The three new aluminum alloys exhibit high compression fracture strength of ~563 MPa, a high compression yield strength of ~420 MPa, and an elongation above 12%.
What are High Entropy Alloys?
High entropy alloys (HEAs) are defined as alloys of five or more metallic elements in equimolar or near-equimolar concentrations between 5 and 35 atomic%. These alloys exhibit excellent mechanical properties, corrosion-oxidation resistance, and wear behavior. The main goal of this approach is to stabilize a single-phase solid solution (SS) by reducing Gibbs free energy to form stable phases instead of brittle intermetallic compounds (IC).
HEAs are composed of low-density elements, known as lightweight HEAs (LW-HEAs), such as aluminum (Al) or titanium (Ti) alloys. Al-based alloys have received considerable attention, especially in the automotive and aerospace industries, due to their lightweight, formability, and machinability.
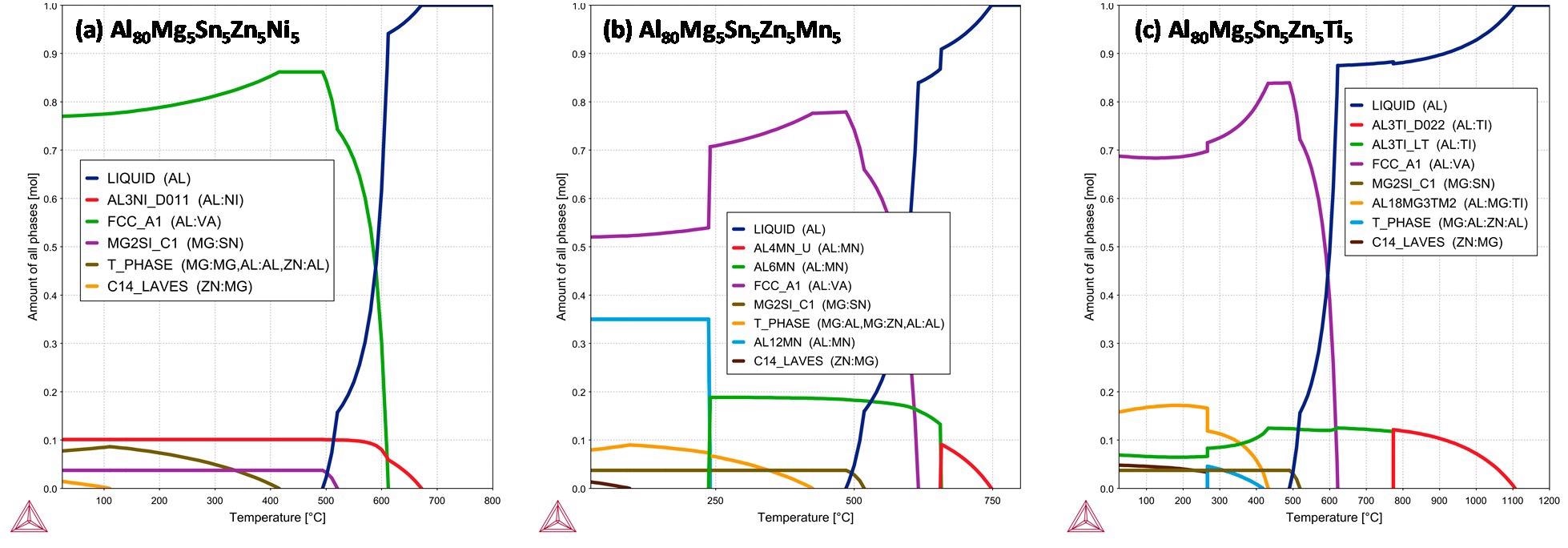
Equilibrium phase diagrams in function of temperature of the selected alloys (a) Al80Mg5Sn5Zn5Ni5; (b) Al80Mg5Sn5Zn5Mn5 and (c) Al80Mg5Sn5Zn5Ti5. Image Credit: Sanchez, J et al., Metals
Although the initial concept for obtaining single-phase SS in HEAs was to maximize the configurational entropy, till now, only Al20Li20Mg10Sc20Ti30 LW-HEA avoided the formation of brittle IC and displays a single-phase SS microstructure. LWHEAs are hard, brittle, and strong multiphase materials due to large precipitation and/or solution hardening.
These alloys are also called low-density multi-principal elements alloys or lightweight complex concentrated alloys (CCAs). The SS phase formation in HEAs can be investigated using Hume-Rothery criteria and thermodynamic parameters. These parameters commonly include the enthalpy of mixing in the liquid phase (ΔHmix), atomic size difference (δr), Pauling electronegativity difference (Δχ), and the Ω-parameter.
About the Study?
In this research, ingots of three novel CCAs viz. Al80Mg5Sn5Zn5Ni5, Al80Mg5Sn5Zn5Mn5, and Al80Mg5Sn5Zn5Ti5 are developed via gravity permanent mold die casting process. First, pure Al is melted in an alumina crucible inside an induction furnace. Then tablets of Al-Ni, Al-Mn, and Al-Ti containing 75 wt.% of Ni, 80 wt.% of Mn, and 75 wt.% of Ti, respectively, are added separately into them. Finally, Sn, Mg, and Zn are added to all the molten alloy samples and held between 776 and 795 ℃ for 10 min to reach complete dissolution followed by pouring into pre-heated steel molds to obtain ingots.

SEM image and EDS elemental mapping of the as-cast Al80Mg5Sn5Zn5Ni5 alloy. Image Credit: Sanchez, J et al., Metals
Observations
The restrictive values previously proposed for HEAs for the stabilization of disordered SS of alloys containing low-density elements are -1 kJ/mol ≤ ΔHmix ≤ 5 kJ/mol, δr < 4.5%, Ω-parameter > ~10, and Δχ ≤ 0.175.
From the measured thermodynamic parameters of developed three Al alloys, it is evident that the very restrictive values of ΔHmix between -1 and 5 kJ/mole to avoid the formation of ICs in LW-HEAs are very difficult to satisfy. Also, only the Al80Mg5Sn5Zn5X5 system satisfies the proposed criteria of δr below 4.5% and Δχ equal or below 0.175 to form SS, whereas all the obtained values of Ω-parameter are very far away from the values considered favorable for the formation of SS in LWHEAs. The Al80Mg5Sn5Zn5X5 system is lightweight than other similar Al-based alloy systems.
From the equilibrium phase diagrams and thermo-physical parameters for phase formation in HEAs, all the alloys are found to be composed of a mixture of SS with ICs microstructures. All microstructures are composed of a major α-Al phase enriched with Zn and Mg, a cubic Mg2Sn intermetallic phase, and an unanticipated hexagonal Zn phase.
The Al-alloys containing Ni and Mn formed acicular phases, whereas the addition of Ti forms a globular phase. This is reflected in the better mechanical properties of Al80Mg5Sn5Zn5Ti5 alloy over alloys containing Ni and Mn. Also, Ni, Mn, and Ti affected the grain size of the alloys differently, the Mn-containing alloy has the finest microstructure followed by Ni-containing alloy and Ti-containing alloy.
This is related to different precipitation temperatures of the Al3Ni, Al6Mn, and Al3Ti phases. The equilibrium phase diagrams of the alloys revealed that Al3Ti is the compound with the highest precipitation temperature of 1108 ℃.
The formation and growth of this phase at high temperatures have not allowed it to act as a grain refiner, as is common in commercial aluminum alloys. In contrast, Al3Ni with precipitation temperature of at 672 ℃ and Al6Mn with precipitation temperature of 749 ℃ is closer to the formation of the α-Al matrix (precipitation temperature around 610 ℃), thus they are more efficient as grain refiners.

SEM image and EDS elemental mapping of the as-cast Al80Mg5Sn5Zn5Mn5 alloy. Image Credit: Sanchez, J et al., Metals
Conclusions
In this study, three new Al-based CCAs are fabricated with very low densities through a standard permanent mold gravity die casting process. All the alloys are found to be composed of a mixture of SS with ICs microstructures. Al-Ni and Al-Mn phases are more efficient grain refiners than Al-Ti phases.
The formation of unexpected hexagonal Zn phase indicates the deviation between CALPHAD predictions and experimental results, which is attributed to partial oxidation of Zn during the fabrication process.
This study further suggests taking the effect of different heat treatments in the mechanical and tribological properties into consideration.
Reference
Sanchez, J., Pascual, A., Vicario, I., Albizuri, J., Guraya, T., Galarraga, H., Microstructure and Phase Formation of Novel Al80Mg5Sn5Zn5X5 Light-Weight Complex Concentrated Aluminum Alloys. Metals, 2021, 11(12), 1944. https://www.mdpi.com/2075-4701/11/12/1944
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.