Jul 9 2018
Light is used to cure an ever-increasing number of coatings, such as printing inks, varnishes, and even tooth fillings. However, it is not possible to create customized, homogenous polymer networks and the materials are also likely to be fragile, limiting the use of photopolymers in various applications like microelectronics, biomedicine, and 3D printing.
 © Wiley
© Wiley
In the Angewandte Chemie journal, scientists presented a technique which can be used for making homogenously cross-linked, methacrylate-based, customized tough polymers, even at high resolution for 3D printing.
Usually, light-curing is a radical chain polymerization. Light energy is used to split an initiator into radicals. These radicals subsequently attack the monomer, for example, the C=C double bond present in a vinyl group, which creates a new radical that turns out to be the starting point of an increasing growing polymer network by attacking more number of monomers and adhering to them.
More recent techniques to better control radical photopolymerization as well as the products’ material properties are likely to slow the curing process, which is not suitable for 3D printing. For economical production times and high spatial resolution, a short irradiation phase is crucial.
A research team, headed by Robert Liska at the Technical University of Vienna (Austria), has developed a novel method through which tailored methacrylate-based photopolymers can be produced without hindering the curing process. Their success depends on the inclusion of an ester-activated vinyl sulfonate ester, or EVS, which serves as a chain transfer agent. EVS is activated since it effortlessly splits off one portion itself.
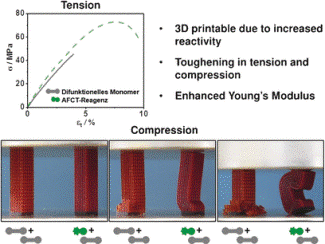
An intermediate is formed when the growing polymer network attacks EVS rather than the next monomer. This intermediate immediately breaks up to create a terminated polymer chain in the network as well as a highly reactive radical, or tosyl radical, which triggers a new chain reaction. The average chain length in the polymer network will be shorter if more EVS is added. The risk of shrinkage cracks at the time of curing process is considerably reduced, because shorter polymer chains stay mobile for a longer time. Unlike traditional chain transfer agents, the polymerization is not inhibited. This is because reversible reaction steps or stable intermediates are not involved. The splitting off of the tosyl radical is preferred.
Using a methacrylate copolymer, the team prepared a scaffold-like sample structure. Separate layers with 50-µm thickness were spatially well resolved. The material is solid yet elastic, extremely homogenous, resistant to impacts, and has high tensile strength. It is possible to adjust these properties by altering the amount of EVS added. The material was extremely brittle without EVS. This latest technique prepares the way for using strong photopolymers in biomedicine, for example as fillings for teeth and shape-memory polymers for tissue growth.