Closed-vessel microwave synthesis is a highly effective technique for synthetic processes today. Since the technology is not restricted to a solvent boiling point, it can be applied to a much greater range of reaction sequences. Research has moved from days and hours to minutes mainly due to the ability to operate at higher pressure and temperature values.
The first key advancement with a moderate temperature and medium pressure capability are single-mode microwave reactors. Multi-mode reactors in comparison to their single-mode counterpart allow multiple reactions to be carried out with a single operation.
There are drawbacks in both these technologies, single-mode units are limited in scale and though multi-mode systems work on a larger scale and run multiple reactions, the technology doesn’t allow any change in reaction conditions such as solvent, catalyst and large variation in reactants without compromising the efficiency of the chemistry.
Single Reaction Chamber Technology
A new approach to microwave synthesis is single reaction chamber technology. As the name implies, the SRC is a large, pressurized stainless steel reaction chamber into which all reactions are placed and performed simultaneously. The pressurized chamber in SRC serves as the reaction vessel and the microwave cavity and the reaction vessel, enabling the intensity and distribution of the microwave energy to be optimized with the shape and size of the reaction vessel.
Even heating is ensured by this design and the need to rotate reactions during a method is eliminated. Since reactions are placed in a pressurized vessel, individual pressure vessels are not required. Reagents are added to disposable glass vials with the appropriate solvent and placed in a rack similar to an auto sampler tray. It is possible to replace glass with Teflon for specific reaction media.
Vial caps prevent condensation from the rood of the chamber into the reactions. The rack is arranged in the chamber roof and connected to a stirring motor to provide a method for stirring all reactions as shown in Figure 1.
The reaction chamber is sealed automatically and pre-pressurized with argon or nitrogen to a desired pressure. This makes sure there is no loss of volatile reaction components, no boiling or cross-contamination during the course of the method. The increase in temperature during the microwave program causes an increase in pressure in the chamber hence boiling does not take place.

Figure 1. SRC - SynthWAVE.
Benefits of SRC
The benefits of SRC are:
- It can run multiple parameters or reactions simultaneously.
- It operates at very high temperature and pressure, up to 300°C and 200 bar thus allowing a wider range of reaction types to be successfully completed without several set-up configurations.
- Furthermore, since all reactions are in the same vessel, nearly any reaction type can be processed simultaneously.
- The need for batching C-C couplings away from simple substitution reactions is eliminated
- Several stoichiometry combinations and element changes can be modified for inorganic hydrothermal syntheses in a single run, saving weeks of research labor. Control of every reaction is made possible by direct pressure and temperature control.
The high-pressure capability in the reactor design enables additional gas inlet ports as shown in Figure 2 to open reaction flexibility to reactive gas additions and in situ sampling.

Figure 2. Gas addition.
SRC Technology in Operation
SRC operation is highly simple and automated. Figure 3 describes the process.
The process involves the following steps:
- Reactants are weighed into vials with a stir bar, solvent is added and the reactions are placed on a rack.
- The rack is fitted to the chamber top with a mechanical stirrer, which is lowered automatically into the chamber.
- Uniform temperature conditions are achieved by positioning the reactions in a pool of microwave absorbing material liquid (several options are available).
- The chamber is secured, and the chamber is pre-pressurized with N2 to 40 bar.
- The microwave program used is normally the same for any reaction type and includes time -to-temperature (ramp time) and time-at-temperature (hold time).
- When the program stops, a circulating water-cooling jacket rapidly cools the chamber, pressure is gently released, and gases and vapors are directed away to exhaust.
- Controlled-cooling to specific temperatures and times can also be used as a method step for materials research and as a tool for crystal formation. The sealing clamp is then released, and the sample rack automatically lifts clear of the chamber as shown in Figure 4.
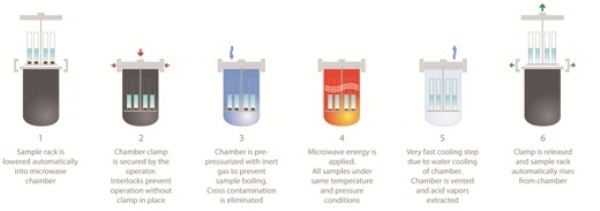
Figure 3. Sequence of SRC Operation.

Figure 4. Post reaction.
Early Illustration of Reaction Examples
Equation 1 shows an example of a difficult hydrogenolytic dearomitization of an aromatic ring containing no heteroatoms as a challenging organic transformation.
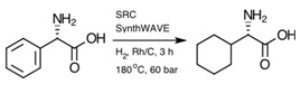
Equation 1. Milestone in collaboration with Eberhard Heller at University of Wurzburg.
The Pd catalyzed alkoxycarbonylation reaction with CO with a range of starting materials is a recent example of multiple reactions run simultaneously under a reactive gas atmosphere as shown in Equation 2.
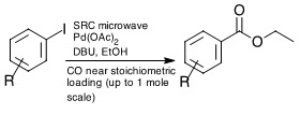
Equation 2. N. Leadbeater et al. Org. Process Res. Dev. 2009 13(3), 634-637.
A recent example of modifying a single-walled carbon nanotube is a good example how traditional organic chemistry is being applied today. The following example using SRC technology provides the functionalization of SWCNTs with a 1,3-cycloaddition of carbonyl Ylides and is shown in Equation 3.
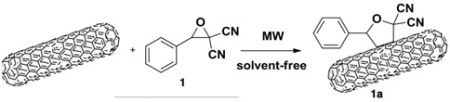
Equation 3. G. Cravotto Synlett 2012 23, 1459-1462.
Conclusion
Most of the challenges of traditional microwave designs are overcome by a new SRC microwave synthesis technology. Early examples illustrate new capabilities for organic and materials science synthesis, including higher temperature and pressures, simple and safe capability of reactive gas additions, rapid or time-controlled cooling.

This information has been sourced, reviewed and adapted from materials provided by Milestone.
For more information on this source, please visit Milestone.