The world is warming due to human activity, with increasingly extreme weather events, forest fires, and flooding all reported over the past decade. Rapidly decarbonizing industry and removing atmospheric carbon dioxide is crucial to avoid the worst effects of climate change and keep the world in line with international agreements to reach net-zero carbon emissions by 2050.
Carbon capture and storage (CCS) technologies have been widely proposed as a key strategy to reduce atmospheric carbon dioxide or recovered carbon dioxide from power plants and industrial activity and either reuse it or sequester it deep underground. The main storage locations that have been proposed for recovered carbon dioxide are geological reservoirs such as undeveloped deep coal seams, gas and oil reservoirs, and deep brine aquifers.
Methods to trap and remove carbon have been investigated in recent studies, with solubility trapping emerging as a safe and effective technique. In this method, CO2 is dissolved in water. This creates a CO2-rich brine, also termed live brine, which is denser than the original water and sinks to the bottom of the reservoir where it can be easily recovered. However, sequestering this brine in coal seams is problematic as it has a highly acidic pH of 3-4, which means it can chemically react with the coal.
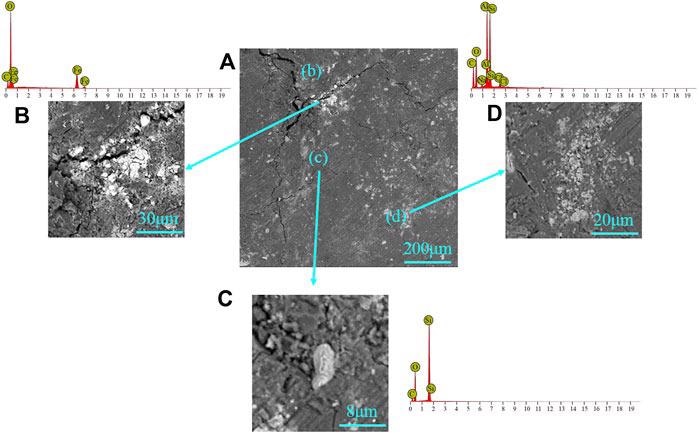
SEM-EDS for the mineral in the coal sample: (A) coal matrix, (B) siderite, (C) quartz, and (D) kaolinite. Image Credit: Li, X et al., Frontiers in Energy Research
Studying the Chemical Reaction of Coal and CO2
Studying the chemical reactions between CO2 and coal is important to characterize the effects of carbon sequestration and storage in coal seams. Whilst there have been extensive studies on these chemical reactions in the context of acid stimulation, the body of research on quantitative micro-pore scale analysis is limited.
Acid stimulation reactions lead to the opening and interconnection of the cleats, which are natural opening-mode fractures, in coal seams. Cleats account for much of the porosity and permeability of coalbed gas reservoirs and have a significant effect on the success of engineering projects and the overall stability of coalbeds.
Studies have demonstrated that the magnesite, dolomite, and calcite dissolved in the CO2-rich brine causes the opening of originally closed or semi-closed pores. Additionally, the mechanical properties of coal can be changed by the dissolved minerals filling the pores and cracks in the seam.
Current research has mainly focused on the qualitative description of geochemical changes such as mineral changes and element migration. Scanning Electron Microscopy (SEM) has been widely employed to study the evolution of coal cleats under these conditions and to analyze the cleat geometry of coal. Studies have observed the cleat morphology using SEM and presented its nanoscale mechanical effects and have described pore connectivity and types in coal.
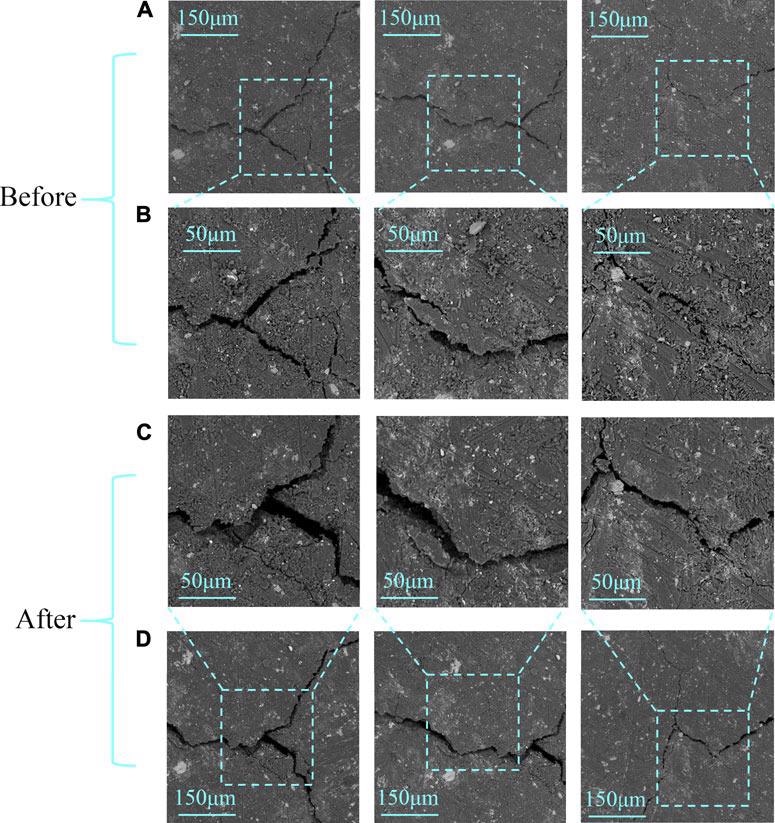
SEM images before (A,B) and after (C,D) the reaction; rows (B,C) are in high resolution, while rows (A,D) are in low resolution. Image Credit: Li, X et al., Frontiers in Energy Research
The Study
Microstructure changes affect the porosity and permeability of coal and the overall mechanical properties of the material, but research is lacking in current literature. The study published online in Frontiers in Energy Research has investigated microstructural changes caused by CO2 sequestration in coal seams by using a reactor under simulated high-temperature and high-pressure conditions.
To analyze and evaluate the microstructural changes caused by the influence of CO2-saturated brine on coal, SEM and energy dispersive spectroscopy (EDS) were employed by the authors. The changes in specific minerals and the microstructure were identified and imaged before and after the chemical reaction.
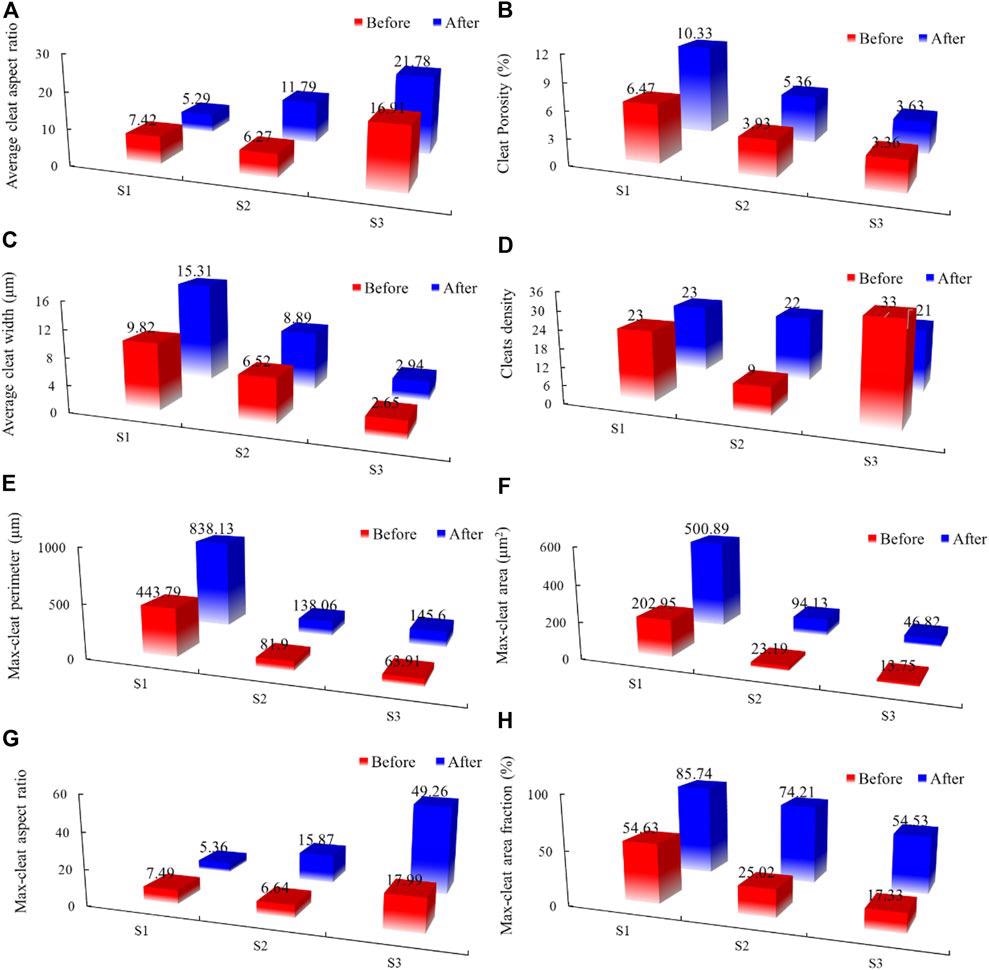
Histogram of the variation of cleat parameters in coal: (A) total aspect ratio; (B) porosity; (C) average width of all cleats; (D) fracture density; (E) perimeter; (F) area; (G) aspect ratio of maximum cleat; (H) the maximum cleat fraction. Image Credit: Li, X et al., Frontiers in Energy Research
Three samples were obtained from the Collie Basin, which is located 150 km southeast of Perth in Western Australia. Covering around 230 km2, this is one of the largest coal basins in the area. The CO2-saturated brine was obtained by injecting CO2 into deionized water with 5 wt% NaCl dissolved in it. Samples were polished and imaged with SEM at room temperature and pressure and then placed in the reactor and vacuumed to remove all air.
Temperature and pressure in the reactor were kept at 50 oC and 20 MPa and constantly monitored. After 24h, the sample was removed and vacuum-dried before analysis was performed.
Results of the analysis revealed several microstructural changes. These changes include a volume fraction change caused by mineral dissolution at the edges of the cleat, leading to isolate cleat connection an increase in maximum cleat size from 25 to 72.4%. This volume fraction change increased the storage space available for CO2 storage and increased coal seam permeability.
Overall, the authors have stated that the microstructural changes of coal caused by the injection of CO2-saturated brine should be measured on the microscale and combined with quantitative analysis to improve the reliability and accuracy of carbon dioxide storage in deep coal seams.
Further Reading
Li, X et al. (2022) The Influence of CO2 Saturated Brine on Microstructure of Coal: Implications for Carbon Geo-Sequestration [online] Frontiers in Energy Research 10 | frontiersin.org. Available at: https://www.frontiersin.org/articles/10.3389/fenrg.2022.802883
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.