under the high-pressure synthesis conditions, disordered layers present within the glassy carbon buckle, blend and then connect in different ways. This process develops an overall structure that in fact lacks a long-range spatial order, but possesses a short-range spatial organization on the nanometer scale.
Light materials with high strength and robust elasticity like this are very desirable for applications where weight savings are of the utmost importance, even more than material cost. What’s more, we believe that this synthesis method could be honed to create other extraordinary forms of carbon and entirely different classes of materials.
Zhisheng Zhao, A Former Carnegie Fellow and current Yanshan University Professor
The other members of the team include: Meng Hu, Julong He, Wentao Hu, Dongli Yu, Hao Sun, Lingyu Liu, Zihe Li, Mengdong Ma, Jian Yu Huang, Zhongyuan Liu, Bo Xu, Yongjun Tian of the State Key Laboratory of Metastable Materials Science and Technology; Yanbin Wang of the University of Chicago; and Stephen J. Juhl of Penn State University.
This research was supported by the National Natural Science Foundation of China, a grant for Distinguished Young Scholars of Hebei Province of China, the Postgraduate Innovation Project of Hebei Province of China, the Defense Advanced Research Projects Agency, EFree—funded by the U.S. Department of Energy (DOE), Office of Science, and the NSF. HPCAT gets support from DOE-NNSA and DOE-BES with partial instrument support from the NSF. APS. is supported by DOE-BES.